Just a Drop of Blood: An Easier Way to Test for COVID-19
As researchers around the world race to respond to the COVID-19 pandemic, UR CTSI Co-Director Martin Zand, M.D., Ph.D., is rapidly adapting previous research to develop a potential diagnostic test that would require just a few drops of blood to detect the fast-spreading disease.
The current research was born out of a past UR CTSI pilot project in which Zand’s lab created a finger-stick test that can detect immunity to over 50 strains of the flu from just a few drops of blood. The test, which is similar to a blood sugar test for diabetes, comes in a convenient kit – complete with detailed instructions, a small lancet, and blood collection swabs in an easy-to-mail case.
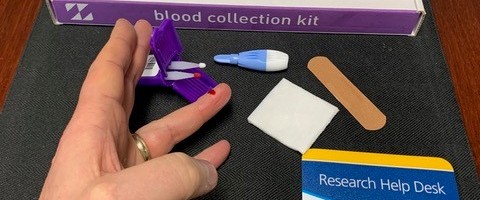
At-home Mitra® Blood Collection Kits from Neoteryx are being used for UR CTSI’s coronavirus research study.
Currently validated for use in research, the test allows clinical study participants to collect their own blood samples at home and mail them to the lab. This cuts down on time and travel for study participants, which is extremely convenient regardless of global pandemics, but also reduces their risk of COVID-19 exposure.
As Zand’s lab pivots to use the test to detect immunity to COVID-19, they hope it could also offer a quick and easy way to test immune responses to experimental vaccines.
“We're hoping this could make coronavirus research - especially vaccine trials - faster and more convenient for those who volunteer for them,” said Zand, who is also senior associate dean for Clinical Research at the University of Rochester Medical Center. “And hopefully, that will allow us to enroll many, many more people and get more and better information.”
Once the samples are mailed to Zand’s lab, his team can glean an astounding amount of information from each tiny blood spot. In each sample, they will look for antibodies (a crucial part of the immune response) against the virus that causes COVID-19, called SARS-CoV-2, as well as related coronaviruses that cause the common cold, and the SARS-Cov-1 virus that emerged in 2003.
The test will help us understand whether prior exposure to seasonal coronaviruses might protect some people from developing a COVID-19 infection. This could help explain why some COVID-19-positive people have very mild symptoms or no symptoms at all. It might also tell us whether SARS-CoV-2 can hijack antibodies – which usually help you fight off viruses – to instead help it infect more of your cells and make you sicker.
That hijacking, called antibody-dependent enhancement, happens in viral infections that are similar to COVID-19, like SARS and MERS, as well as Zika and Dengue. This phenomenon can make it more difficult to develop vaccines against these viruses, so it is critical to know whether it happens in COVID-19. (See Zand’s recent article on this topic in the Journal of Clinical and Translational Science.)
Zand is currently seeking funding for his lab’s coronavirus research, but hopes to eventually test one thousand people in Rochester and abroad (pending a collaboration with an Italian research group). The project would focus on testing healthcare workers (who are at high risk), people who tested positive for COVID-19, and people who had symptoms, but were never tested.
“Ultimately, if we can get this test validated and approved by the FDA,” Zand said, “it would be an inexpensive and easy way to monitor who is immune to COVID-19, so we can get people back to work safely.”
Learn more about other coronavirus diagnostic and sensor technologies at the University of Rochester.
##
The UR CTSI pilot study described above was supported by the University of Rochester CTSA award (UL1 TR000042) from the National Center for Advancing Translational Sciences of the National Institutes of Health. UR CTSI Pilot Awards provide seed funding for highly innovative research that spans the translational spectrum.
Michael Hazard |
4/20/2020
You may also like