B Cells and Immune Diseases

Jennifer Anolik, MD, PhD
Principal Investigator
B Cell Roles in Rheumatoid Arthritis
One of the research areas in the Anolik lab is directed toward understanding the pathophysiology of RA with a focus on the role of B cells. Studies from our lab have extensively characterized B cells and identified pathogenic B cell subsets that play a role in the bone erosion process. We have recently demonstrated that B cells can be a prime secretor of RANKL, thereby promoting osteoclast differentiation and bone erosion in RA. Ongoing work in the lab is investigating the signaling pathways in the RA synovium that promote B cell mediated osteoclast activation and osteoblast inhibition in RA. Approaches include defining disease specific pathways within human RA synovial tissue using novel technologies including laser capture micro-dissection and RNA sequencing of discrete immune and bone cell populations as well as the application of animal models.
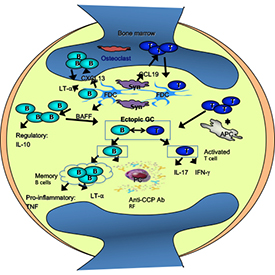
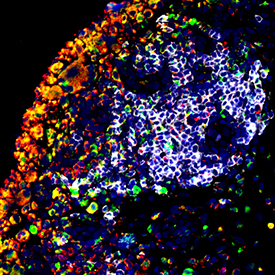
Figure legend:
(Left) Schematic of cellular interactions in the rheumatoid arthritis synovium and subchondral bone marrow.
(Right) Rheumatoid arthritis synovium showing infiltrating CD20+ B cells, some expressing RANKL and interacting with RANK+ cells to influence bone homeostasis.
Immunologic and ultrasound biomarkers in Primary Sjögren’s Syndrome
These studies seek to elucidate abnormalities in both the peripheral blood and salivary gland target tissue in the B cell and T cell compartment in Sjögren’s syndrome in order to develop biomarkers of disease and treatment. We are using flow cytometry to examine the B cell and T cell compartment and microarray to evaluate transcriptomic profiles of pSS patients. We are also exploring ultrasound of the salivary glands as a diagnostic approach and relating imaging characteristics to B cell activation and other immunologic abnormalities.
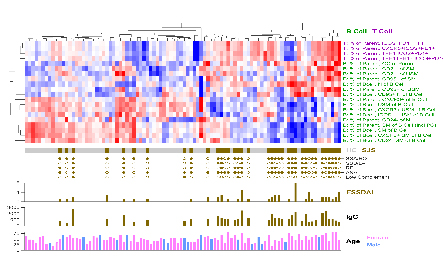
Figure legend:
Phenotypic profiles of primary Sjögren’s (pSS) and normal control characterized by multi-parameter flow cytometry. Cluster analysis reveals two distinct cluster of pSS patients based on T and B cells.
Development of Novel Treatment Approaches in Systemic Lupus Erythematosus (SLE)
Early work in our lab defined the critical role of B cells in SLE by both autoantibody dependent and independent mechanisms through the study of the immune systems of patients undergoing B cell depletion therapy (BCDT). However, durable clinical remissions in SLE after BCDT typically lead to eventual auto-antibody normalization. This suggests that the most effective therapeutic interventions impact multiple aspects of the immune system, including decreasing the number of pathogenic auto-reactive plasma cells (PCs). Notably, most therapies currently used to treat SLE, and B cell targeted therapies under development do not target PCs and the auto-antibodies they produce. A major focus in the lab is to define novel approaches that may eliminate autoreactive PCs. One project is examining the effect of selective inhibition of nuclear pore export, an approach that has effectively targeted malignant PCs in multiple myeloma. Utilizing a lupus mouse model, we have shown that administration of a small inhibitor of nuclear export (SINE) has a pronounced impact on the auto-immune humoral response by targeting auto-reactive GCs in the spleen and kidney of lupus prone mice. Our current goal is to further define the factors regulating the development and survival of autoreactive plasma cells in lupus patients using multiple approaches including in vitro co-culture of plasma cells, flow cytometry and RNA sequencing in order to develop multi-pronged strategies to decrease PC survival. In mouse models combinations of SINE drugs and other approaches, such as proteasome inhibition will be examined.
Vehicle
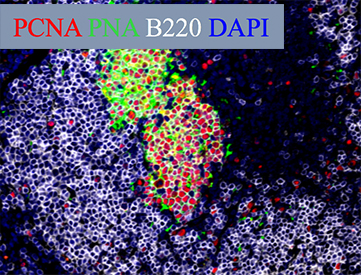
KPT-350 7.5 mg/kg
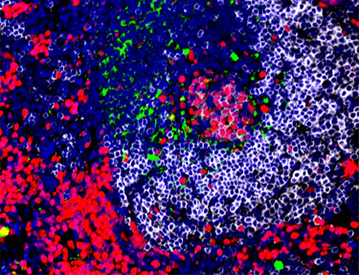
Figure legend:
(Left and Right) Reduction of germinal center in lupus prone mice after treatment with a small inhibitor of nuclear export KPT-350
Regulation of Tolerance in Bone Marrow B Cell Development in SLE
Systemic Lupus Erythematous (SLE) is a classic autoimmune disease with complex pathogenesis involving abnormalities in both the adaptive and innate immune system, with loss of B cell tolerance a key event in early disease development and possibly an important factor in ongoing amplification of disease. B cell development is a carefully regulated process that involves the sequential differentiation of bone marrow (BM) precursors into immature and transitional B cells, with auto-reactivity normally censored at these early stages. We hypothesize that environmental signals in the bone marrow play a critical role in shaping B cell development and these signals maybe aberrant in SLE contributing to loss of tolerance and ongoing B cell activation during B cell development. We have recently demonstrated type-I IFN activation in human and murine SLE BM. The IFN activation was associated with higher autoantibodies and transitional B cell expansion. We found that neutrophils were a prime producer of IFN in the SLE BM and other key mediators that likely affect B cell development. Ongoing work is examining the impact of the larger family of IFNs and other factors on B cell tolerance using single cell molecular approaches.
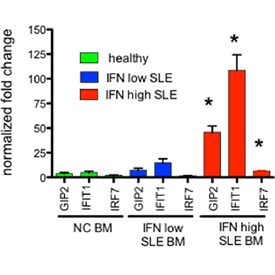
Figure legend:
Lupus patients have a type I interferon signature in the bone marrow. We hypothesize that this disturbed microenvironment impact developing cells including B cells and mesenchymal stem cells.
The Role of B Cells in the Pathogenesis of Autoimmune Disease
Our research focus is to understand the underlying pathogenic mechanisms of autoimmune diseases, specifically systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA), on the role of B cells in the development and progression of these diseases. Autoimmune diseases are characterized by the presence of antibodies against self-antigens. As an antibody producing cell, the B cell is a key player in the pathogenesis of autoimmune diseases. However, B cells also have other functions beyond antibody production. Our research focus is to investigate autoantibody independent functions of B cells including interaction with other immune cells and other relevant organ systems. Our research focus in RA is to understand the mechanism of how B cells interact with osteoclasts and osteoblasts to contribute to bone erosion process in this disease.
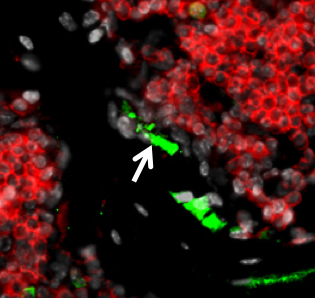
Figure legend:
B cells interact with OCN+ osteoblasts to inhibit new bone formation, thus contributing to erosion progression and joint damage