Endothelin Signaling in Glaucomatous Neurodegeneration
The endothelin (EDN) system has been implicated in human and animal models of glaucoma. Activation of the EDN system has been shown to be an early, pathogenic response in animal models of glaucoma. The EDN system consists of three ligands, EDN1, EDN2, EDN3, and two receptors, endothelin receptor subtype A and B (EDNRA and EDNRB respectively). Many organ systems, including the central nervous system, use the EDN system to maintain normal physiology. As with many signaling systems that have a physiological role, EDN signaling also has been broadly implicated in the pathophysiology of numerous diseases, including several retinal diseases. There are numerous lines of evidence that support a role for EDN signaling in human glaucoma. Genetic studies have shown an association between EDN family genes and several types of glaucoma. In human glaucoma, EDN levels are elevated in both the plasma and aqueous humor of patients with primary open angle glaucoma. Interestingly, the canonical role of EDN signaling is to regulate vasoconstriction and blood flow. Changes in blood flow have been documented in human glaucoma and it is hypothesized that these changes could be important factors in the development and progression of glaucoma. In animal studies, intravitreal injection of EDN1 or EDN2 results in RGC loss and axonal degeneration. Also, multiple components of the EDN system are upregulated in both the retina and optic nerve after a variety of glaucoma-relevant insults. Importantly, inhibiting the EDN system lessens glaucomatous neurodegeneration in three different systems used to model glaucomatous neurodegeneration. Determining the mechanism of EDN-induced glaucomatous neurodegeneration is complex because multiple glaucoma-relevant cell types express EDN receptors. For instance, EDNRA is expressed in cells of the retina and optic nerve head vasculature while EDNRB is expressed on retinal neurons and retinal glia. As these receptors are expressed on different tissue types, it is unclear if the injury mediated by EDN ligands is a direct injury to the RGCs or an indirect injury mediated by non-neuronal cells such as glial or vascular cells. Furthermore, the EDN system could be involved in several cell biological events implicated in glaucomatous neurodegeneration, including: macroglia reactivity, monocyte infiltration, breakdown of the blood brain barrier, constriction of blood vessels and alteration of blood flow, diminished axonal transport, and direct neurotoxicity to RGCs. To understand this important system in retinal disease, we are combining genetic, cell biological, and genomic approaches to identify important signaling events both upstream and downstream of EDN signaling critical for the early stages of glaucomatous neurodegeneration.
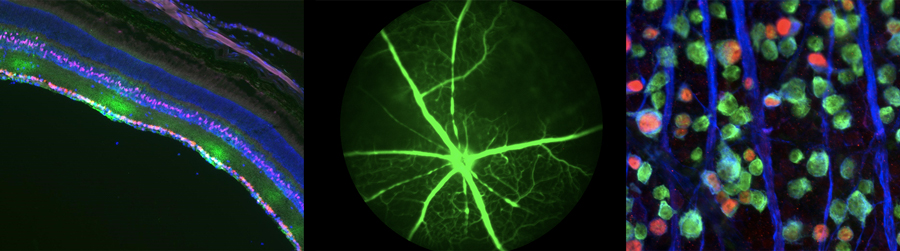
Left: Retinal section depicting hypoxia (green) of RGCs (red) and Muller glia (pink) after endothelin treatment
Middle: Fluorescein angiography depicting loss of retinal vascular perfusion after endothelin treatment
Right: Retinal whole-mount depicting RGCs (green and blue) with an accumulation of the pro-apoptotic transcription factor JUN (red) after endothelin treatment