Developmental Immunotoxicology
1. Use of a comparative biology approach in Xenopus to define how postembryonic exposure to endocrine disruptor contaminants impacts immune cell development and weaken antimicrobial immune response later in life: Human population and aquatic vertebrates such as amphibians are increasingly exposed to water contaminated with mixtures of endocrine disruptor chemicals (EDCs) at levels below government regulation. These common environmental pollutants are often innocuous when considered each separately, but in mixture their activity may synergize and cause long-lasting deleterious developmental and immune defects. Despite this, the causalities and extent of these immune compromises remains unexplored largely due to the lack of appropriate, cost-effective animal models.
We have established Xenopus as an experimental platform for the study of toxicological effects of common waterborne pollutants in mixture or alone to the development of immune cells and effective immune response to virus (ranavirus FV3) and mycobacteria. Notably, metamorphosis parallels the perinatal period in humans. Using the Xenopus-FV3 experimental platform, we have shown that certain herbicides (atrazine) and insecticides (carbaryl) contaminating water at low but ecologically-relevant concentrations induce dramatic acute and persistent defects in anti-FV3 immune responses. We have also shown that exposure to the mixture of 23 UOG-EDCs induces acute immune perturbation in tadpoles and further showed immunotoxicity of 6 of these chemicals (10.1016/j.mce.2020.110722).
We are now focusing on EDCs that alter thyroid hormone function, postulating that developmental exposure to TH disrupters alters T cell differentiation and function. This in turn can result in long-term perturbation of immune homeostasis and poorer antimicrobial immunity.
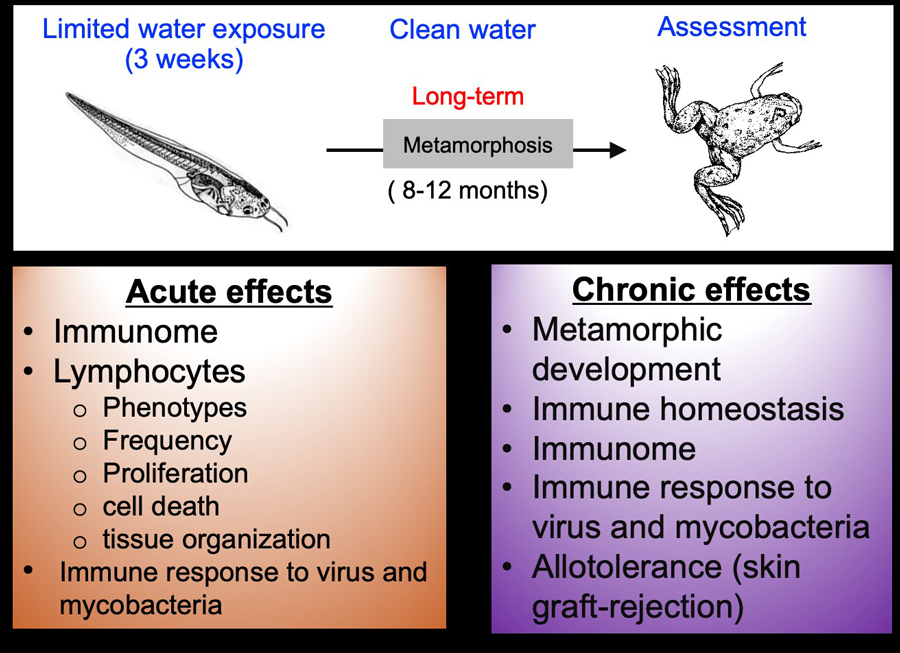
Schematic of our experimental design
2. Effects of microplastics on Xenopus immunity:
Microplastic (MPs) debris sizing from sizing from 10 mm to 100 nm, have become notoriously ubiquitous pollutant of marine and freshwater ecosystems worldwide, and are thought to pose a serious threat to aquatic ecology and aquatic organisms. In particular MPs have been detected a large amount in Upstate New York lake and river systems. There is an urgent need of a sensitive, standardized and reliable methodology to detect and quantify MPs in organisms as well as a rigorous assessment of the biodistribution and biological effects of MPs ingestion and their potential contribution to weakened resistance to pathogens.
Our goal is to leverage our Xenopus -FV3experimental system to rigorously assess the MPs biodistribution and accumulation in tadpole tissues, then evaluate the biological effects resulting from MPs ingestion on development, fitness as well as immune homeostasis and immune response to pathogens.
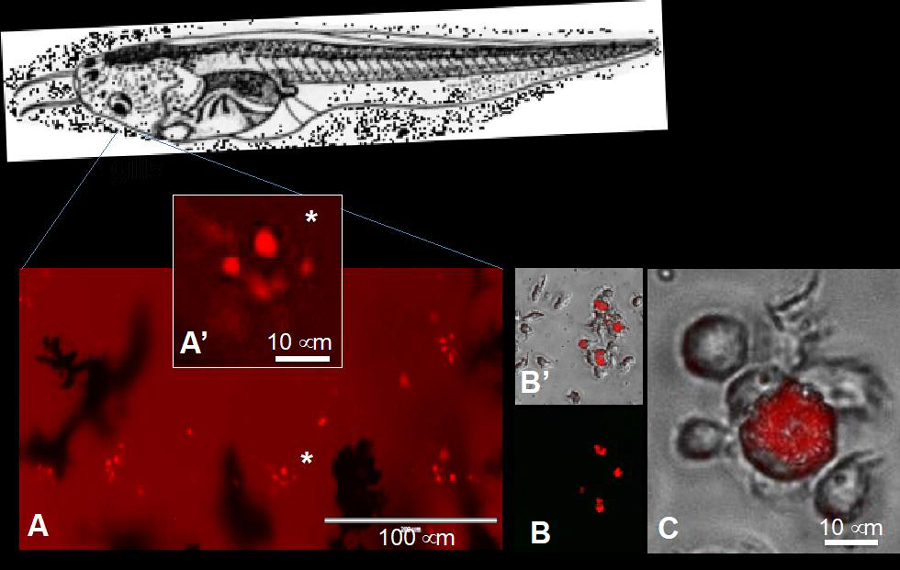
Detection of accumulating fluorescent MPs in gills phagocytes in vivo (A) and in peritoneal macrophages in vitro (B, C).