Epigenetic Regulation of Zebrafish Development
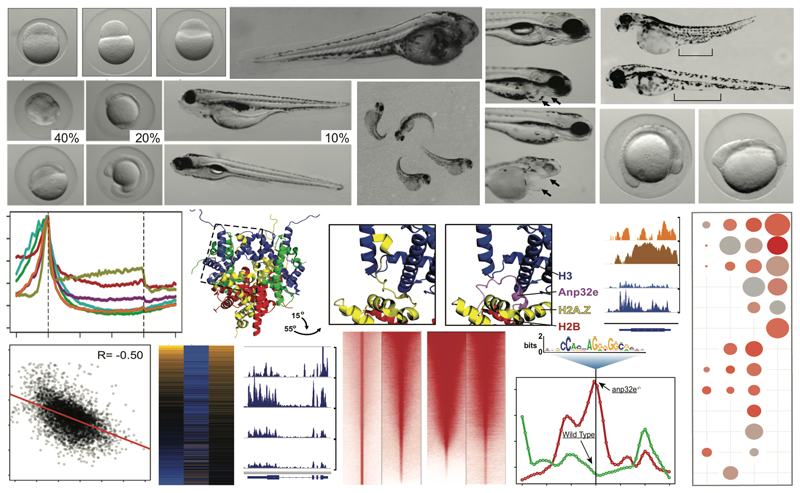
During early zebrafish development, the DNA methylation patterns of the two parental genomes are harmonized such that genes important for early development lack DNA methylation, while the rest of the genome (including genes involved in terminal differentiation) becomes highly methylated. Prior to our recent results, it was unknown how these genes underwent DNA methylation reprogramming. However, one thing was clear – the maternal genome was reprogrammed to become identical to the paternal genome, and the paternal genomic DNA methylation patterns were maintained. To explain how genes became demethylation on the maternal genome, we hypothesized that a ‘Placeholder’ chromatin feature was inherited from sperm and established on the maternal genome to facilitate DNA demethylation by blocking DNA methyltransferases (DNMTs). During his postdoctoral training in Dr. Brad Cairns laboratory, Dr. Murphy tested this hypothesis, and was successful in identifying epigenetic marks that makeup the ‘Placeholder’ nucleosome (Murphy PJ et al. Cell 2018).
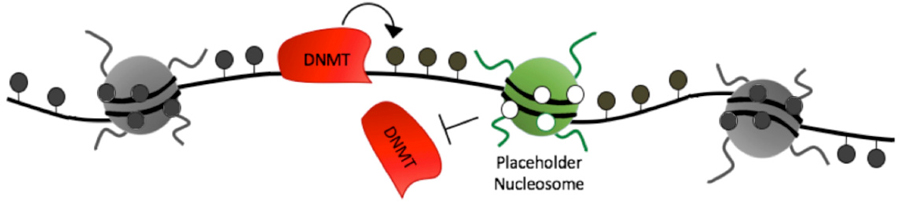
DNA methylation reprogramming during zebrafish cleavage phase relies on both establishment and antagonism of Placeholder nucleosomes to enable proper transcriptional activation. Placeholder is established at regions gradually undergoing maternal DNA demethylation, and antagonized at regions undergoing default acquisition of DNA methylation. This mechanism ensures that both housekeeping and many developmental genes acquire Placeholder, and lack DNA methylation. Concurrent with zygotic transcriptional activation, the histone mark H3K4me3 is acquired at activated housekeeping genes, and bivalent marks are established at ‘poised’ developmental genes. Developmental genes expressed much later in development lack Placeholder, and are DNA methylated. As follow-up to these studies, the Murphy laboratory is now investigating mechanisms by which Placeholder functions at the molecular level, and testing whether Placeholder patterning is important for cell type specification at later stages of development. We are also investigating whether environmental toxins, including cigarette smoking and oxidative stress agents (Murphy PJ et al. PLOS Genetics 2020) function in part through disruption of Placeholder nucleosomes.


For many reasons, the zebrafish model is a powerful tool for studying various aspects of vertebrate diseases and normal developmental processes. These fish have a short generation time, are small, and are highly manipulatable for functional studies. Additionally, zebrafish are transparent as embryos. For these reasons as well as others, zebrafish have become a prevailing model for studying cell state transitions during vertebrate differentiation and in carcinogenesis. More recently, zebrafish have emerged as incredibly valuable when combined with genomics and biochemical studies. This is largely because zebrafish fertilization and early organismal development occur ex utero, and a single breeding pair can produce over 200 offspring per mating. Therefore, many thousands of cells can be harvested for complex studies that would be costly, and require dissection, if other vertebrate models were used.