Methylmercury Metabolism and Elimination Status (MerMES) in Humans
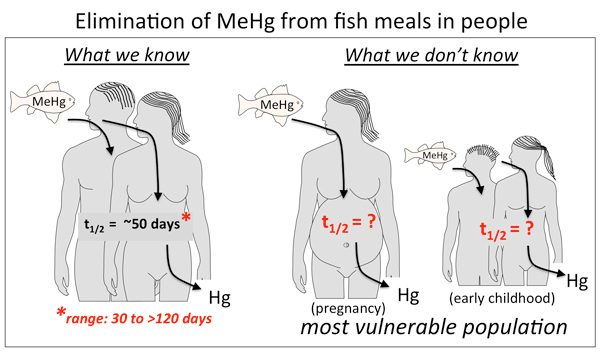
Evaluating the potential for methylmercury (MeHg) toxicity relies on accurately predicting the mercury (Hg) body burden that results with eating fish. The Hg body burden is directly determined by the slow elimination kinetics of MeHg in the human body (t1/2~ 50days). Existing studies are limited in size and number, yet show that the biological half-life of MeHg in humans can vary widely (t1/2 = 30 to >150days). Since the half-life directly determines how much MeHg accumulates from repeated exposures (e.g. with routine fish eating) the variation in half-life documented to date has instilled great uncertainty in the derivation of a MeHg Reference Dose (RfD) for recommended daily intake. We predict that MeHg toxicokinetics in humans is subject to a large degree of naturally occurring biological variation that is influenced by genetic background, the gut microbiome and dietary co-exposures. Understanding what contributes to individual differences in MeHg metabolism and elimination could lead to more concrete methods of identifying a susceptibility to MeHg toxicity, and better inform patients on MeHg exposure from fish on an individual basis.
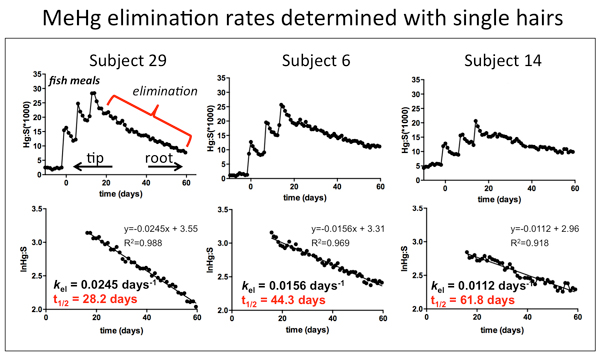
We recently characterized variation in human MeHg metabolism and elimination rate (half-life) in 37 adult volunteers who consumed three fish meals. MeHg elimination rates were determined via longitudinal Hg analysis of single hairs from each subject using laser ablation ICP-mass spectrometry. MeHg metabolism to inorganic mercury (I-Hg, biotransformation) was measured via speciation of Hg in feces collected from the subjects. We found an average half-life of t1/2 = 44days across the 37 subjects, which is 5 days faster than the value used in the current EPA RfD. However, half-life across all the subjects varied more than two-fold (t1/2 = 28-62days). Overall, the rate of MeHg biotransformation showed a positive association with elimination rates in the subjects, indicating that “de-methylation” of MeHg results in more rapid excretion and supporting the hypothesis that the gut microbiome plays a role in the biotransformation and elimination of MeHg from the human body.
Our studies will continue to characterize MeHg metabolism and kinetics in people, focusing next on the most vulnerable populations: pregnant women and young children. In parallel, we are investigating the gut microbiome in more depth for its role in the biotransformation process. Finally, we are using our data to inform development of a physiologically based pharmacokinetic (PBPK) model in silico capable of predicting MeHg kinetics in people.