Transgenic Technology
Current Transgenic Technology
Plucky, a X. laevis transgenic albino
expressing the green fluorescent
protein (GFP) in her eye.
Kindly provided by D. Papermmaster.
Known as restriction enzyme-mediated insertion (REMI), this method involves mixing transgene DNA with purified and permeabilized sperm, along with a small amount of restriction enzyme and a high-speed interphase egg extract. This mixture is then injected into unfertilized dejellied eggs (Kroll & Amaya, 1996). The extract partially decondenses the sperm chromatin but does not promote replication and the restriction enzyme stimulates recombination and integration by creating double stranded breaks in the sperm chromatin. This technique permits large-scale transgenesis in X. laevis embryos. Unlike embryos injected with plasmids, the transgenic embryos show correct spatial and temporal regulation of integrated promoter constructs. It is generally considered that one of the great advantages of this system in X. laevis over transgenesis in mice or zebrafish is that the transgene is usually integrated into the male genome prior to fertilization. Therefore, the resulting embryos are not chimeric and breeding of animals is not required. However, exchange of genetic material between male and female genomes as well as multiple gene insertions have been observed (Kobel & Du Pasquier, pers, comm.)
Generation Of Transgenic Clones
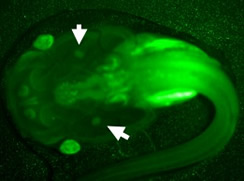
Generation of transgenic isogenetic X. laevis clone with the "Sleeping Beauty"
transposase. Dejellied LG-6 eggs were co-injected with
102 ng of transposase mRNA and 15 ng in 10mL volume of vector
containing a GFP reporter transgene under the control of the
X. laevis Ef-1α promoter. Live larvae were screened for GFP expression
with a fluorescence stereomicroscope at a pre-metamorphic
stage (st 56, 1 month old) Arrows: thymus
We are adapting transgenic (Tg) technology to our X. laevis clones (e.g., LG-6, LG-15) for immunological studies (reviewed in Robert et al., 2009). The advantage of using these clones is that they are MHC defined. Furthermore, since progeny from cloned Tg founders are produced by gynogenesis (UV-irradiated spermatozoa are used to activate diploid eggs and do not contribute any genetic material to the offspring), no time consuming screening is required. Three different Tg techniques that allow the direct insertion of transgenes into X. laevis genomic DNA are currently used in our lab: the φC31 integrase (Allen & Weeks,2005), the I-Sce meganuclease (Pan et al., 2006; Ogino et al.., 2006), and the transpossae "Sleeping Beauty" (Sinzelle et al., 2006). These techniques are used to express reporter GFP genes under the regulation of immunologically relevant genes, over-expressing or knocking down by siRNA immunologically relevant genes.
RNAi-Mediated Loss Of Function By Transgenesis With Meganuclease
Fig. 1: RNAi-mediated loss of function by transgenesis.
(A) Map of the double promoter expression cassette vector
used for I-SceI meganuclease transgenesis.
Representative F0 transgenic tadpole (B) and
young adult (C) of the isogenetic clone LG-15
showing uniform constitutive expression of the green fluorescence protein.
A powerful reverse genetic approach has been developed Xenopus, which combines transgenesis with RNA interference for studying immune functions (Nedelkovska et al., 2013; Nedelkovska and Robert, 2012).
Transgenic Xenopus with knockdown (KD) of expression of a gene of interest are generated using a double expression cassette vector flanked by I-SceI meganuclease recognition sequences that contains the shRNA targeting the gene of interest under the control of the human U6 RNA polymerase III promoter (hU6) and a GFP reporter gene driven by the human EF-1α promoter (Fig. 1A). This construct is digested with I-SceI meganuclease and the reaction is then microinjected into one cell stage eggs. This technique not only works with fertilized outbred or inbred eggs but also with diploid isogenetic LG eggs activated by gynogenesis. The injected embryos are reared until tadpole stage 56 and then screened for GFP expression by fluorescence microscopy.
The I-SceI meganuclease Tg technique is very efficient (35-50% Tg efficiency and high survival rate >35%), and results in bright nonmosaic GFP expression as well as stable germline transmission with100% of the progeny carrying the transgene. A key advantage of this technique is that the high efficiency and nonmosaic transgene expression permit the direct use of F0 animals. Typically Tg animals are selected at larval stage 55-56 (3-4 weeks of age) and controlled for effective KD by qPCR using a tail clip. A fraction of the animals are maintained as founders for F1 progeny while the remaining are used for research.
Currently, our resource maintains several X. laevis lines and LG clones including both F0 founder and F1 generations with knockdown of β2-microglobulin (b2m; globally affecting b2m, MHC class Ia and class Ib surface expression) and nonclassical MHC class Ib XNC10 as well as F0 animals with knockdown of XNC11and XNC14. This gene loss-of-function method can be applied to non-immune genes and has the advantage of morpholinos to work at late stage of development and in F0 adults.
References
Nedelkovska, H., Edholm, E.S., Haynes, N., Robert, J., 2013. Effective RNAi-mediated beta2-microglobulin loss of function by transgenesis in Xenopus laevis. Biol Open 2, 335-342.
Nedelkovska, H., Robert, J., 2012. Optimized transgenesis in Xenopus laevis/gilli isogenetic clones for immunological studies. Genesis 50, 300-306.