URMC Selected for NIH Initiative to Map Connections in the Brain
Tuesday, September 26, 2023
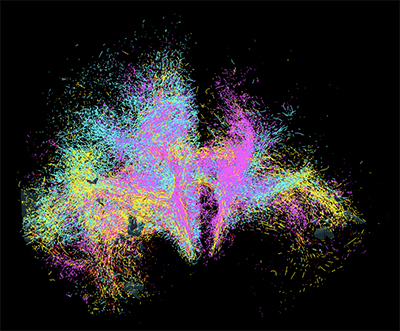 Goal is to Develop Cutting-Edge Tools to Image Neural Networks in Unprecedented Detail
Goal is to Develop Cutting-Edge Tools to Image Neural Networks in Unprecedented Detail
Scientists at the University of Rochester Medical Center are joining research teams across the globe to develop next-generation tools for visualizing connections in the human brain. Imaging and understanding the brain’s intricate circuitry at the cellular and microscopic level will advance new approaches to treat brain disorders like Tourette’s syndrome, Parkinson’s disease, and obsessive-compulsive disorder.
The research is supported by the National Institutes of Health’s Brain Research Through Advancing Innovative Neurotechnologies® Initiative, or The BRAIN Initiative®. Principal investigator Suzanne Haber, PhD, professor of Pharmacology and Physiology at URMC, will lead a team of scientists with co-principal investigators Anastasia Yendiki of Massachusetts General Hospital and Elizabeth Hillman of Columbia University.
Haber’s lab will launch the team’s efforts by providing the “ground truth”—animal tracing data that is considered the gold standard for understanding the complex connectivity of the human brain.
“We can’t develop tools without a guide to start with, just like you can’t navigate the ocean without a map,” noted Haber, who is also a professor of Neuroscience, Brain and Cognitive Science, and Psychiatry. “Once we identify how axons traverse the brain in animal models, we use that information to develop tools to accurately track connections in the human brain. These novel technologies for imaging axonal projections will increase our understanding of the circuitry and cellular mechanisms that underlie brain abnormalities.”
Today, diffusion MRI (dMRI) is used to examine the connectivity and functional networks in the human brain. However, this is an indirect method with inherent problems, particularly in specific situations that make it difficult to accurately follow the axons. This leads to many false positives and false negatives.
 Haber says the time is right for this initiative. “We’ve seen so many exciting developments in optics and engineering over the past few years…the field has really exploded,” she notes. “We’re excited to be part of the team that’s pushing the boundaries to better understand the circuitry in humans. Ultimately, we hope this work sets the stage for the creation of more accurate diagnostics and therapeutics for brain diseases.”
Haber says the time is right for this initiative. “We’ve seen so many exciting developments in optics and engineering over the past few years…the field has really exploded,” she notes. “We’re excited to be part of the team that’s pushing the boundaries to better understand the circuitry in humans. Ultimately, we hope this work sets the stage for the creation of more accurate diagnostics and therapeutics for brain diseases.”
Haber’s work, which will be funded by $1.5 million from NIH over five years, is part of the center for Large-scale Imaging of Neural Circuits (LINC). In addition to the University of Rochester, Massachusetts General Hospital and Columbia University, the LINC project includes collaborators from Brigham and Women’s Hospital, Harvard University, Massachusetts Institute of Technology, University College London, and Weill Cornell Medicine.
The BRAIN Initiative is a partnership between federal and non-federal partners, with the NIH taking the leading role in achieving the goal of revolutionizing our understanding of the human brain by accelerating the development of innovative neurotechnologies. The program is made up and managed by 10 NIH institutes and centers whose missions and current research portfolios complement the goals of the BRAIN Initiative. The initial round of awards is projected to total $150 million over 5 years, with a large portion of the funds dedicated to tool development.
