About Us
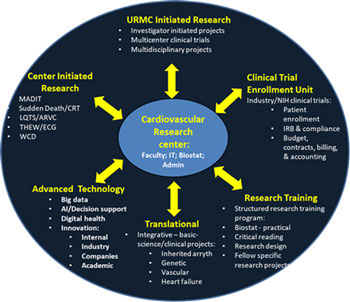
For more than four decades, we have been involved in the design, conduct, and analysis of small and large scale, national and international clinical research studies. These studies ranged from small or large observational studies to Phase II-IV trials testing specific drug or device interventions, funded by the NIH and industry partners, many of them linked to subsequent FDA approval. Our studies use the latest basic science, bioengineering, genetic, biostatistical, and clinical approaches to optimize the acquisition of new knowledge in order to improve the diagnostic and therapeutic foundation of clinical medicine.
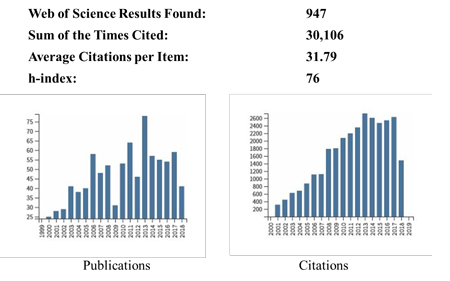
The CCRC has an outstanding reputation for scientific excellence, we have published almost a thousand scientific, peer-reviewed articles in the past 20 years that were cited 30,106 times (average citation 31.79) with an H-index of 76.
Our Clinical Trials Coordination Center
As an academic-based, full-facility Clinical Trials Coordination Center with access to a vast array of University based services, CCRC can provide the highest professional standards to meet all aspects of the diverse management requirements of clinical cardiovascular research studies.
Our Mission
Committed to advancing cardiovascular science by conducting and/or providing services for multicenter randomized or observational clinical research studies, and initiating novel innovative strategies through the highest quality of clinical research.
Our Values
Respect: We honor autonomy, competence, and relatedness.
Integrity: We uphold the highest values in our research conduct.
Professionalism: We focus on professional results and timeliness.
Courage: We tackle important scientific question to improve cardiac health.
Commitment: We are persistent and loyal to our research endeavors.