Nanomembrane Research Group
The Nanomembrane Research Group (NRG) includes students, engineers, scientists, faculty and entrepreneurs at UR, a local nanomembrane manufacturer SiMPore Inc., and the Rochester Institute of Technology (RIT) and a growing network of academic and industry collaborators from around the world. Together, we have grown a serendipitous material discovery into a growing Rochester-based enterprise.
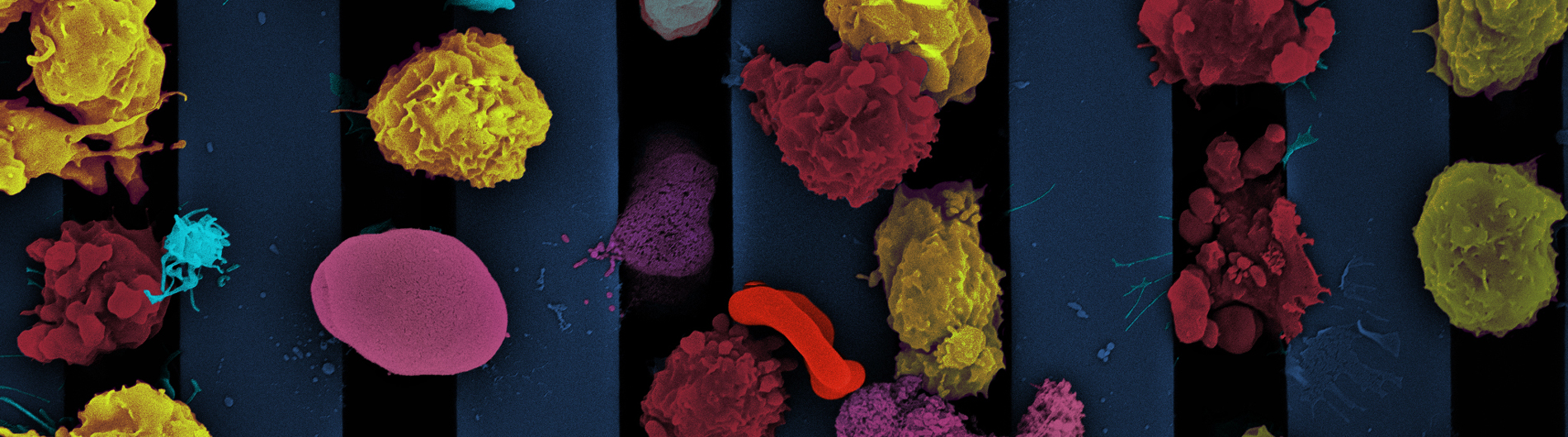
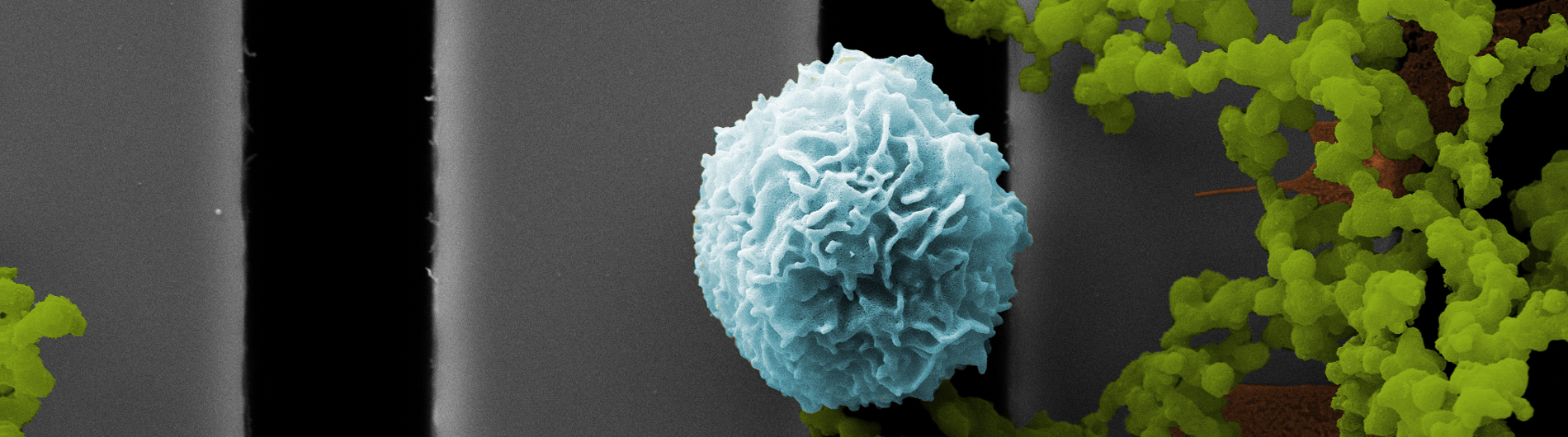
We introduced the first practical ultrathin freestanding nanoporopus membranes at the University of Rochester in 2007. Today, we manufacture and apply a variety of nanoporous and microporous membranes with the common characteristics that they are ultrathin (15 nm - 400 nm) and made with silicon-based manufacturing. Because these 'nanomembranes' are orders-of-magnitude thinner than conventional membranes, they are orders-of-magnitude more permeable to both diffusing molecules and pressurized flows. Molecular scale thickness also enhances the resolution of separations when the membranes are used as sieves. The silicon platform enables the ready assembly of membranes into devices using easily customized, but also highly scalable, layer-by-layer assembly. The unique properties of silicon nanomembranes have sparked paradigm-shifting research programs in:
- Biological tissue models
- Small format hemodialysis
- Biosensors
- Electrokinetic devices
In addition to these applications, our team focuses on the basic science of ultrathin membranes including studies of transport and mechanics.

James L. McGrath
Principal Investigator

Team Science Showcase: The People and Partnerships Moving Microplastics Research Forward
