Welcome to the Topham Lab
Viruses that infect the respiratory tract are responsible for extensive morbidity and mortality in human populations worldwide. Influenza virus is a particular concern because of its ability to periodically cause deadly pandemics as occurred in 2009 after the spread of a swine-origin H1N1 virus to humans. Novel avian influenza viruses such as H5N1 and H7N9 continue to cause sporadic cases of severe disease in humans and are an ongoing pandemic threat. The COVID-19 pandemic caused by SARS-CoV-2 coronavirus is our newest threat. An understanding of how the immune system controls influenza, coronavirus, and other respiratory viruses and provides long-term protection is critical for the rational development of effective vaccination and treatment strategies.
Studies in our lab are primarily aimed at understanding the multiple roles of virus-specific B and T cells in determining the outcome of viral infection of the respiratory tract. In particular, we are interested in the character, longevity, and protective capacity of B and T cell memory induced by infection and vaccination. A large component of work in the lab focuses on the response of the human immune system to infection and vaccination; other work uses a variety of animal model systems to investigate basic immunological mechanisms. This work will identify strategies for engineering new antivirals and improving vaccines.
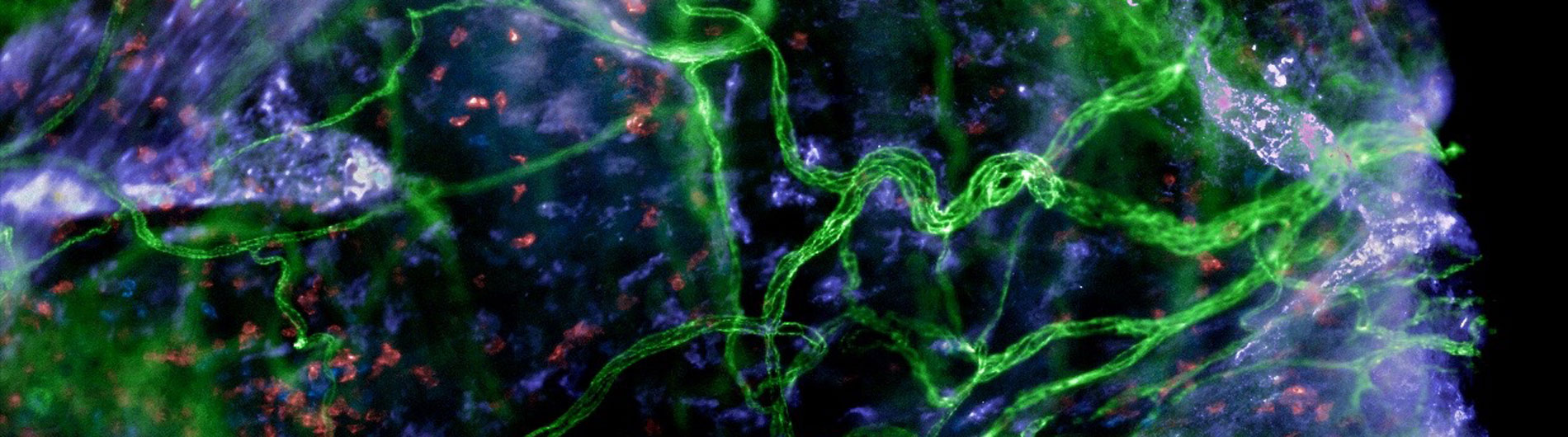
Tissue Resident Memory (TRM) CD8 T cells are a special non-lymphoid tissue resident memory cells that are established after an infection but do not recirculate in the body. They are present in all barrier tissues such as the respiratory tract and provide rapid immune protection during re-encounter with the same or variant viruses. We study how they form, their function, motility, and genetic programming using a combination of imaging, immunology, and genomics.
Many projects involve strong collaborative interactions within centers at the University of Rochester that focus on immunity to respiratory pathogens. These centers include the Respiratory Pathogens Research Center (RPRC), the New York Influenza Center of Excellence (NYICE), and the University of Rochester Genomics Research Center (URGRC).

David J. Topham, Ph.D.
Principal Investigator
Projects
View All ProjectsPublications
View All Publications- Single cell analysis of neonatal naïve CD8α + T cells reveals novel subsets bridging the innate-adaptive spectrum.; bioRxiv : the preprint server for biology. 2026 Mar 09.
- Anti-IsdB Antibody-Secreting Cells Found in S. aureus-Infected Periprosthetic Bone Marrow: Case Series of Total Hip Arthroplasty Patients.; Medical research archives; Vol 14(1). 2026 Jan.
- Association of disease severity and genetic variation during primary Respiratory Syncytial Virus infections.; BMC medical genomics; Vol 17(1), pp. 165. 2024 Jun 19.
- MousebreedeR: A novel software to assist in the design of breeding schema for complex genotypes of experimental organisms.; Journal of open source software; Vol 9(98). 2024 Jun 04.
- Correction for Piepenbrink et al., "Highly Cross-Reactive and Protective Influenza A Virus H3N2 Hemagglutinin- and Neuraminidase-Specific Human Monoclonal Antibodies".; Microbiology spectrum. 2024 Jan 30.
Affiliations
- David H. Smith Center for Vaccine Biology and Immunology
- Microbiology & Immunology
- Immunology, Microbiology, and Virology Ph.D. Program
- Respiratory Pathogens Research Center (RPRC)
- New York Influenza Center of Excellence (NYICE)
- Center for Biomedical Informatics (CBI)
- Program for Advanced Immune Bioimaging
- Health Science Center for Computational Innovation (HSCCI)
News
December 2, 2024
URMC conducts research on possible COVID-19 nasal booster
October 16, 2024
Future COVID Boosters Could Be Nasal: URMC Researchers Lead Study on Next-Gen Vaccines
June 20, 2024
What Is H5N1 Bird Flu, How it Spreads to Humans, and Safety Concerns
September 19, 2023
A Game Plan for Timing Your Flu, Covid and RSV Shots This Fall with Commentary by David Topham
Contact Us
Topham Lab
KMRB 3-9852
601 Elmwood Ave,
Rochester, NY 14642
(585) 273-2452