Projects
Cellular Mechanisms of Metabotropic Glutamate Receptor Dimer Formation
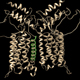 mGluRs function as stable, covalently linked dimers. This means that a functional receptor consists of two mGluR proteins. The proteins can either be identical, called a homodimer, or different, called a heterodimer. While all mGluRs can form homodimers, several can heterodimerize with some other mGluR proteins, but this process is not promiscuous. Only certain combinations of mGluRs can heterodimerize. Moreover, the organization of mGluRs into heterdimers can profoundly alter how the functioning receptors respond to pharmacological ligands.
mGluRs function as stable, covalently linked dimers. This means that a functional receptor consists of two mGluR proteins. The proteins can either be identical, called a homodimer, or different, called a heterodimer. While all mGluRs can form homodimers, several can heterodimerize with some other mGluR proteins, but this process is not promiscuous. Only certain combinations of mGluRs can heterodimerize. Moreover, the organization of mGluRs into heterdimers can profoundly alter how the functioning receptors respond to pharmacological ligands.
Learn more about Cellular Mechanisms of Metabotropic Glutamate Receptor Dimer Formation
Functional Consequences of Heteromeric Receptor Association
 mGluRs are known to function as obligate dimers. We and other labs have shown that the dimeric nature of mGluRs leads to certain pharmacological and functional consequences. For example, unlike most GPCRs which exist mainly as monomers, mGluRs require two agonists bound to each receptor dimer for activation. More recent work from the Kammermeier lab is beginning to show a novel interaction between specific mGluRs (mGluR1&5) in which receptor homodimers appear to interact in a way that each receptor allosterically modulates the function of its partner.
mGluRs are known to function as obligate dimers. We and other labs have shown that the dimeric nature of mGluRs leads to certain pharmacological and functional consequences. For example, unlike most GPCRs which exist mainly as monomers, mGluRs require two agonists bound to each receptor dimer for activation. More recent work from the Kammermeier lab is beginning to show a novel interaction between specific mGluRs (mGluR1&5) in which receptor homodimers appear to interact in a way that each receptor allosterically modulates the function of its partner.
Learn more about Functional Consequences of Heteromeric Receptor Association
The Role of Accessory 2αδ Subunits in the Trafficking and Expression of Voltage Gated Calcium Channels
 Voltage dependent calcium (CaV) channels act as key mediators of several fundamentally important processes in both excitable and non-excitable cells by initiating calcium entry leading to muscle contraction, changes in gene transcription, and vesicular secretion of neurotransmitters or hormones. Expression of the α1, pore forming subunit of CaV channels (as well as some of their biophysical properties: voltage dependence, kinetics, etc.) is highly dependent on the accessory proteins β and α2δ. In short, the β subunits are important in aiding the trafficking of CaV channels through the ER and to the plasma membrane, and α2δs in stabilizing them once there, probably by slowing internalization and aiding in recycling of internalized channel complexes, but specific roles of individual α2δ subunits remain unknown.
Voltage dependent calcium (CaV) channels act as key mediators of several fundamentally important processes in both excitable and non-excitable cells by initiating calcium entry leading to muscle contraction, changes in gene transcription, and vesicular secretion of neurotransmitters or hormones. Expression of the α1, pore forming subunit of CaV channels (as well as some of their biophysical properties: voltage dependence, kinetics, etc.) is highly dependent on the accessory proteins β and α2δ. In short, the β subunits are important in aiding the trafficking of CaV channels through the ER and to the plasma membrane, and α2δs in stabilizing them once there, probably by slowing internalization and aiding in recycling of internalized channel complexes, but specific roles of individual α2δ subunits remain unknown.
Regulation of Metabotropic Glutamate Receptor Signaling by the Post-Synaptic Scaffold Homer
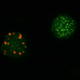 Group I mGluRs (mGluRs 1&5) are the predominant mGluRs expressed near the post-synaptic density at glutamatergic synapses. These receptors are anchored near the PSD by the post-synaptic scaffolding proteins called Homer. Work in the Kammermeier lab has been among the first to show that Homer association can act as a molecular switch for group I mGluR signaling, uncoupling mGluR1&5 from a large set of effectors in the absence of Homer scaffolds (voltage gated ion channels expressed extrasynaptically) to a separate set of effectors located near the PSD (ligand gated glutamate receptors, IP3 receptors, and DAG lipases).
Group I mGluRs (mGluRs 1&5) are the predominant mGluRs expressed near the post-synaptic density at glutamatergic synapses. These receptors are anchored near the PSD by the post-synaptic scaffolding proteins called Homer. Work in the Kammermeier lab has been among the first to show that Homer association can act as a molecular switch for group I mGluR signaling, uncoupling mGluR1&5 from a large set of effectors in the absence of Homer scaffolds (voltage gated ion channels expressed extrasynaptically) to a separate set of effectors located near the PSD (ligand gated glutamate receptors, IP3 receptors, and DAG lipases).