URMC / Labs / Benoit Lab / Projects / Tissue Engineered Periosteum Approaches to Heal Bone Allograft Transplants
Tissue Engineered Periosteum Approaches to Heal Bone Allograft Transplants
Project Collaborators:
Dr. Hani Awad, Dr. Regine Choe, Dr. Wakenda Tyler
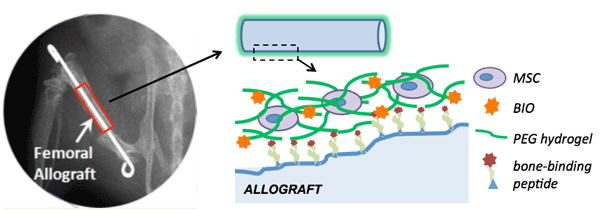
Figure1. Schematic detailing proposed tissue-engineering periosteum for improved allograft integration. Patient
specific mesenchymal stem cells (MSCs), and a soluble drug factor to increase cell proliferation (BIO) will be
encapsulated within a degradable hydrogel network used to surround the bone allograft, thus mimicking the native
periosteum. Osteogenic potential of the delivered stem cell population will be controlled by cell-matrix
mechanotransduction.
Although most orthopaedic fractures heal, the clinical management of critical (>3mm) segmental defects continues to present major challenges for both amputation and limb salvage approaches. The periosteum plays an essential role in the healing process of both fractures and autografts. Periosteal stem cells have been shown necessary for the induction of robust endochondral and intramembraneous bone formation, essential for effective healing and neovascularization of autografts. However, allografts are processed such that there is no viable stem cells or periosteum remaining. Therefore, by providing a pseudo-periosteum to revitalize allografts, a similar robust healing response may be induced.
We aim to exploit hydrogel cell/drug delivery vehicles as a tissue-engineered periosteum, evaluating different delivery regimes and hydrogel mechanical properties to promote stem cell proliferation and differentiation in vitro and in vivo within a murine segmental defect model. Our global hypothesis is that a tissue-engineered periosteum will promote allograft integration and healing thus reducing allograft failure rates.
As detailed in Fig. 1, we will encapsulate patient-specific MSCs within a hydrogel surrounding the allograft implant. Hydrogel degradation will be tuned to match the rate of host tissue healing, enabling host bone and vascular network infiltration. Facile tethering of hydrogels to the allograft will be achieved through machined molds and bone-binding peptide sequences to ensure stable localization of the hydrogel-encapsulated MSCs at the allograft surface (Fig.1). In addition, we will include signals to enhance stem cell proliferation through 6-bromoindirubin-3'-oxime (BIO) controlled dosing and temporal availability and osteogenesis through scaffold mediated mechanotransduction.
« back to all projects