Detailed Research Information
Development of More Effective Cancer Therapies
Our cancer research demonstrates how discovery of general biological principles, integration of multiple discoveries, and analysis of complex medical problems provide the foundation for development of novel therapeutic approaches for challenging afflictions. Read an expanded overview of our cancer research program and a detailed review is provided in this article.
Development of Novel Treatments for Glioblastoma (GBM) that are Suitable for Rapid Translation to Clinical Evaluation
Glioblastomas are among the most deadly of all cancers, with a median survival of 15 months from initial diagnosis even with optimal treatment. They represent some of the most difficult cancers of all to treat, and thus provide a particularly challenging disease on which to test the potency of our general strategy. Intrinsically resistant to known therapies, they also contain some of the best characterized of cancer stem cell populations – which are even more resistant to known treatments yet must be eliminated if new tumors are not going to rapidly emerge. GBM cells also migrate extensively in the CNS, so that surgical removal of the tumor is still followed quickly by tumor recurrence. Thus, treatments must target the GBM stem cells as well as other cells of the tumor and must be able to pass through the blood-brain barrier to reach cells far removed from the tumor’s primary origin. Moreover, all this must occur without causing unacceptable levels of damage to the normal cells of the central nervous system.
Attempts to develop new treatments for GBM have been ongoing for many years, and thus far have been unsuccessful. The lack of success of multiple efforts to develop more effective treatments for GBM could mean that these tumors are inherently not treatable, or that strategies employed thus far are incorrect. We think the latter hypothesis is the correct one – but how can we do better?
Our development of promising novel treatments for GBM, which are suitable for rapid transition to clinical analysis, was achieved via the following discoveries:
- We discovered a novel regulatory pathway, called the redox/Fyn/c-Cbl pathway that controls multiple aspects of normal glial progenitor cell function. In this pathway, small increases in oxidative status sequentially activate Fyn kinase and the c-Cbl ubiquitin ligase. The ability to activate c-Cbl via this pathway is of great interest because c-Cbl regulates the degradation of multiple receptors and other proteins of critical importance in the function of stem cells, progenitor cells and cancer cells. From NIH - Chemically Diverse Toxicants Converge on Fyn and c-Cbl to Disrupt Precursor Cell Function.
- There is no point in developing anticancer agents that cause unacceptable levels of damage to normal tissue, a problem of particular importance in the central nervous system. As part of our studies on the toxicity of standard anti-cancer therapies, we discovered that multiple existing chemotherapeutic agents are more toxic for the normal progenitor cells and oligodendrocytes of the central nervous system than they are for human cancer cells. These findings appear to provide the cellular foundations underlying the neurological complications that are far too frequent in patients treated with systemic chemotherapy. They also provide a starting point for discovering the reasons for these differences in vulnerability, and for development of potential protective strategies.
- From NIH - CNS progenitor cells and oligodendrocytes are targets of chemotherapeutic agents in vitro and in vivo
- From NIH - Systemic 5-fluorouracil treatment causes a syndrome of delayed myelin destruction in the central nervous system
- From NIH - MEK1/2 inhibition suppresses tamoxifen toxicity on CNS glial progenitor cells
- The necessary next question is how to attack GBM cells without also targeting normal cells of the brain. We discovered that GBM cells prevent c-Cbl activation, including activation via the redox/Fyn/c-Cbl pathway, by producing a protein (called Cool-1/ß-pix) that forms a complex with c-Cbl in tumor biopsies and tumor cells, but not in normal brain tissue or normal glial progenitor cells. This feature of GBM biology distinguishes these cells from normal brain cells. Cool-1-mediated inhibition of c-Cbl modulates multiple critical properties of glioblastomas, including the ability to generate tumors in vivo
- Cool-1 knockdown restores normal c-Cbl regulation suppresses GBM cell division and increases sensitivity to chemotherapeutic agents – but Cool-1 knockdown does not suppress division or increase chemosensitivity of normal glial progenitor cells. Thus, this appears to be a specific vulnerability of the cancer cells. Cool-1-mediated inhibition of c-Cbl modulates multiple critical properties of glioblastomas, including the ability to generate tumors in vivo
- Restoring c-Cbl activity by Cool-1 knockdown eliminates the ability of GBM cells to form tumors, and otherwise eliminates cancer stem cells. Thus, inhibition of c-Cbl appears to be essential in order to generate a GBM. Cool-1-mediated inhibition of c-Cbl modulates multiple critical properties of glioblastomas, including the ability to generate tumors in vivo
- Restoring normal c-Cbl regulation enables simultaneous targeting of multiple proteins of critical importance in GBM biology (and particularly in GBM stem cell biology), including the epidermal growth factor receptor, Notch-1, ß-catenin,Sox2 and Cd133. In contrast with other therapeutic approaches, our therapies enable co-ordinated attack on multiple targets of interest with a single intervention. Cool-1-mediated inhibition of c-Cbl modulates multiple critical properties of glioblastomas, including the ability to generate tumors in vivo
- We have discovered pharmacological agents that enable us to restore normal c-Cbl function in GBM cells. Effects of combining these agents with standard of care for these tumors are extremely promising in stringent mouse models of human GBM.
