Project 2:
Understanding the pathophysiology of ALI by studying the link between endothelial cell (EC) cytoskeletal dynamics and inflammatory gene expression.
Our second project involves dissecting the mechanistic link between endothelial cell (EC) cytoskeletal dynamics and inflammatory gene expression and to understand the relevance of this linkage in the pathophysiology of ALI.
Cytoskeletal structures occur throughout the animal and plant kingdom and are involved in essentially all structural and dynamic aspects of living cells including cellular signaling. These intricate signaling networks play a central role in maintaining cellular homeostasis and thus any dysfunction in the signaling pathway results in a diseased state.
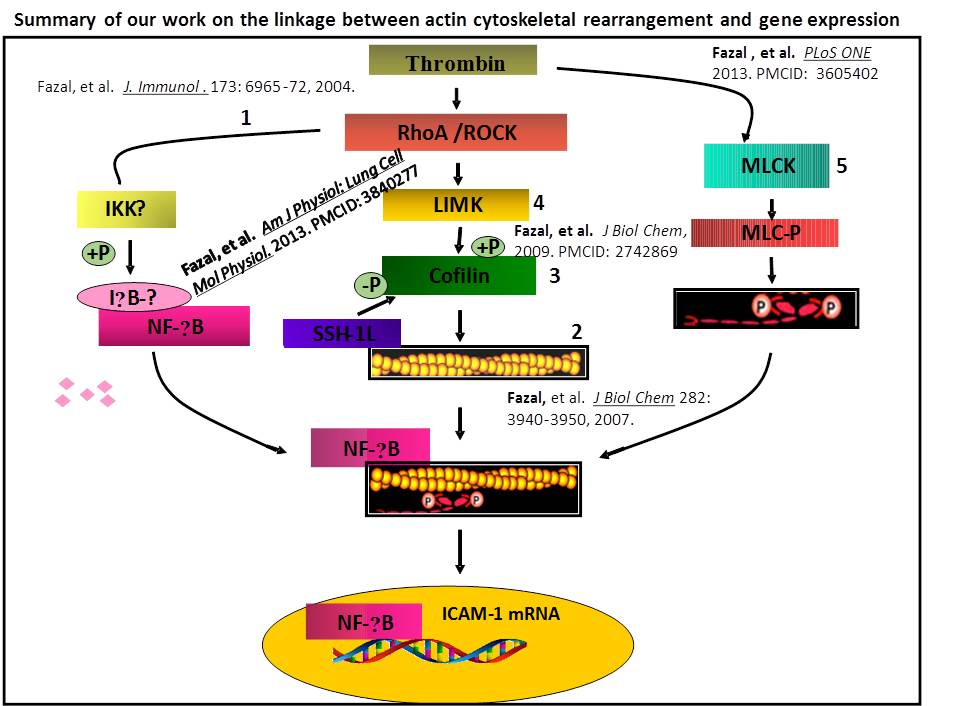
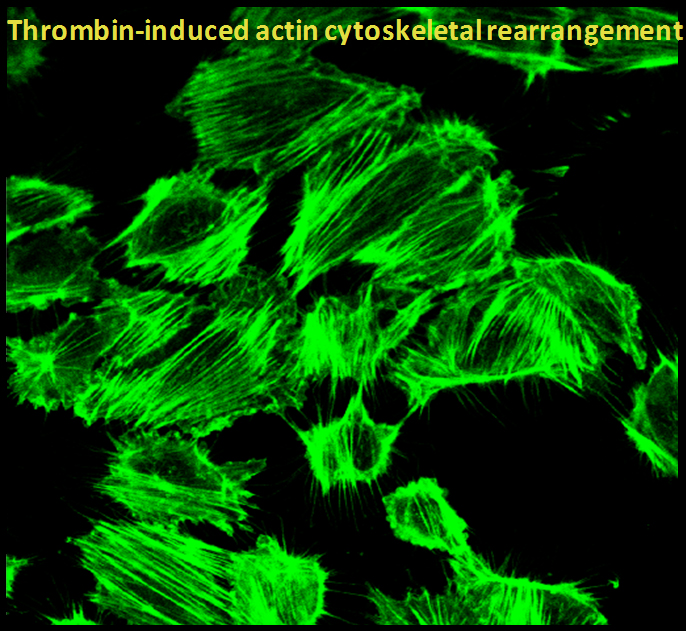
The pathogenesis of acute lung injury (ALI) involves bidirectional cooperation and close interaction between inflammatory and coagulation pathways. A key molecule linking coagulation and inflammation is the procoagulant thrombin, a serine protease whose concentration is elevated in plasma and lavage fluids of patients with ALI and acute respiratory distress syndrome (ARDS). We have developed a new mouse model that permits investigation of lung inflammation associated with intravascular coagulation induced by thrombin. Using this mouse model and in vitro approaches, we identified non-muscle myosin light chain kinase (nmMLCK) as a critical mediator of endothelial cell (EC) inflammation and lung neutrophil (PMN) infiltration in ALI. In addition our work has provided evidence that thrombin-induced actin cytoskeletal rearrangements are used as tracks for the cytoplasmic trafficking of the liberated NF-ĸB to the nucleus for the expression of proinflammatory adhesion molecules, cytokines and chemokines. Also we have identified the role of cytoskeletal proteins cofilin1, LIM Kinase1 (LIMK1) and slingshot 1 (SSH-1L) in mediating EC inflammation and permeability, hallmarks of ALI.