Pediatrics / Palis Lab / Current Research Projects / Response of the Hematopoietic System to Ionizing Radiation
Response of the Hematopoietic System to Ionizing Radiation
While radiation causes bone marrow damage and drops in peripheral blood counts, the response of the erythroid and megakaryocyte lineages to irradiation has not been well characterized. We have developed novel approaches to quantify erythroid and megakaryocyte precursors using multispectral imaging flow cytometry (McGrath, et al. J Immunol Methods, 2008; Niswander, et al. Cytometry A, 2014). Our studies indicate that erythroid progenitors and precursors, but not megakaryocyte precursors, are extremely sensitive targets of radiation damage. The recovery of the erythroid lineage is dependent on the action of erythropoietin on late-stage erythroid progenitors (Peslak, et al. Blood, 2012). Radiation injury also leads to SDF-1-mediated movement of megakaryocytes within osteoblastic and endothelial niches (Niswander, et al. Blood, 2014). We are currently investigating the potential role of directed therapy to mitigate damage and enhance the recovery of hematopoietic cells following a bioterrorist attack or nuclear disaster.
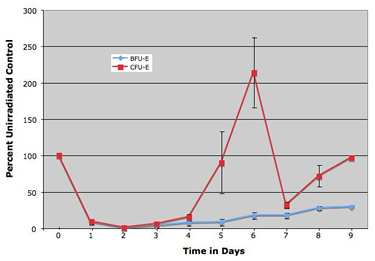
Differential response of early-stage erythroid progenitors (BFU-E) and late-stage erythroid progenitors (CFU-E) to a single dose of 4 Gy radiation.
References
- Saunders II J, Niswander LM, McGrath KE, Koniski A, Catherman SC, Ture SK, Medhora M, Kingsley PD, Calvi LM, Williams JP, Morrell CN, Palis J. Long-acting PGE2 and lisinopril mitigate H-ARS. Radiation Research 2021. (in press)
- Calvi LM, Frisch BJ, Koniski A, Kingsley PD, Kennedy J, Love T, Williams J, Palis J. Acute and late effects of combined internal and external radiation exposures on the hematopoietic microenvironment. International Journal of Radiation Biology 195:1447-1461, 2019.
- Niswander LM, Kingsley PD, McGrath KE, Palis J. SDF-1 dynamically regulates megakaryocyte niche occupancy and thrombopoiesis. Blood 124:277-286, 2014.
- Peslak SA, Wenger J, Bemis JC, Kingsley PD, Koniski AD, McGrath KE, Palis J. EPO-mediated expansion of late-stage erythroid progenitors in the bone marrow initiates recovery from sublethal radiation stress. Blood 120:2501-2511, 2012.
- Peslak SA, Wenger J, Bemis JC, Kingsley PD, Koniski AD, Chen Y, Williams JP, McGrath KE, Dertinger SD, Palis J. Sublethal radiation injury uncovers a functional transition during erythroid maturation. Experimental Hematology, 39:434-445, 2011.