Projects
The Antioxidant Glutathione (GSH)
 Discovered more than a century ago (De Rey-Pailhade, J., 1885), the most abundant antioxidant in the cell is glutathione (GSH). A tripeptide composed of glutamate, cysteine and glycine, the rate-limiting, non-redundant step in GSH synthesis is the condensation of glutamate and cysteine. This step is governed by a holoenzyme called glutamate-cysteine ligase which consists of a catalytic subunit (GCLC) and modifying subunit (GCLM).
Discovered more than a century ago (De Rey-Pailhade, J., 1885), the most abundant antioxidant in the cell is glutathione (GSH). A tripeptide composed of glutamate, cysteine and glycine, the rate-limiting, non-redundant step in GSH synthesis is the condensation of glutamate and cysteine. This step is governed by a holoenzyme called glutamate-cysteine ligase which consists of a catalytic subunit (GCLC) and modifying subunit (GCLM).
Learn more about the Antioxidant Glutathione (GSH)
GSH and Cancer Initiation
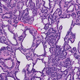 There has been a long-standing belief that supplementation with antioxidants could prevent or treat cancer. However, results of large-scale clinical trials have proven the opposite: supplementation with antioxidants promote, rather than prevent, cancer. While these findings were indisputable, the mechanism behind these surprising results was lacking. A mechanism explaining the failure of antioxidants in preventing cancer was provided when it was demonstrated that the antioxidant GSH was required for cancer initiation (Harris, I.S., Cancer Cell, 2015).
There has been a long-standing belief that supplementation with antioxidants could prevent or treat cancer. However, results of large-scale clinical trials have proven the opposite: supplementation with antioxidants promote, rather than prevent, cancer. While these findings were indisputable, the mechanism behind these surprising results was lacking. A mechanism explaining the failure of antioxidants in preventing cancer was provided when it was demonstrated that the antioxidant GSH was required for cancer initiation (Harris, I.S., Cancer Cell, 2015).
Learn more about GSH and Cancer Initiation
GSH and Protein Homeostasis
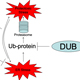 While GSH synthesis was required for tumor initiation, it was found that reducing GSH levels in established tumors was not sufficient to inhibit tumor growth. In this context, additional pathways provided a backup system allowing cancer cells to survive in the absence of GSH. To interrogate these support systems, a high-throughput pharmacologic screening technique was developed to study vulnerabilities in cancer cells. Denoted Multifunctional Approach to Pharmacologic Screening (MAPS, below), enables users to determine the cellular response to perturbations of hundreds of molecular pathways.
While GSH synthesis was required for tumor initiation, it was found that reducing GSH levels in established tumors was not sufficient to inhibit tumor growth. In this context, additional pathways provided a backup system allowing cancer cells to survive in the absence of GSH. To interrogate these support systems, a high-throughput pharmacologic screening technique was developed to study vulnerabilities in cancer cells. Denoted Multifunctional Approach to Pharmacologic Screening (MAPS, below), enables users to determine the cellular response to perturbations of hundreds of molecular pathways.
Learn more about GSH and protein homeostasis
MAPS High-throughput Technology
 The Multifunctional Approach to Pharmacologic Screening (MAPS) is a high-throughput pharmacologic screening technique used by the Harris Lab to study vulnerabilities in cancer cells. This platform enables users to quickly and easily determine the cellular response to perturbations of hundreds of molecular pathways. The MAPS compound library is comprised of more than 500 inhibitors of cancer-related proteins as well as lesser studied metabolic enzymes. In contrast to traditional screening approaches, the MAPS library contain compounds arrayed across 10 dilution points, allowing for a robust understanding of the differential drug sensitivities.
The Multifunctional Approach to Pharmacologic Screening (MAPS) is a high-throughput pharmacologic screening technique used by the Harris Lab to study vulnerabilities in cancer cells. This platform enables users to quickly and easily determine the cellular response to perturbations of hundreds of molecular pathways. The MAPS compound library is comprised of more than 500 inhibitors of cancer-related proteins as well as lesser studied metabolic enzymes. In contrast to traditional screening approaches, the MAPS library contain compounds arrayed across 10 dilution points, allowing for a robust understanding of the differential drug sensitivities.
Learn more about MAPS High-throughput Technology
Genetically-Engineered Mouse Models
 For decades, genetically engineered mouse models (GEMMs) have been used to better understand cellular processes and their effects within a living animal. GEMMs can be used to study normal physiology, such as mammary gland developed (right), as well as pathophysiology, such as the MMTV-PyMT mouse model that spontaneously develops breast cancer. In the Harris lab, we use a variety of GEMMs to understand the roles of antioxidants in disease, with a focus on cancer development and progression.
For decades, genetically engineered mouse models (GEMMs) have been used to better understand cellular processes and their effects within a living animal. GEMMs can be used to study normal physiology, such as mammary gland developed (right), as well as pathophysiology, such as the MMTV-PyMT mouse model that spontaneously develops breast cancer. In the Harris lab, we use a variety of GEMMs to understand the roles of antioxidants in disease, with a focus on cancer development and progression.