Dopamine and Stress: Circuits Through the Extended Amygdala
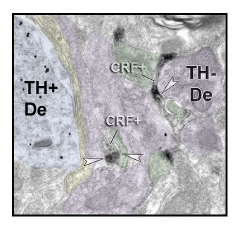
Figure 1. Pre-embedding dual-immunoreactivity electron microscopy (EM) shows DAB-filled CRF+ axons (gray) and Gold-labeled TH+ dendrites (black dots) in a lateral region of the A10 neurons. Excitatory synapses (asymmetric, black arrow heads), inhibitory synapses (symmetric, white arrow heads). Blue overlay= TH+ dendrite, purple overlay= dendrite, green overlay= dendritic spine, yellow overlay= presynaptic terminal
In 2000, we found that a structure known as the ‘extended amygdala’ had a direct impact on dopamine system in primates(1, 2). Although the idea was novel—and somewhat controversial-- at that time, subsequent work from other laboratories and our own indicate that the extended amygdala-dopamine pathway is an important pathway mediating the effects of stress-induced behaviors, including depressive-like symptoms and drug-seeking.
We now focus on the microcircuitry of the extended amygdala-dopamine path, particularly the role of afferent inputs containing corticotropin releasing factor (CRF), which amplifies circuit responses. We recently found that all projections from the extended amygdala largely avoid the midline ‘classic’ VTA in nonhuman primates, targeting instead the more lateral regions of the VTA. Importantly, CRF-containing terminals tend to form inhibitory synapses on GABAergic interneurons (rather than directly on the DA cells). This suggests a mechanism in which CRF-GABA terminals inhibit GABAergic interneurons, and releasing the brakes on DA efflux under stressful circumstances.
Dopamine release is best-studied in ‘reward’ paradigms, but dopamine plays a role in pain, novelty, and uncontrollable stress. Importantly, dopaminergic neurons are now understood to contain a variety of co-transmitters, including glutamate and GABA. While groundbreaking studies in rodents have shown circuit-specific roles for ‘multiplexed’ dopamine neurons, almost nothing is known in higher species. Our new studies indicate that ‘multiplexed’ DA neurons are even more abundant in nonhuman primates than in rodents. Planned studies will compare how they participate in DA projections to various sites in the mesolimbic circuit.
- Fudge JL, Haber SN (2000): The central nucleus of the amygdala projection to dopamine subpopulations in primates. Neuroscience. 97:479-494
- Fudge JL, Haber SN (2001): Bed nucleus of the stria terminalis and extended amygdala inputs to dopamine subpopulations in primates. Neuroscience. 104:807-827