Please Welcome our Faculty member, Manoela V. Fogaca, PhD - February 1, 2023 to the Department of Pharmacology and Physiology
Monday, January 9, 2023
 Dr. Fogaça’s research focuses on understanding the molecular basis of behaviors relevant to stress and the actions of antidepressant and anxiolytic drugs, aiming at identifying specific circuits, neuronal subpopulations and synaptic mechanisms involved in these responses, as well as novel pharmacological strategies to treat neuropsychiatric disorders. Dr. Fogaça received her Master’s and PhD in Pharmacology from the Department of Pharmacology at the University of São Paulo (Brazil), where she studied molecular mechanisms of action of cannabinoid compounds in multiple brain systems involved in stress-related disorders. As a postdoctoral fellow at Yale University, Dr. Fogaça has investigated cellular and synaptic mechanisms underlying the rapid and sustained effects of fast-acting antidepressants, including ketamine and ketamine-like drugs, with the goal of contributing to the development of more effective medications to treat major depression disorder (MDD).
Dr. Fogaça’s research focuses on understanding the molecular basis of behaviors relevant to stress and the actions of antidepressant and anxiolytic drugs, aiming at identifying specific circuits, neuronal subpopulations and synaptic mechanisms involved in these responses, as well as novel pharmacological strategies to treat neuropsychiatric disorders. Dr. Fogaça received her Master’s and PhD in Pharmacology from the Department of Pharmacology at the University of São Paulo (Brazil), where she studied molecular mechanisms of action of cannabinoid compounds in multiple brain systems involved in stress-related disorders. As a postdoctoral fellow at Yale University, Dr. Fogaça has investigated cellular and synaptic mechanisms underlying the rapid and sustained effects of fast-acting antidepressants, including ketamine and ketamine-like drugs, with the goal of contributing to the development of more effective medications to treat major depression disorder (MDD).
Because currently available antidepressants have serious limitations for treating MDD, including low response rates, a significant number of treatment resistant patients, and a time-lag before there is a therapeutic response, exploring the mechanisms of action of rapid antidepressants is an important strategy to understand the pathophysiology of MDD and to guide efforts to develop safer and better-tolerated drugs. Low doses of ketamine, an NMDA receptor (NMDA-R) blocker, can induce rapid (2 h) and sustained (up to 7 days) antidepressant effects in chronically stressed mice and in patients diagnosed with MDD, even in patients that are refractory to current antidepressants. Early findings suggest that these drugs initially target specific subpopulations of GABA interneurons in the medial prefrontal cortex (mPFC) and promote a fast enhancement of neurotrophic factors release, such as the brain-derived neurotrophic factor (BDNF), as well as glutamate- and GABA-induced neuroplasticity, leading to protein synthesis, synaptogenesis and new spine formation. One hypothesis is that this local re-organization re-establishes the excitation:inhibition balance (E:I) in the mPFC, leading to a restoration of correct firing patterns and the integrity of signal transfer to target regions, and thereby promoting antidepressant effects. At the University of Rochester, research at Fogaça’s lab will expand these studies to understand how complex local and long-range neural circuits interact and modulate brain plasticity that culminate in distinct behavioral outcomes. Her lab will combine molecular neuropharmacology, genetic approaches and circuit-level studies of neurobiological systems to investigate how specific subpopulations of GABAergic and glutamatergic neurons crosstalk to modulate the cortical E:I network dynamics that lead to phenotypes relevant to stress disorders. On the long-term, Dr. Fogaça’s research will span the mPFC to additional brain circuits, with the objective of mapping downstream regions and neuromodulatory systems to determine how the interaction between the mPFC and its projections promotes stress resilience and rapid antidepressant responses.
Congratulations Chen Li!
Sunday, January 1, 2023
Chen Li, 4th year graduate student in the laboratory of Dr. Craig Morrell was awarded a two-year American Heart Association Predoctoral Fellowship entitled, “Thrombocytopenia Independently Leads to Monocyte Immune Dysfunction in Sepsis”.
Project Summary
In addition to their well-studied hemostatic functions, platelets are immune cells. We have now found that thrombocytopenia leads to monocyte dysfunction, independent of the cause of thrombocytopenia, in a manner that is dependent on direct platelet-monocyte CD47 interactions that regulate monocyte immunometabolism. Using mouse models of thrombocytopenia and sepsis we found that normal platelet numbers are needed to limit immune dysregulation in sepsis. Our studies demonstrate that in healthy conditions resting platelets maintain monocyte immune tolerance by regulating monocyte metabolic processes that lead to changes in gene expression regulation.