Epigenetic Mechanisms Underlying Behavioral Toxicity
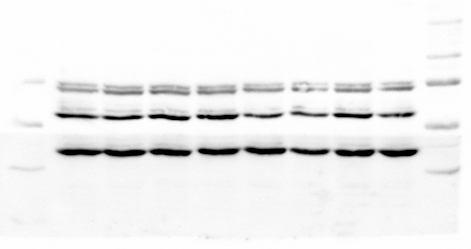
 We have long known that altered developmental exposure to steroid hormones can alter behavior into adulthood; however, the mechanisms of cellular transmission were unknown. Increasingly, epigenetic mechanisms of cellular memory across the lifespan have been identified. In collaboration with Dr. Deborah Cory-Slechta, we study how developmental endocrine disruption through exposure to lead (Pb), prenatal stress (PS), and their combination alter epigenetic profiles in mice, including DNA methyltransferase concentrations, post translational histone modifications, and DNA methylation, in the frontal cortex and hippocampus across the lifespan. Importantly, the epigenetic alterations produced by Pb exposure are significantly modified by the type of behavioral experiences that mice are subsequently exposed to. Our lab is developing a model of behavioral experience as a secondary ‘hit’ for neurodevelopment. We are beginning to understand the epigenetic consequences of behavioral experience alone. These data underscore the potential for behavior to modify neurotoxicity. Such research has substantial promise for identifying mechanisms by which behavioral experiences can act as protective factors or as risk factors for brain and behavioral function.
We have long known that altered developmental exposure to steroid hormones can alter behavior into adulthood; however, the mechanisms of cellular transmission were unknown. Increasingly, epigenetic mechanisms of cellular memory across the lifespan have been identified. In collaboration with Dr. Deborah Cory-Slechta, we study how developmental endocrine disruption through exposure to lead (Pb), prenatal stress (PS), and their combination alter epigenetic profiles in mice, including DNA methyltransferase concentrations, post translational histone modifications, and DNA methylation, in the frontal cortex and hippocampus across the lifespan. Importantly, the epigenetic alterations produced by Pb exposure are significantly modified by the type of behavioral experiences that mice are subsequently exposed to. Our lab is developing a model of behavioral experience as a secondary ‘hit’ for neurodevelopment. We are beginning to understand the epigenetic consequences of behavioral experience alone. These data underscore the potential for behavior to modify neurotoxicity. Such research has substantial promise for identifying mechanisms by which behavioral experiences can act as protective factors or as risk factors for brain and behavioral function.
In addition, in collaboration with Dr. Martha Susiarjo, we have begun following these epigenetic changes across generations. Our research has shown significant and sex-dependent transgenerational effects of Pb, prenatal stress, and their combination. We aim to track the direct cellular and environmental mechanisms of transmission for behavioral variability across generations. This includes examining the role lineage and sex play in the transmission of a phenotype.
These studies will serve as the basis for defining epigenetic mechanisms by which developmental exposure to neurotoxicants, including EACs such as Pb, ultimately impact cognitive function. Such studies can be used translationally to define behavioral experiences that may act as a positive intervention for cognitive deficits following developmental EAC exposures, possibly erasing prior epigenetic programming, and thereby improving future behavioral function.