Pediatric Research Newsletter
June 2017
Candidate Vaccine Antigen Identified for Pneumocystis Pneumonia
By Meghan Plog M.S.
 Francis Gigliotti M.D.
Francis Gigliotti M.D.The lab of Francis Gigliotti, M.D., Professor of Pediatrics and Microbiology and Immunology and his colleague Terry Wright, Ph.D., Associate Professor of Pediatrics and Microbiology and Immunology is one step closer to developing a possible vaccine for Pneumocystis pneumonia (PcP), which would be a major breakthrough for prevention of this potentially fatal disease. Immunization against Pneumocystis using a variety of immunogens has been studied for the past 35 years, but to date, none have been particularly successful in animal models of Pneumocystis pneumonia. Dr. Gigliotti’s research group at the University of Rochester, however, has recently published new findings in the journal Infection and Immunity that reveal promising results.
 Terry Wright, Ph.D.
Terry Wright, Ph.D.Dr. Gigliotti has dedicated much of his career to studying Pneumocystis pneumonia, an opportunistic fungal infection that affects immunocompromised patients, commonly including those undergoing chemotherapy treatments for cancer or taking immunosuppressant medication for autoimmune diseases. Modern medicine has led to an increase in immunosuppressing agents, putting more and more patients at risk for PcP. Untreated, the disease is 100% fatal. However, even with antibiotic treatment (Bactrim being the current treatment of choice), the mortality is still relatively significant due to the disease’s bystander inflammatory injury to the lung, which the current treatment does not adequately prevent.
The lab of Dr. Gigliotti, including former pediatric infectious diseases fellows, Brenda Tesini, M.D., and Zachary Hoy, M.D. have been working towards the development of a vaccine in hopes of disease prevention through active immunization and disease treatment through passive immunization. They have had success with both approaches, which is quite the feat, as a major complication associated with working with Pneumocystis is the host range specificity – organisms that infect one mammalian species (i.e. mice) do not infect other mammalian hosts (i.e. humans). Thus, an antigen that protects against the disease in one mammal will not likely protect another. However, Dr. Gigliotti’s group has identified a molecule, the Pneumocystis cross reactive antigen 1 (Pca1), which induces a cross reactive antibody response in mice and humans. As revealed in their recent paper, they have tested the mouse antigen in an established mouse model with excellent results – nearly all mice immunized with Pca1 were completely protected against infection, whereas all unimmunized control mice developed PcP, as did the control protein-immunized mice.
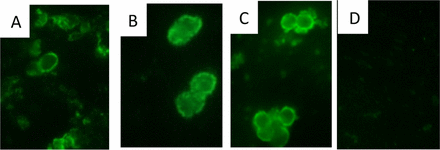
The next step will be to test the human antigen in this same mouse model in hopes of a similar response, and if successful, a clinical trial could be in the future. The work that has led up to this discovery spans nearly three decades, and additional collaborators who Dr. Gigliotti credits having a significant role throughout the years include Constantine G. Haidairis, Ph.D. in the Department of Microbiology and Immunology; P.J. Simpson-Haidaris, Ph.D. at the Clinical & Transnational Science Institute at URMC; and Allen Harmsen, Ph.D. now at Montana State University.
Drs. Gigliotti and Wright are in the process of applying for a grant to support the rest of their active immunization study and look forward to the work that lies ahead. They are also in the process of finalizing a manuscript outlining their passive immunization findings, which have great potential for clinical use. With an estimated 10,000 U.S. PcP cases each year, and more than 400,000 cases worldwide, this work has the potential to alleviate thousands of patients who are at risk for or who have developed Pneumocystis.
Novel T Cell Development Model Provides Insight Into Respiratory Disease In Premature Infants
By Meghan Plog M.S.
 Kristin Scheible, M.D.
Kristin Scheible, M.D.The lab of Kristin Scheible, M.D. studies T cell differences in the fetus and infant with specific interest in identifying how these differences may cause disease susceptibility and severity in premature babies. While it is known that the cytokine IL-8 is produced in newborns, little else is known about the adaptive immune system in preterm infants and how their fetal programming may change the way their adaptive immune system reacts to inflammation and infection. Dr. Scheible’s lab has discovered and established a fetal T cell development model that reveals novel insight in regards to T cell development and transformation throughout the gestational period, shedding new light on the cause of respiratory vulnerability in preterm infants.
 This model reveals that early on in the gestational period when thymic output is lower, peripheral T cells proliferate in response to rapid fetal growth. Throughout this process, the peripherally expanded cells lose CD31 expression and IL-8 effector function, and gain TNF-α function. As a baby continues to grow to term, this peripheral expansion decreases as thymic output increases, and the T cell profile changes to reveal a predominance of IL-8 producing cells. These findings have serious implications for infants born prematurely. According to this model, the majority of a preterm infant’s T cells will produce TNF (pro-inflammatory), as opposed to the requisite IL-8, which has been proven to be more protective against disease, less inflammatory, and essential in establishing normal memory that will aid in protecting against disease in the future.
