Research Projects
Engineered Salivary Gland Tissue Chips
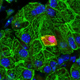
 For more than 550,000 patients annually diagnosed with head and neck cancers worldwide, severe loss of salivary gland function (xerostomia) is an unavoidable outcome of radiation therapy. Efforts to discover effective radioprotective and regenerative strategies have been hampered by the inability to culture salivary gland secretory cells in vitro, due to loss of the secretory cell phenotype.
For more than 550,000 patients annually diagnosed with head and neck cancers worldwide, severe loss of salivary gland function (xerostomia) is an unavoidable outcome of radiation therapy. Efforts to discover effective radioprotective and regenerative strategies have been hampered by the inability to culture salivary gland secretory cells in vitro, due to loss of the secretory cell phenotype.
Learn more about Engineered Salivary Gland Tissue Chips
Homeostasis and Regeneration
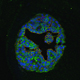
 We are working to identify cells involved in maintenance and regeneration of head and neck exocrine glands. We recently demonstrated that salivary gland homeostasis is based predominantly on self-duplication of differentiated secretory acinar cells. However, the mechanisms – and cell types- involved in gland regeneration remain unresolved.
We are working to identify cells involved in maintenance and regeneration of head and neck exocrine glands. We recently demonstrated that salivary gland homeostasis is based predominantly on self-duplication of differentiated secretory acinar cells. However, the mechanisms – and cell types- involved in gland regeneration remain unresolved.
Learn more about Homeostasis and Regeneration
Radiation Damage and Radioprotective Strategies
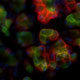
 The informed design and application of radioprotective strategies first requires a clear understanding of mechanisms underlying salivary gland damage. We use genetic mouse models, complemented with bioengineered tools, to address the unanswered questions regarding loss of salivary gland function, and in continuing efforts to design improved radioprotective strategies
The informed design and application of radioprotective strategies first requires a clear understanding of mechanisms underlying salivary gland damage. We use genetic mouse models, complemented with bioengineered tools, to address the unanswered questions regarding loss of salivary gland function, and in continuing efforts to design improved radioprotective strategies
Learn more about Radiation Damage and Radioprotective Strategies
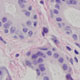
Specialized Cells in the Salivary Gland
The salivary gland ducts serve as an epithelial barrier between secretory components of the gland and the oral cavity. An essential function of such a barrier is the detection and response to external insults. In many tissues, scattered single chemosensory cells (SCCs) function as sentinels, which relay external cues into transduced signals that activate an immune response.
We are investigating whether SCCs in the salivary glands play a critical role in mounting an immune response, and might be involved in the mechanisms leading to acute and chronic inflammation.