Fazal Lab
Overview
“The contents inside a eukaryotic cell are not static, but are constantly moving, following a choreography more precise and complex than that of a computer chip! The slightest of disruption in this precise choreography results in a pathological condition (disease)!”
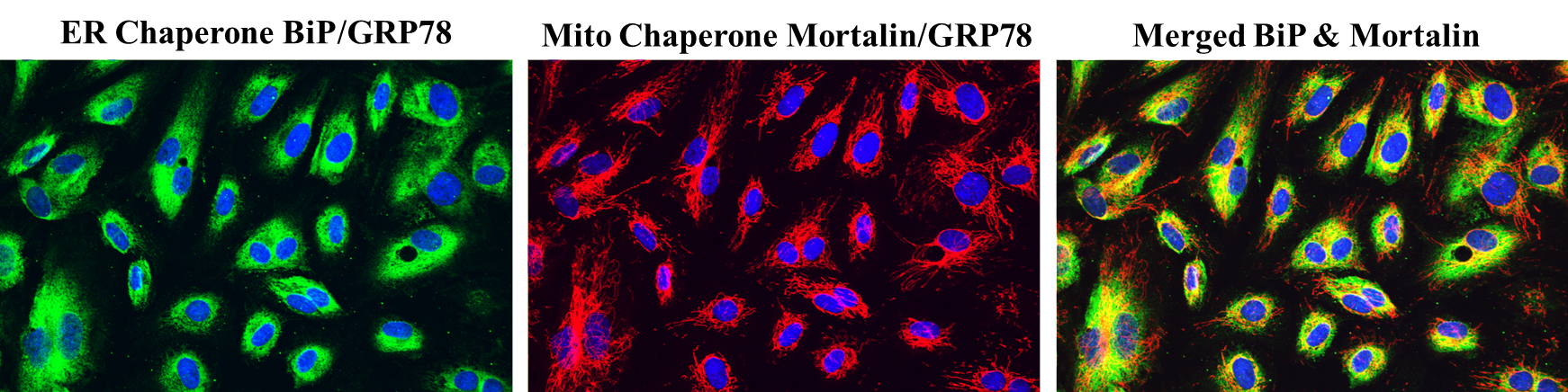
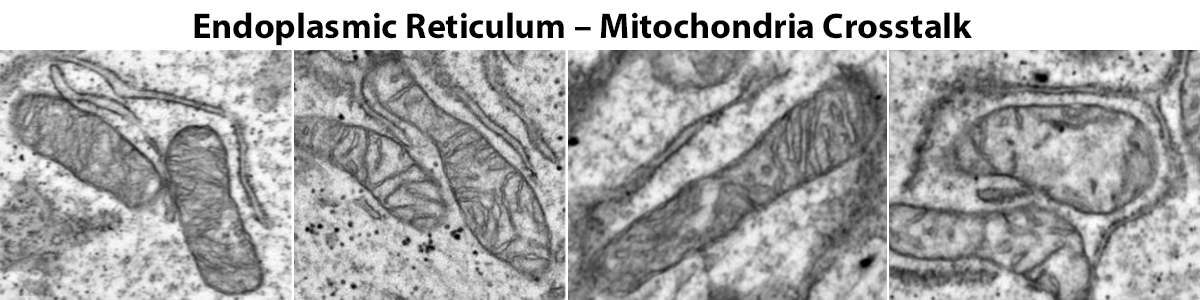
“Eucaryotic Cell is like a City! Cell membrane is city’s border; cytoskeleton is city’s transport system; cytosol is city’s streets; mitochondria is like a city’s powerhouse and endoplasmic reticulum is like a city’s industrial park …! For the proper functioning of the city and the cell, these player have to not only perform their designated task but have to communicate with each other! Thus “Inter-organellar” communication is key to cellular homeostasis – which in turn is key to OUR health and well-being!!!
Introduction
My lab focuses on understanding the pathogenesis of Acute Lung Injury (ALI)/Acute Respiratory Distress Syndrome (ARDS), a life threatening pulmonary inflammatory condition, which occurs as a response to or escalation of various lung injuries, including direct insults to the lungs such as viral or bacterial pneumonia, or indirect insults to the lungs such as sepsis. It is a common syndrome, occurring in approximately 10 percent of all patients admitted to hospital Intensive Care Units and 23 percent of all patients who are placed on mechanical ventilation. The annual incidence alone is over a quarter of million patients with a mortality rate of approximately 40 percent. There are also significant socio-economic ramifications, as ALI patients have an average stay in the hospital of 17 days, resulting in an annual cost of approximately10 billion dollars. Patients who survive this disorder are at high risk for cognitive decline, depression, post-traumatic stress disorder, and persistent skeletal-muscle weakness. Compounding the issue is the lack of any effective medicines to treat patients with ALI. Care focuses on what is called “supportive therapy,” referring to fluid management and/or further injury prevention via lung-protective ventilation. This field is in a desperate need of new and effect therapy.
Central to the pathogenesis of ALI are two key features: 1) disruption of lung endothelial and epithelial barriers, leading to the accumulation of fluid within the alveolus/airspace, and hence compromised lung function and 2) lung inflammation, which involves excessive migration of neutrophils, one of the critical innate immune cell, from blood through the interstitium into the airspace, where they not only kill the invading microbes but contributes to the progression of the disease.
The long term goal of my research is to identify novel mechanistic targets and assess the preventive and therapeutic benefit of targeting them against evolving ALI. In our lab we use integration of in vitro and in vivo techniques to dissect the pathways that are critical to endothelial cell (EC) inflammation and permeability. We use multidisciplinary approaches ranging from biochemical, cellular, and molecular biology to in vivo gene delivery and lung physiology.
- Critical Role of Mortalin/GRP75 In Endothelial Cell Dysfunction Associated With Acute Lung Injury.; Shock (Augusta, Ga.). 2019 Sep 04.
- Critical role of autophagy regulator Beclin1 in endothelial cell inflammation and barrier disruption.; Cellular signalling. 2019 May 01.
- Selective Inactivation of Intracellular BiP/GRP78 Attenuates Endothelial Inflammation and Permeability in Acute Lung Injury.; Scientific reports; Vol 9(1). 2019 Feb 14.
- Importins α and β signaling mediates endothelial cell inflammation and barrier disruption.; Cellular signalling. 2018 Jan 10.
Personnel
Lab News
- Dr. Fabeha Fazal and Dr. Arshad Rahman receive new NIH R01 grant from the National Institute of General Medical Sciences (NIGMS) to study “Regulation of Endothelial-Neutrophil Interaction and Lung Vascular Permeability in Sepsis”.
Contact Us
Fazal Lab
Department of Pediatrics, Box 850
601 Elmwood Ave
Rochester, NY 14642
Shipping Address
University of Rochester Medical Center
Dept of Pediatrics
ATTN: Dr. Fabeha Fazal
575 Elmwood Avenue (Room 3-11301 MRBX)
Rochester, NY 14642
(585) 275-9917
Labs: (585) 275-7674