This model reveals that early on in the gestational period when thymic output is lower, peripheral T cells proliferate in response to rapid fetal growth. Throughout this process, the peripherally expanded cells lose CD31 expression and IL-8 effector function, and gain TNF-α function. As a baby continues to grow to term, this peripheral expansion decreases as thymic output increases, and the T cell profile changes to reveal a predominance of IL-8 producing cells. These findings have serious implications for infants born prematurely. According to this model, the majority of a preterm infant’s T cells will produce TNF (pro-inflammatory), as opposed to the requisite IL-8, which has been proven to be more protective against disease, less inflammatory, and essential in establishing normal memory that will aid in protecting against disease in the future.
This new model has been supported through two large translational studies – the Prematurity and Respiratory Outcomes Program (PROP) led by Gloria Pryhuber, M.D. and Thomas Mariani, Ph.D. and the Prematurity, Respiratory Outcomes, Immune System, and Microbiome (PRISM) study led by Gloria Pryhuber, M.D., Mary Caserta, M.D. and Steven Gill, Ph.D. through the Respiratory Pathogens Research Center (RPRC). Through measuring respiratory outcomes at one year (PROP) and infections throughout the first year (PRISM), Dr. Scheible’s findings were consistent with the model – premature infants with low CD31+ IL-8 producing cells upon reaching gestational age not only went on to have respiratory disease, but were more susceptible to earlier infections. While it has been known that premature infants are at greater risk for developing respiratory viruses in their first year of life, this new information lays the groundwork as to what intrinsic differences in T cell phenotype and function may be contributing to this fact.
Dr. Scheible looks forward to using this new information to further identify the functional potential of these different types of T cells in relation to disease susceptibility and severity. She gives much credit to the excellent mentorship she has had through her primary mentors, David Topham, Ph.D. and Gloria Pryhuber, M.D., as well as the collaborations that have helped support this work.
Research Publication Highlights
Oct 2016 – Feb 2017 Selected Publications
- Neurocognitive Function in Children with Primary Hypertension. Lande MB, Batisky DL, Kupferman JC, Samuels J, Hooper SR, Falkner B, Waldstein SR, Szilagyi PG, Wang H, Staskiewicz J, Adams HR. J Pediatr. 2016 Sep 27. pii: S0022-3476(16)30859-9. doi: 10.1016/j.jpeds.2016.08.076. PMID: 27692987
- Auditory Toxicity in Late Preterm and Term Neonates with Severe Jaundice. Amin SB, Saluja S, Saili A, Laroia N, Orlando M, Wang H, Agarwal A. Dev Med Child Neurol. 2016 Oct 8. doi: 10.1111/dmcn.13284. PMID: 27718221
- Effects of Phone and Text Message Reminders on Completion of the Human Papillomavirus Vaccine Series. Rand CM, Vincelli P, Goldstein NP, Blumkin A, Szilagyi PG. J Adolesc Health. 2016 Nov 8. pii: S1054-139X(16)30355-X. doi: 10.1016/j.jadohealth.2016.09.011. PMID: 27836533
- Medication Identification in Pediatric Asthma (MED ID): The Reliability and Validity of a Novel Screening Tool. Frey SM, Fernandez ID, Ossip DJ, Halterman JS. J Asthma. 2016 Nov 18:0. PMID: 27860526
- Etonogestrel Implants in Adolescents: Experience, Satisfaction, and Continuation. Obijuru L, Bumpus S, Auinger P, Baldwin CD. J Adolesc Health. 2016 Mar;58(3):284-9. doi: 10.1016/j.jadohealth.2015.10.254. PMID: 26903428
- Development of a Global Respiratory Severity Score (GRSS) for Respiratory Syncytial Virus Infection in Infants. Caserta MT, Qiu X, Tesini B, Wang L, Murphy A, Corbett A, Falsey AR, Holden-Wiltse J, Walsh EE. J Infect Dis. 2016 Dec 23. pii: jiw624. doi: 10.1093/infdis/jiw624. PMID: 28011907
- Immunization with Pca1 Protects Mice Against Pneumocystis Pneumonia and Generates Antibody to Pneumocystis Jirovecii. Tesini BL, Wright TW, Malone JE, Haidaris CG, Harber M, Sant AJ, Nayak JL, Gigliotti F. Infect Immun. 2016 Dec 28. pii: IAI.00850-16. doi: 10.1128/IAI.00850-16. PMID: 28031260
- Methyl Mercury Exposure and Neurodevelopmental Outcomes in the Seychelles Child Development Study Main Cohort at Age 22 and 24 Years. van Wijngaarden E, Thurston SW, Myers GJ, Harrington D, Cory-Slechta DA, Strain JJ, Watson GE, Zareba G, Love T, Henderson J, Shamlaye CF, Davidson PW. Neurotoxicol Teratol. 2016 Oct 27. pii: S0892-0362(16)30125-8. doi: 10.1016/j.ntt.2016.10.011. PMID: 27989696
- Preventing Permeability Transition Pore Opening Increases Mitochondrial Maturation, Myocyte Differentiation and Cardiac Function in the Neonatal Mouse Heart. Lingan JV, Alanzalon RE, Porter GA Jr. Pediatr Res. 2017 Jan 31. doi: 10.1038/pr.2017.19. PMID: 28141792