URMC / Labs / Sant Lab / Projects / The Role of DM in Selecting Epitopes for CD4 T Cell Responses
The Role of DM in Selecting Epitopes for CD4 T Cell Responses
The MHC-encoded DM molecule is a critical component of the class II presentation pathway and catalyzes peptide loading onto class II molecules by binding to the class II protein and stabilizing a conformation that promotes peptide exchange. Early studies on endogenous antigen presentation by MHC class II molecules by our laboratory and other laboratories were the first that suggested that DM functions as a peptide exchange protein and can act as either a positive or negative regulator of the cell surface expression peptides:class II complexes and thus the ability of these complexes to recruit and promote expansion of the CD4 T cells. This observation prompted us to revisit the phenomena of immunodominance and crypticity in CD4 T cell responses. We speculated that cryptic peptides might fail to elicit T cell responses when introduced as part of an intact antigen because within endosomal compartments, they are removed from the MHC class II presenting molecule by DM.
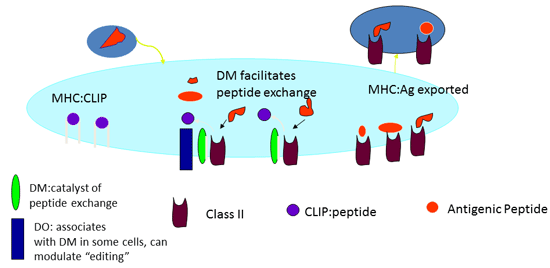
"MHC class II : Peptide" represents the end product of an exchange reaction in endosomal compartments of antigen presenting cells: DM modulates this exchange = "DM editing". Our transfection experiments indicate that within antigen presenting cells, DM promotes accumulation of some peptides and "strips off" other peptides from class II. Those peptides that bind to class II with great affinity (off rates >150 hours) are promoted by DM. This property of a peptide can be rationally modified.
To investigate this, T cells specific for cryptic and immunodominant peptide epitopes were collected for analyses and studied using class II positive APC that differed only in their expression of DM. Thus far, we have observed a complete concordance between DM effects and immunodominance. All immunodominant determinants are enhanced in their presentation by DM expression in APC, while all of the cryptic epitopes are antagonized by DM function within the APC. This relationship between immunodominance in DM editing provides a novel mechanism to explain immunodominance in vivo. Because the priming APC in vivo is a DM-positive dendritic cell, DM editing is likely to antagonize or completely eliminate the presentation of the cryptic peptides on class II molecules. These complexes will therefore be unavailable to recruit CD4 T cells. Through a series of molecular and cellular experiments, we have found that the parameter that dictates the effects of DM on accumulation of peptide class II complexes at the cell surface is the strength of interactions between the peptide and the MHC class II molecule. This property can be intentionally modified using molecular techniques to change anchor residues in the peptide and thus the specificity of the CD4 T cell response can be altered by harnessing the editing property of the DM protein.
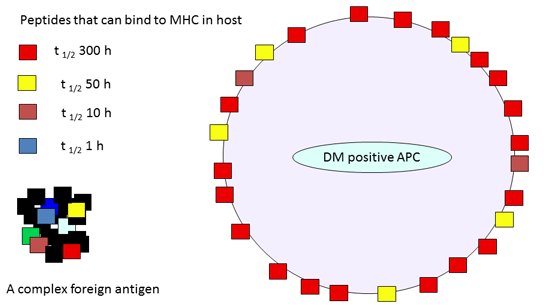
One major mechanism underlying the link between the intrinsic kinetic stability of peptide: MHC and immunodominance = initial epitope density on the surface of the priming antigen presenting cell (APC).