Triploidy
Triploidy
Clinical History
A 32-year-old woman is in her 20th week of gestation. Four weeks prior she noted bright red vaginal bleeding and was placed on bed rest. Ultrasound studies revealed decreased amniotic fluid volume, and prominent lateral ventricles. Amniocentesis was performed and confirmed cytogenetic abnormalities, as discussed below. A repeat ultrasound was done which diagnosed fetal demise and dilation and extraction was performed.
Seven months later, the same woman underwent a spontaneous abortion of a 12-week fetus in her subsequent pregnancy.
Gross Examination
Gross examination of the first fetus revealed a 17-week fragmented female fetus (20 weeks by dating). Of the identifiable tissue, right micropthalmia, syndactyly of digits 3-4 on both hands, and syndactyly of digits 4-5 on the right foot and 3-4 on the left foot were observed.
The second fetus was consistent with a 12-week macerated fetus of indeterminable gender. There was bilateral syndactyly of the 3rd and 4th digits.
Microscopic Examination
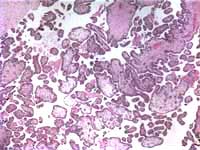
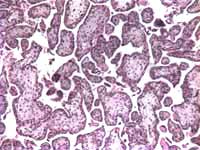
Placenta, umbilical cord, both eyes, and non-descript tissue of the first fetus was sent for histologic review. Fetal tissue and the placenta, which was consistent with a second trimester placenta, contained no pathological abnormalities.
The only histological findings of the second fetus were some mildly hydropic villi (Figures 1 and 2). Neither pregnancy had evidence of a partial hydatidiform mole.
Click an image to view larger format.
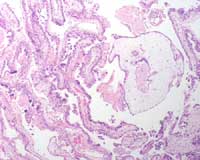
Figure 3: H & E stain of a partial
hydatidiform mole (different case);
note the large hydropic villus with a
trophoblastic inclusion surrounded
by smaller villi
Cytogenetics
Karyotype analysis of both amniotic fluid and placental tissue of the first fetus showed a 69, XXX (triploid) karyotype.
Karyotype analysis of placenta and skin of the second fetus showed a second triploidy, with 69, XXY karyotype.
Discussion
Triploidy syndrome refers to a complete extra set of haploid chromosomes in the embryo. The syndrome is estimated to occur in 1 - 2% of conceptuses. Most conceptuses with triploidy syndrome are lost as miscarriages and they account for about 20 per cent of all chromosomally abnormal spontaneous abortuses. For every triploid conceptus that survives to term, approximately 1200 conceptuses result in fetal death. The extra haploid set can be derived from maternal (digynic) or paternal (diandric) origins. Digynic triploidy result from fertilization of a diploid ovum by a haploid sperm and can therefore result in a XXX, or XXY genotype. Diandric triploidy can result from fertilization of a haploid ovum by either a diploid sperm, or by two haploid sperm and can result in a XXX, XXY, or XYY genotype. The majority of triploid cases are of paternal origin, with 66% attributed to fertilization by two sperm, 24% due to fertilization with a diploid sperm, and 10% resulting from fertilization of a diploid oocyte.
Phenotypically, diandric triploidy conceptuses are associated with an enlarged placenta that is histology consistent with a partial hydatidiform mole (see attached figure 3). In the case described above, the triploid conceptuses occurred in subsequent pregnancies, and neither showed evidence of a partial hydatidiform mole suggesting that the triploidy was attributable to maternal origin. A suggested maternal mechanism resulting in triploidy is nondisjunction in meiosis I or meiosis II of oogenesis.
The triploidy syndrome can result in many congenital abnormalities including: micrognathia, prominent forehead, dysplastic cranial bones, low-set malformed ears, microphtalmia, hypertelorism, cleft lip and palate, macroglossia, omphalocele, syndactyly and asymmetric development. Of all cases of triploidy, the live birth rate is 1/10,000 live births, and those children are severely mentally retarded. Triploidy usually results in fetal death in-utero. Of those that are live born, there are no reported survivors beyond 10.5 months.
Derek Masden, Student Fellow 2004
References
- Jones, KL. Smith’s Recognizable Patterns of Human Malformation, 5th ed, WB Saunders Company, Philadelphia 1997.
- Jacobs PA, Szulman AE, Funkhouser J, Matsuura JS, Wilson CC. Human triploidy: relationship between parental origin of the additional haploid complement and development of partial hydatidiform mole. Ann Hum Genet 1982;46:223-231
- Sherard J, Bean C, Bove B, DelDuca V Jf, Esterly KL, Karcsh HJ, Munshi G, Reamer JF, Suazo G, Wilmoth D, Dahlke MB, Weiss C, Borgaonkar DS. Long survival in a 69, XXY triploid male. Am J Med Genet 1986;25:307-312.
- Cotran, R, Kumar, V, Collins, T, et al. Robbins Pathologic Basis of Disease. 1999; 741-743.
- Pergament E, Confino E, Zhang JX, Roscetti L, Chen PX, Wellman D. 2000. Recurrent triploidy of maternal origin. Prenat Diagn 20: 561-563.
- http://www.tdh.state.tx.us/tbdmd/risk/risk24-triploidy.htm
- http://www.nlm.nig.gov/mesh/jablonski/syndromes/syndrome214.html