Multimodal Precursor-derived AstroCyte Therapy (MPACT)
Multimodal Precursor-derived AstroCyte Therapy (MPACT)
Injuries to the brain, whether caused by trauma or degenerative disease, are highly debilitating and currently without effective treatment. The common denominator to both traumatic and neurodegenerative disease in CNS is the disruption of the environment at the injury site.
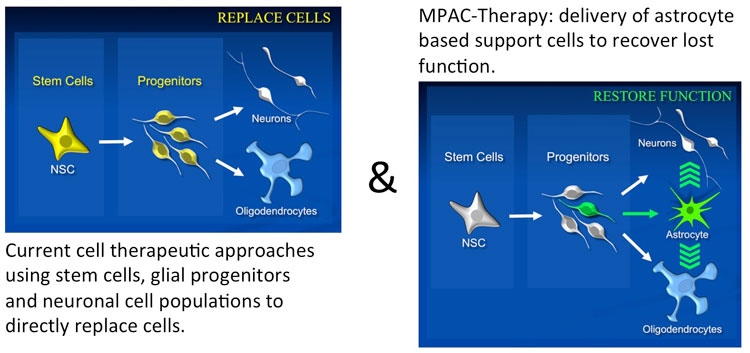
This disruption prevents the repair of damaged neurons and perpetuates the disease process. My laboratory has focused on restoring the tissue environment to stop disease progression and to enable regeneration. In this context we have made the fundamental discovery that restorative remodeling of the injury site can be readily achieved through transplantation of a specific astrocyte population derived from a novel progenitor cell. We refer to these astrocytes as MPACs (Multimodal Precursor-derived AstroCytes). Using MPACs we have achieved tissue repair with near complete functional recovery in animal models of both traumatic and neurodegenerative CNS disease. Our current results suggest that rather than simply replacing lost neural cells, these astrocytes act through several paracrine pathways to reprogram the lesion environment and to orchestrate endogenous neural, vascular and immune cells to repair the injury.
Synaptophysin staining of neuronal synapses.
As we move forward, the broad benefits observed not only provide new insights into repair mechanisms, but also allow us to test this therapeutic approach in several CNS disease models, including traumatic brain injury, stroke, Parkinson’s, Huntington’s and Alzheimer’s disease.
At the same time, it is equally important to bring this novel therapeutic approach to the clinic. Our ability to produce cryopreserved, transplantable human astrocytes is an important step towards facilitating collaborative efforts with non-human primate models in the near term, and to enable clinical trials in the future.