Public concern about microplastics and human health is growing. Learn how University of Rochester scientists and community partners are advancing research through interdisciplinary teams and innovative collaborations.
Read the full story →- Nanomembrane-based microfluidic platform with embedded electrical pressure transducer for on-chip nanoparticle quantification.; Lab on a chip. 2026 Apr 30.
- Bacterial extracellular vesicles indirectly destabilize a human stem cell-derived blood-brain barrier on-chip through pro-inflammatory stimulation of immune cells.; Lab on a chip. 2026 Apr 14.
- A human synovial tendon-on-a-chip models key features of peritendinous adhesions and offers a new approach methodology for testing anti-fibrotic drugs.; bioRxiv : the preprint server for biology. 2026 Apr 07.
- Type III Collagen Promotes a Pro-Fibrotic Microenvironment for In Vitro Tendon Healing Microphysiological Systems.; Journal of orthopaedic research : official publication of the Orthopaedic Research Society; Vol 44(4), pp. e70192. 2026 Apr.
News
Affiliations
- Biomedical Engineering
- Del Monte Institute for Neuroscience
- Environmental Health Sciences Center
- James P. Wilmot Cancer Center
- NIH T32 Training Grant in Immunology
- Pulmonary T32 Training Grant
- Biomedical Engineering Ph.D. Program
- Biophysics, Structural & Computational Biology Ph.D. Program
- Neuroscience Ph.D. Program
February 9, 2026
Team Science Showcase: The People and Partnerships Moving Microplastics Research Forward
February 23, 2021
Rochester to advance research in biological imaging through new grant
September 29, 2020
Awad, McGrath and Miller receive a $4M NCATS/NIAMS Clinical Trials on a Chip grant to study tendon inflammation and fibrosis
April 21, 2020
Rochester researchers pursue quick ways to detect COVID-19—and better understand it
Contact Us
McGrath Lab
306 Goergen Hall
Rochester, NY 14620
(585) 273-4746