Engineering Scarless Repair of Tendon Injuries
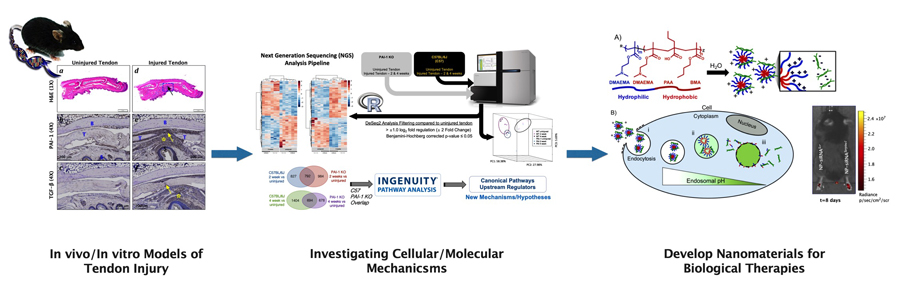
Injuries to tendons can be acute, resulting from work-, sport-, or trauma-related full or partial tissue rupture, or can be chronic resulting from repetitive accumulation of microdamage due to overuse or aging, leading to a spectrum of painful, degenerative injuries collectively known as tendinopathy. The major injuries typically involve a variety of tendons including Achilles, Patellar, Quadriceps, Hamstring, Supraspinatus (rotator cuff), and hand and wrist flexor tendons. The incomplete understanding of the complex cellular and molecular processes involved in the fibrotic healing process in tendon has hindered the development of biological therapies for regenerative tendon repair.
We are pioneering regenerative treatments to injuries to flexor tendons in zone II of the hand, which are among the most difficult injuries for surgeons to repair. Restoration of function during healing is often impaired due to the formation of debilitating adhesions and the incidence of repair rupture typically necessitates additional surgery. Our research focuses on the mechanobiology regulating tendon healing with the goal of manipulating target pathways and processes to develop clinically translatable biological therapies to improve outcomes of tendon healing. Our approach uses genetic mouse models of zone II flexor tendon injury and novel in vitro platforms to uncover the cellular and molecular mechanisms of fibrotic healing, and innovative biomaterials approaches for drug delivery to target candidate pathologic pathways. Some specific foci of research include:
- The role TGF-beta/PAI-1 in regulating fibrotic tendon healing and downstream effectors of fibrosis and adhesions.
- Nanomaterials approaches to deliver biological therapies to inhibit PAI-1 and downstream targets.
- Immunophenotyping and transcriptomics of fibrotic tendon adhesions in mice and humans.
- Novel in vitro microphysiological systems of inflammation and fibrosis in acute and chronic tendon pathologies.
Collaborators: Danielle Benoit, Alayna Loiselle, James McGrath, Constantinos Ketonis
Funding: NIH/NIAMS R01 AR056696