Projects
Establishing a novel somato-visual biomarker for psychotic disorders
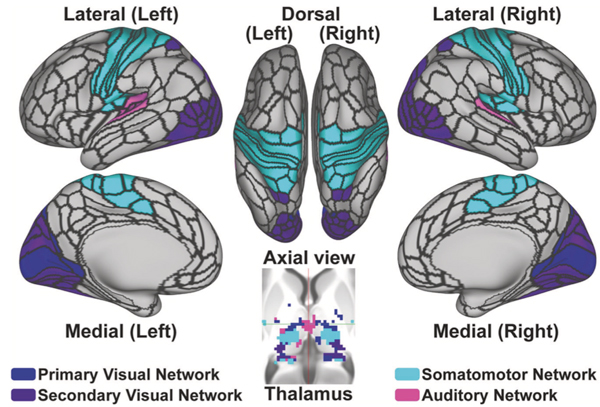 We have recently established a highly robust “somato-visual” biomarker for psychosis, which is defined by thalamo-cortical hyperconnectivity and cortico-cortical hypoconnectivity in somatomotor and higher-order visual network (Keane et al., 2025, Molecular Psychiatry; see sky blue and purple cortical regions in the accompanying figure). A current goal in our lab is to harness cutting-edge methods in acquisition and preprocessing to examine the clinical relevance, behavioral implications, and diagnostic specificity of this biomarker.
We have recently established a highly robust “somato-visual” biomarker for psychosis, which is defined by thalamo-cortical hyperconnectivity and cortico-cortical hypoconnectivity in somatomotor and higher-order visual network (Keane et al., 2025, Molecular Psychiatry; see sky blue and purple cortical regions in the accompanying figure). A current goal in our lab is to harness cutting-edge methods in acquisition and preprocessing to examine the clinical relevance, behavioral implications, and diagnostic specificity of this biomarker.
Fine-scale eye movements in schizophrenia and their contribution to abnormal vision
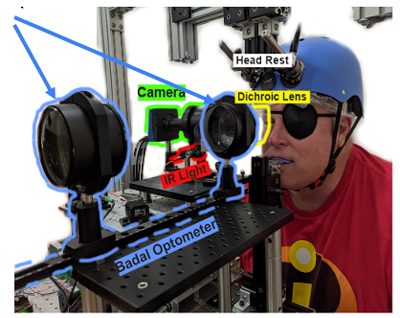 Eye movement abnormalities in schizophrenia have been studied for over a 100 years but there are shockingly few that have examined fine-scale eye movements in this population. Of those have considered this question, none did so with high spatial precision, none have ruled out ocular pathology or refractive error confounds, and none have considered their task relevance. My collaborators and I (from the “Active Perception Lab”) are employing state-of-the-art eye-tracking and screening methods to determine to what extent fine-scale eye movement abnormalities exist in schizophrenia; we are also considering how such eye movements might contribute to poor visual acuity and reading.
Eye movement abnormalities in schizophrenia have been studied for over a 100 years but there are shockingly few that have examined fine-scale eye movements in this population. Of those have considered this question, none did so with high spatial precision, none have ruled out ocular pathology or refractive error confounds, and none have considered their task relevance. My collaborators and I (from the “Active Perception Lab”) are employing state-of-the-art eye-tracking and screening methods to determine to what extent fine-scale eye movement abnormalities exist in schizophrenia; we are also considering how such eye movements might contribute to poor visual acuity and reading.