Gene Therapy
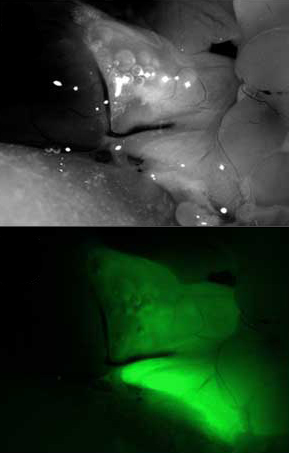
Rat lungs were electroporated with GFP-expressing
plasmids and three days later, expression was
visualized throughout the lungs. No trauma or
inflammation are associated with the technique.
Despite all of the hype regarding gene therapy, at present, gene therapy is a dream due to insufficient levels of gene transfer and expression at desired sites. One way to increase gene expression is to target more DNA to the cell nucleus. Since the nucleus is the site of transcription, without the movement of plasmids through the cytoplasm, translocation into the nucleus, and localization to the appropriate subnuclear domain, no gene expression, or gene therapy can take place.
Ongoing projects in my laboratory are studying the mechanisms of cytoplasmic trafficking of plasmids along the cytoskeleton, plasmid nuclear entry, subnuclear organization, and exploiting what we learn to improve gene therapy.
We have shown that plasmids are able to enter the nucleus in a sequence-specific manner that appears to be mediated by transcription factor binding. My lab is interested in identifying the proteins required for this activity and the regulation of their nuclear import. Based on our model, we have created cell-specific plasmids by incorporating DNA sequences that bind to cell-specific transcription factors. At present, we have examples of smooth muscle, alveolar epithelial cell, and endothelial cell-targeting vectors that we hope to use for the treatment of a number of pulmonary diseases, and we are working to expand our repertoire to selectively target expression to any desired cell or tissue.
We also are developing methods for extracellular delivery of non-viral vectors in animal models for disease. Using electroporation, we have obtained very high levels of gene expression in the vasculature and lungs of animals. We have used this approach to prevent and/or cure existing disease in models of acute lung injury and asthma in mice and rats. Our next goal is to optimize these approaches in more relevant preclinical models to move toward clinical application.

David A. Dean, Ph.D.
Principal Investigator
- Correction: Upregulation of alveolar fluid clearance is not sufficient for Na+,K+-ATPase β subunit-mediated gene therapy of LPS-induced acute lung injury in mice.; Scientific reports; Vol 16(1). 2026 May 04.
- Anticodon-edited transfer RNAs (ACE-tRNAs) encoded as therapeutic nonviral minimal DNA vectors.; Nucleic acids research; Vol 54(4). 2026 Feb 05.
- Subnuclear organization and mislocalization of plasmids reduce transgene expression.; Molecular therapy. Nucleic acids; Vol 36(4), pp. 102730. 2025 Oct 09.
- Anticodon Edited Transfer RNAs (ACE-tRNAs) Encoded as Therapeutic Nonviral Minimal DNA Vectors.; bioRxiv : the preprint server for biology. 2025 Sep 06.
News
Affiliations
- Pediatrics
- Biomedical Engineering
- Pharmacology & Physiology
- Center for Inhalation and Flavoring Toxicological Research (CIFT)
- Lung Biology and Disease Program
- Pulmonary T32 Training Grant
- Cellular and Molecular Pharmacology and Physiology Ph.D. Program
- Master’s Degree in Pharmacology
- Master’s Degree in Physiology
- PhD Program in Pathology - Cell Biology of Disease
- Toxicology Ph.D. Program
Contact Us
Dean Lab
601 Elmwood Ave
MRBX 3-11311
Rochester, NY 14642
(585) 756-7780