Molecular and Signaling Mechanisms of Synaptic Plasticity in Memory Formation and Mental Health
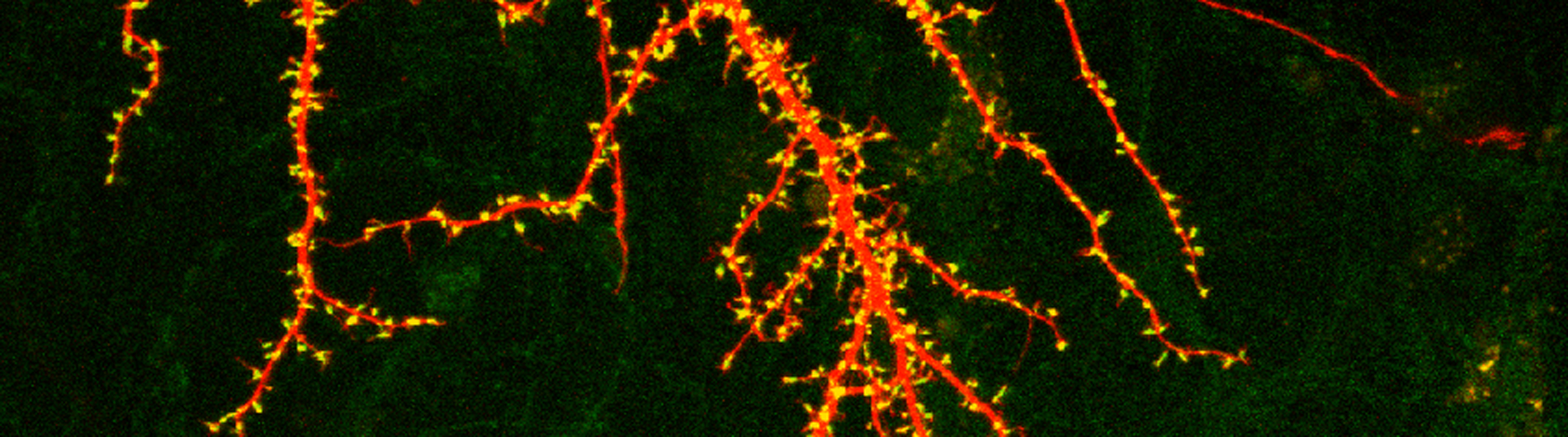
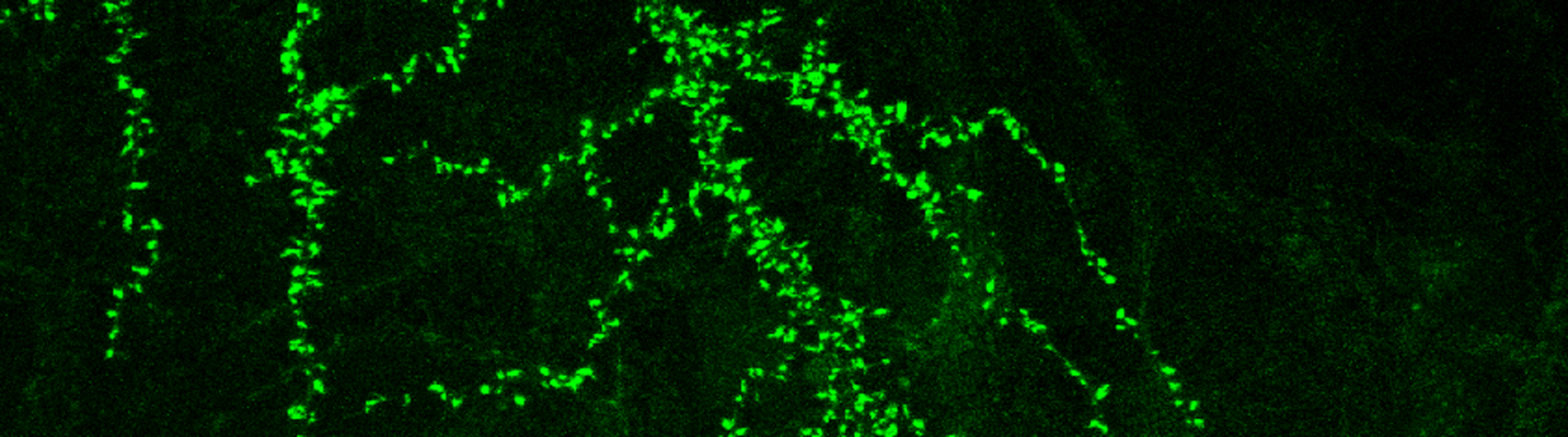
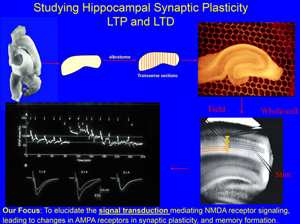 Synapse has been shown to undergo persistent modifications in response to different patterns of activity which is termed synaptic plasticity, with long term potentiation (LTP) and long term depression (LTD) being its major forms. Synaptic plasticity has been hypothesized to underlie the experience-dependent brain functions, such as learning, memory and anxiety. We are interested in how NMDA receptors function in inducing these many forms of synaptic plasticity. We are not only interested in short term modifications in the synaptic protein composition through calcium mediated signaling pathway, but also CREB mediated gene transcription which provides new proteins for long term modification of the synapse.
Synapse has been shown to undergo persistent modifications in response to different patterns of activity which is termed synaptic plasticity, with long term potentiation (LTP) and long term depression (LTD) being its major forms. Synaptic plasticity has been hypothesized to underlie the experience-dependent brain functions, such as learning, memory and anxiety. We are interested in how NMDA receptors function in inducing these many forms of synaptic plasticity. We are not only interested in short term modifications in the synaptic protein composition through calcium mediated signaling pathway, but also CREB mediated gene transcription which provides new proteins for long term modification of the synapse.

Houhui Xia, M.Sc., Ph.D.
Principal Investigator
Publications
View All Publications- PP1β opposes classic PP1 function, inhibiting spine maturation and promoting LTP.; bioRxiv : the preprint server for biology. 2023 Jan 27.
- Regulation of PP1 interaction with I-2, neurabin, and F-actin.; Molecular and cellular neurosciences. 2022 Jan 25.
- Nuclear Inhibitor of Protein Phosphatase 1 (NIPP1) Regulates CNS Tau Phosphorylation and Myelination During Development.; Molecular neurobiology. 2022 Jan 06.
- Protein phosphatase-1 inhibitor-2 promotes PP1γ positive regulation of synaptic transmission.; Frontiers in synaptic neuroscience; Vol 14, pp. 1021832. 2022 Jan 06.
News
Affiliations
- Pharmacology & Physiology
- Neuroscience
- Del Monte Institute for Neuroscience
- Cellular and Molecular Pharmacology and Physiology Ph.D. Program
- Master’s Degree in Pharmacology
- Master’s Degree in Physiology
- Medical Pharmacology Master's Degree
- Neurobiology & Anatomy Ph.D. Program
- Neuroscience Ph.D. Program
- Toxicology Ph.D. Program
November 19, 2020
Karl Foley, Receives NIH F30 Award
July 7, 2017
Houhui Xia appointed Paul Stark Professor in Pharmacology
Contact Us
Xia Lab
601 Elmwood Ave
Rochester, NY 14642