Our discoveries are also applicable to other cancers. We found that basal-like breast cancer cells also inhibit c-Cbl, and this inhibition also is critical for cancer stem cell function in these tumors. From NIH - Inhibition of redox/Fyn/c-Cbl pathway function by Cdc42 controls tumour initiation capacity and tamoxifen sensitivity in basal-like breast cancer cells
We also have promising results in several other types of cancer.
Finally, in collaboration with Dr. Margot Mayer-Pröschel, we also have been studying the problem of treating lymphomas in a mouse model of the human genetic disease ataxia telangiectasia, where our therapeutic approaches also appear to offer significant promise in providing improved anticancer therapies. Treating the tumors that arise in individuals with ataxia telangiectasia is an even more difficult problem than is the case for treatment of most cancers, as individuals with this disease have mutations in an enzyme (called ATM) of vital importance in repairing DNA damage. Moreover, ataxia telangiectasia is associated with ongoing degeneration in parts of the central nervous system. Thus, treating these cancers without causing unacceptable levels of damage is even more difficult than is usually the case.
Diseases of Lysosomal Dysfunction
The success of our studies on the development of potential new therapies for GBM and other cancers by integrated analysis of fundamental biological principles raises the question of whether we have developed research strategies that are equally applicable to other difficult medical problems.
One of the areas that we have targeted successfully is treatment of lysosomal dysfunction. We chose this particular challenge because such dysfunction is emerging as a critical contributor in multiple diseases, such as Parkinson’s disease, Alzheimer’s disease, diabetes – and is also at the heart of understanding devastating developmental diseases called lysosomal storage disorders. Lysosomal dysfunction also is an important component of aging.
We initiated this research by targeting lysosomal storage disorders, the most severe diseases of lysosomal dysfunction. Our studies have revealed new mechanisms of damage in these diseases, new treatment strategies and novel therapeutic agents.
More details will be added as papers are accepted for publication.
Treatment of Acute and Chronic Injury in the Central and Peripheral Nervous System
Astrocyte Transplantation Therapies
In collaboration with Dr. Chris Pröschel, we are helping to develop cell-based therapies for treating spinal cord injury and Parkinson’s disease. Please refer to the Pröschel Lab for more details on these studies and to our previous publications and reviews:
- From NIH - Astrocytes derived from glial-restricted precursors promote spinal cord repair
- From NIH - Transplanted astrocytes derived from BMP- or CNTF-treated glial-restricted precursors have opposite effects on recovery and allodynia after spinal cord injury
- From NIH - Transplantation of specific human astrocytes promotes functional recovery after spinal cord injury
- From NIH - Precursor cell biology and the development of astrocyte transplantation therapies: lessons from spinal cord injury
- Cell therapies for the central nervous system: how do we identify the best candidates?
Promoting tissue repair with drugs suitable for rapid translation from the laboratory to clinical studies
Two of the central goals of regenerative medicine are to discover drugs that enhance the body’s own repair processes and that can be moved rapidly from the laboratory to clinical studies. Although cell replacement therapies will be necessary for many conditions, pharmacological approaches to tissue repair are likely to be easier and less expensive to implement, and thus may enable more rapid progress on a range of important medical needs.
One example of the power of pharmacological enhancement of repair by endogenous cells is provided by our research on overcoming damage to the peripheral nervous system (PNS).
The PNS is the essential connector that enables the brain and/or spinal cord to control the function of target organs throughout the body. If we consider muscle function as an example of this relationship, a complete injury to the sciatic nerve (PNS) that innervates muscles of the leg has the same functional outcome as a complete injury to that part of the spinal cord (CNS) or that part of the brain (CNS). Thus, attempts to repair the CNS need to also consider whether repair of the PNS is also going to be required.
Injuries to the PNS are frequent. In the case of some CNS injuries, such as spinal cord injury (SCI), most cases of traumatic injury are also associated with PNS injury. The PNS can also be injured in many other ways than traumatic accidents, however. For example, several different anti-cancer treatments, multiple genetic diseases, some autoimmune diseases and even such afflictions as diabetes all can cause debilitating PNS damage involving loss of motor and/or sensory function.
