Welcome to the Zhang Lab
Skeletal repair is a dynamic and well-orchestrated process that involves complex and coordinated function of different cellular compartments and integrated molecular pathways. Understanding complex molecular interactions during skeletal healing represents a critical step toward developing effective treatment strategies for improved repair and reconstruction.
Research in my laboratory focuses on skeletal repair and reconstruction, which integrates a number of important research topics in musculoskeletal research. These topics include biology of bone/cartilage development, cell signaling, stem cell biology and bone tissue engineering. Using transgenic mouse models, primary culture of progenitor cells isolated from bone callus, and the-state-of-the-art imaging approaches, we are currently trying to understand how molecular and cellular signals are integrated to provide synergistic action for repair and regeneration. Our research has been continuously funded by grants from NIH, NYSTEM, and foundations.

Xinping Zhang, B.Med., Ph.D.
Principal Investigator
Publications
View All Publications- Masquelet Inspired in Vivo Engineered Extracellular Matrix as Functional Periosteum for Bone Defect Repair and Reconstruction.; Advanced healthcare materials. 2025 Jan 22.
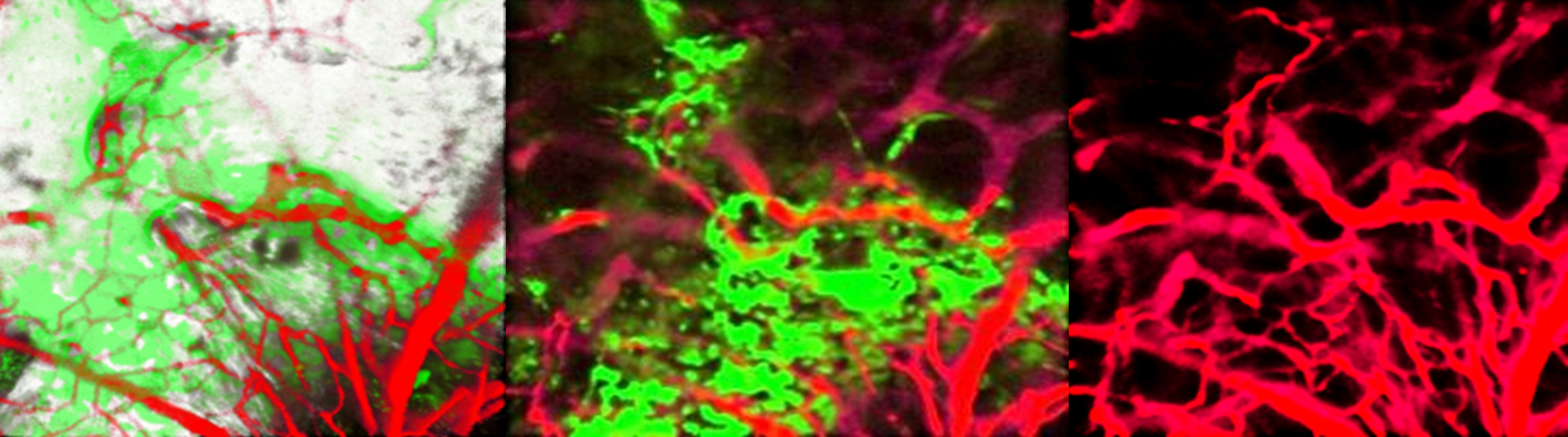
- Differential bone and vessel type formation at superior and dura periosteum during cranial bone defect repair.; Bone research; Vol 13(1), pp. 8. 2025 Jan 13.
- Doctors vs. Nurses: Understanding the Great Divide in Vaccine Hesitancy among Healthcare Workers.; Proceedings : ... IEEE International Conference on Big Data. IEEE International Conference on Big Data; Vol 2022, pp. 5865-5870. 2022 Dec.
- Fabrication of channeled scaffolds through polyelectrolyte complex (PEC) printed sacrificial templates for tissue formation.; Bioactive materials; Vol 17. 2022 Nov.
- A Comprehensive Assessment on the Pivotal Role of Hydrogels in Scaffold-Based Bioprinting.; Gels (Basel, Switzerland); Vol 8(4). 2022 Apr 13.
- High resolution imaging of the osteogenic and angiogenic interface at the site of murine cranial bone defect repair via multiphoton microscopy.; eLife; Vol 11. 2022 Jan 03.
Contact Us
Zhang Lab
601 Elmwood Ave, Box 665
Rochester, NY 14642