Research Projects
 Platelet Regulation of Circulating Immune Cell Responses
Platelet Regulation of Circulating Immune Cell Responses
Monocytes are circulating immune cells that differentiate into longer lived macrophages upon extravascular tissue trafficking. Environmental cues and immune stimuli have differential effects on monocyte functions that can be generally classified as inducing: i) pro-inflammatory monocytes that are phagocytic and produce cytokines that stimulate other inflammatory immune cells; ii) pro-reparative monocytes that produce cytokines that promote would healing and fibrosis. We have discovered that platelets regulate the differentiation of monocytes to either pro-inflammatory or pro-reparative phenotypes, in part through platelet derived beta-2 microglobulin (b2M). b2M is a molecular chaperone for the major histocompatibility class I (MHC I) complex, but b2M may also have less well understood immune functions, as elevated plasma b2M is a risk factor for adverse cardiovascular events. We have now found that platelet derived b2M is a mediator of monocyte pro-inflammatory differentiation. Circulating monocytes from mice lacking b2M only in platelets (Plt-b2M-/-) had a more pro-reparative monocyte phenotype, in part dependent on increased platelet derived TGFb signaling in the absence of b2M. We are now dissecting the molecular signaling pathways involved and ways to regulate b2M mediate monocyte responses. In addition, in collaboration with the lab of Dr. Jim Palis we are currently exploring mechanisms of adverse responses to platelet transfusions in neonates that are in part mediated by platelet-monocyte interactions.
ERK5 Regulation of Platelet Protein Expression and Activation in Ischemic Tissue Environments
We recently published studies demonstrating the role of ERK5 in platelet function following acute and chronic ischemic pathologies, including myocardial infarction and peripheral artery disease. We discovered that select platelet proteins have altered expression in both humans and mouse models of these vascular diseases and demonstrated that the regulation of platelet protein expression occurs at least in part at the level of the platelet itself. We are now actively seeking to define the platelet ‘translatome’ in response to ischemic vascular disease and how ERK5 regulates the activation of the platelet translational machinery in a disease context. Many of these studies are in collaboration with Dr. Roy Silverstein at Medical College of Wisconsin.
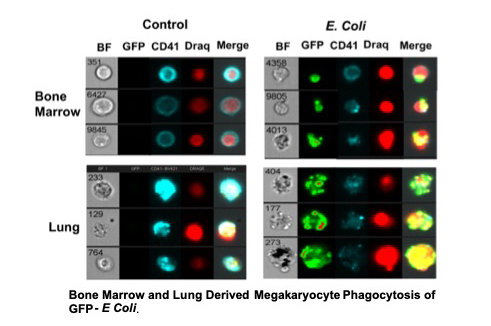
The Platelet/Megakaryocyte Regulation of Acquired Immune Responses
Megakaryocytes (Mks) are typically defined as bone marrow resident platelet progenitors. However, Mks are also present in the lung and recent work by other groups has shown that lung Mks contribute to platelet production. We have now found that compared to bone marrow Mks, lung Mks are differentiated to an immune regulatory phenotype, including the specialization of lung Mks to present antigen to T cells. Aided by unique expertise and research tools we have developed, we now seek to discover mechanisms that regulate Mk functions as professional antigen presenting cells (APCs) in the lung environment. This involves the use of Mk-specific mutant mice to determine how lung Mks participate in antigen dependent and independent T cell responses to lung infection. Outcomes of this work will impact our understanding of many immune disease processes, including immune responses to lung pathogens such as Influenza virus, bacterial pneumonias and the pathogenesis of asthma and allergy.