Promoting tissue repair by pharmacological harnessing of the body’s own repair capacity and discovering new properties of an old drug
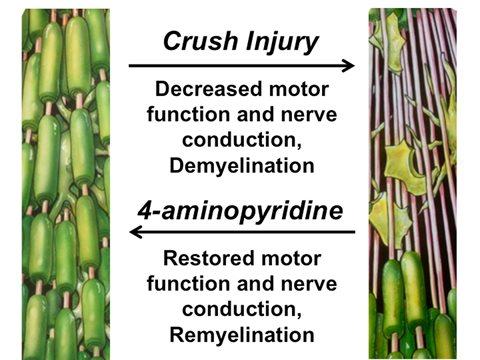
Working together with Dr. John Elfar, we have developed promising new approaches to PNS repair by discovering previously unknown effects of an old drug, called 4-aminopyridine (4AP). 4AP is a potassium channel blocker that has long been known to increase synaptic efficacy. Based on these activities, people started studying it in the early 1980s as a treatment for syndromes in which synaptic function is decreased, such as myasthenia gravis. Moreover, 4AP is used to treat the problem that leakage of potassium out of axons that have lost their myelin renders those axons unable to conduct impulses. By blocking potassium channels, 4AP restores the ability of demyelinated axons to conduct impulses. For this reason, 4AP was also studied in the early 1980s as a means of improving neurological function in patients with multiple sclerosis (MS), and has been approved by the FDA as a treatment for walking disability in these patients.
In studies published in EMBO Molecular Medicine, we discovered that application of 4AP in acute traumatic peripheral nerve injuries is able to cause a durable recovery that remains after treatment is stopped – a type of outcome never before seen with this drug. In chronic injuries, to the CNS or PNS, any benefit provided by 4AP is dependent on the continued presence of drug. In contrast, when we used 4AP to treat acute crush injuries of the sciatic nerve - the major peripheral nerve that connects the spinal cord with muscles and sensory organs of the leg - we caused durable improvements in motor function and electrophysiological function that did not require the constant presence of drug.
We further discovered that treatment of sciatic nerve crush injuries with clinically relevant dosages of 4AP promoted repair of the myelin damage that is an important component of such injuries. Moreover, nerve conduction velocity – which is regulated to a large extent by myelination - improved much more quickly in 4AP-treated mice than occurred in saline-treated mice.
Thus, 4AP provides a small molecule regenerative agent that is clinically relevant and that is able to promote remyelination by enhancing the repair functions of the body’s own cells.
Correct diagnosis is the essential first step to assigning the correct treatments: An additional use of 4AP.
We also found that treatment with 4AP may improve the ability to diagnose different types of nerve lesions, a potentially critical advance in a field that has been relatively unchanged for a long time.
When someone presents in the clinic with nerve damage, the surgeon needs to know if there are axons that traverse the lesion site or whether all axons have been cut - because the treatments for these two types of lesions are mutually exclusive. If there are intact axons, the current therapy is to leave things alone as there is a chance of at least some recovery of function. If all axons are cut, the surgeon needs to clean up the ends of the nerve and rejoin them if there is going to be any chance of recovery.
The problem is that current approaches to diagnosis rely on “watchful waiting,” often for weeks or even months. If an injury gets better spontaneously, then it was right to leave it alone. If it does not get better spontaneously, then surgical rejoining of nerve ends would have been the right thing to do - but the delay in doing so has now decreased the likelihood that the surgery is going to provide benefit.
In contrast with existing approaches, we can tell the difference between a crushed nerve and a cut nerve 24 hours after injury. A single treatment with 4AP 24 hours post-injury causes readily detectable motor improvements if the sciatic nerve is crushed, and no such benefits if the nerve is cut. As indications of benefit are seen rapidly when 4AP is used in people with chronic injuries or illnesses, we are hopeful that we can use this drug to improve our diagnosis of human injuries in similar ways.
4AP and the promise of pharmacological approaches to regenerative medicine
4AP appears to occupy a somewhat unique space in the field of regenerative medicine. The drug that we would use to diagnose peripheral nerve injury patients who would benefit from promoting remyelination – the necessary first step in implementing treatment - is the same drug that we would then use to enhance such repair. Having a single drug with the potential of improving both diagnosis and treatment is exactly the kind of goal to which we aspire.
Our studies thus offer a promising example of promoting tissue repair by pharmacological enhancement of endogenous repair processes.
Our new discoveries on 4AP were accomplished with a small molecule that has been extremely well studied in humans for over 30 years, but only in the context of chronic injury. Those 30 years of prior work offered no prediction of our present outcomes because treatment of chronic injuries or illnesses offers no benefits that remain when drug treatment is stopped. Thus, in the case of chronic injury there is no evidence of regenerative activities of 4AP.
In contrast, analysis of the effects of 4AP in the acute and subacute post-injury period revealed benefits in peripheral nerve injury that remain when drug is no longer present. Nonetheless, even though prior studies on 4AP were not predictive of our present outcomes, they do provide information that is useful in moving forward to the clinic. As with all of our drug repurposing efforts, existing information on such topics as dosage, toxicity, drug metabolism, tissue penetrance and oral availability all is valuable in decreasing the costs of clinical development.
Now that we have the possibility of improving both the diagnosis and treatment of peripheral nerve injuries, the goal is to move these discoveries into the clinic as quickly as possible.
More details on our pharmacological approaches to treatment of neurological injury will be added as papers are accepted for publication.