URMC / Labs / Sant Lab / Projects / Immunodominance in CD4 T Cell Responses
Immunodominance in CD4 T Cell Responses
In specific immune responses pathogens or to protein antigens, T lymphocytes only respond to a limited number of peptide epitopes from the immunogens. These peptides are termed immunodominant. Considering the diversity in the T cell receptor repertoire and the number of peptides contained complex proteins and pathogenic organisms, it is striking that T cells apparently react to such a limited number of peptide epitopes, while seemingly ignoring the others. Our experiments seek to understand the elements in vivo that dictate the narrowed selection of specificities in CD4 T cells during protective immune response, and the competitive events that shape selective recognition of foreign and pathogenic organisms and proteins.
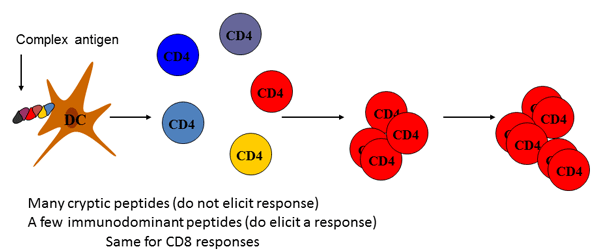
Immunodominance during in vivo responses to CD4 T cells. Skewing of T cell response to a select subset of peptides; where dominant peptides are recognized by a majority of T cells and cryptic peptides can bind MHC and elicit response as a peptide, but are not detected in response against intact antigen.
Our recent studies suggest that one of the key parameters that are selected for in CD4 T cell responses is the ability of peptides to bind in a stable way to the MHC class II molecule. By constructing recombinant proteins that bear antigenic peptides that have altered ability to bind to the presenting class II molecule, we have been able to demonstrate a causative relationship between the kinetic stability of peptide: class II complexes and the ability of that peptide to recruit CD4 T cells. This finding suggests that one can rationally focus the CD4 T cell response toward or against desired peptides in an protein vaccine. One of our major long-term goals is to apply the knowledge that has thus far been gained in animal models towards rational human vaccine design.
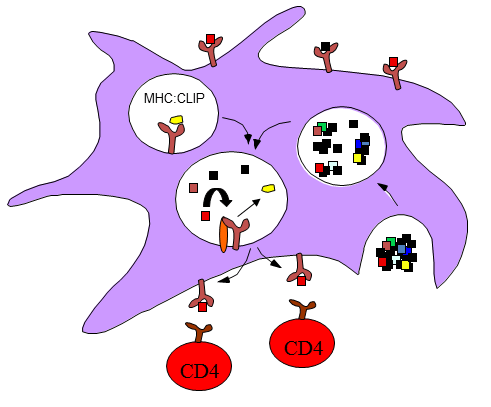
Stable binding of peptides to MHC class II molecules; increased initial epitope density on the surface of APC.