Research Projects
Metabolic Changes in MSCs During Differentiation
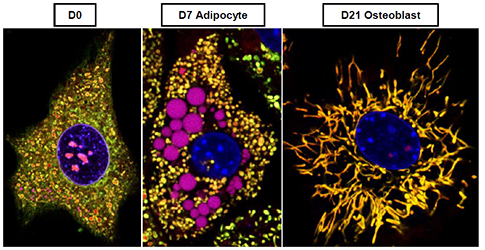
The goal of this project is to determine the mechanism of activation of mitochondria in MSCs during osteogenic differentiation. We study human MSCs and mouse MSCs isolated from bone marrow of either wild-type or genetically modified mice. MSC ability to self-renew and differentiate into various lineages (osteogenic, adipogenic and chondrogenic) is being investigated in the context of cell metabolism. We monitor how mitochondrial function, morphology, ultrastructure and proteome as well as cell metabolome and gene expression, change during differentiation into various lineages. We also use gain- and loss-of-function approach to determine what signaling systems are involved in regulation of cell metabolism during differentiation.
MSC Metabolism and Osteogenic Potential During Aging
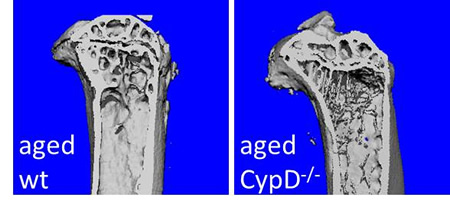
This project is aimed to determine the mechanism of mitochondrial dysfunction in MSCs during aging leading to their decreased osteogenicity, bone loss, and delayed fracture healing. Mitochondrial permeability transition (MPT), a non-selective mitochondrial pore regulated by cyclophilin D (CypD), has been documented as the mechanism of mitochondrial dysfunction in cardiovascular and other systems during aging. The role of the MPT in aged bone has not been elucidated. Our data indicate that the MPT loss of function in CypD knock-out mice leads to improved mitochondrial function and osteogenicity in aged MSCs and less osteoporosis when compared to aged wild type mice. The role of CypD as a potential target to treat osteoporosis and delayed fracture healing and the potential of stem cell therapy involving MSCs with modified CypD function, is being investigated.
Metabolic Reprogramming in Osteosarcoma
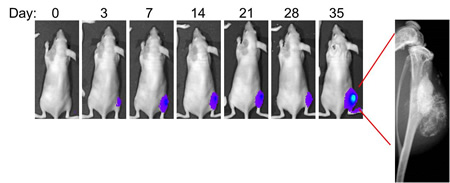
We study how increased glycolytic metabolism and decreased mitochondrial function determine resistance of osteosarcoma cancer stem cells (CSC) to treatments. Our data indicate that CSCs show the most pronounced Warburg effect meaning that they are more glycolytic and less oxidative than the rest of the cells within the tumor. This phenomenon makes them very resistant to apoptosis induced by radiation or other treatments. It also opens a window of opportunity to specifically target these CSCs by shifting their metabolism from glycolytic towards oxidative. We have also found that function of anti- and pro-apoptotic members of the Bcl2 family proteins depends on the level of mitochondrial activity.