Implant Infections in Real Time: New Visualization Could Shape Use of Vaccine, Antibiotics, Bacteria-Resistant Implants
 Edward Schwarz, PhD.
Edward Schwarz, PhD. Chao Xie, MD, MSBA
Chao Xie, MD, MSBAPost-op infection of an artificial joint is a dreaded outcome – and it’s a risk with every new implant and revision surgery. That battle between invasive bacteria and the patient’s immune cells over a newly implanted device has been known as “the race for the surface” since 1987, when pivotal research published in Science explored the phenomenon.
Now, for the first time, scientists in Dr. Chao Xie’s lab at the Center for Musculoskeletal Research at the University of Rochester Medical Center have devised a way to observe the race for the surface while it’s happening – and watch methicillin-resistant Staphylococcus aureus (MRSA) cells right for supremacy in a live mouse model. What they learned could revolutionize the way orthopaedic surgeons
use vaccines, antibiotics and bacteria-resistant implants to prevent this potentially deadly surgical complication.
Researchers shared their discovery at the Orthopaedic Research Society meeting in February. Professor of Orthopaedics and Center for Musculoskeletal Research director Edward Schwarz, PhD., discusses the team’s findings.
Why did you want to visualize the race for the surface?
In Orthopaedics, we’ve accepted the premise of the race for the surface since the 1987 Science paper (“Race for the Surface: Microbial Adhesion vs. Tissue Integration of Biomaterial-Centered Infection.”) But, to fully believe it and understand it, you have to see it. For the first time, we have seen the race for the surface: what it looks like, how rapidly it can occur, how completely bacteria can overwhelm the surgery area. We have videos of MRSA infection inside a living mouse’s femur, with those bacteria fighting off the white blood cells and then capturing the surface of the infected implant. It’s stunning. No one has ever seen this before.
How quickly does bacteria overwhelm the surgical area?
The speed at which this race occurs surprised us. MRSA doubles every 30 minutes, but white blood cells can only double once a day. At the 1-hour mark, you see mostly healthy mouse cells and a few bacteria, but at the 3- hour mark, you see a 50-50 split between healthy cells and bacteria, and from that point on, the bacteria count explodes. Thus, the race for the surface of the implant is over within 3 hours of the surgery – that’s how fast it happens.
How can this work advance research into implant infection and help improve outcomes for patients?
There are many approaches to reducing bone infection, including antibiotics, antimicrobial implants, and vaccines. None of these have ever been demonstrated to affect the race for the surface because nobody has been able to see it. With this technology, we can test – and view – how effectively each approach fights infection. We believe this is the way products will be evaluated in the future.
What should orthopaedic surgeons know about this research?
We’ve seen in an animal model that without intervention, the race for the surface is over in 3 hours. We believe our future research will be a powerful tool in evaluating the effectiveness of new devices and treatments so that those with merit earn FDA approval and help orthopaedic surgeons around the world deliver safer, better outcomes for their patients.

Drs. Edward Schwarz and Chao Xie’s Lab members (from left) 1st Row: Gowrishankar Muthukrishnan (PhD), Chao Xie (MD, MSBA), Kayana Chen (MS), Yue Peng (MS, PhD candidate), Edward M. Schwarz (PhD) 2nd Row: Kyra Sandercock (Class 2023),Youliang Ren (MD, PhD) 3rd Row: Mark Kenney (PhD, MD candidate), Pei Liu (MD, PhD candidate), Moto Saito (MD, PhD) 4th Row: Jason Weeks (BS), Joshua Rainbolt (Class 2023), Himanshu Meghwani (PhD)
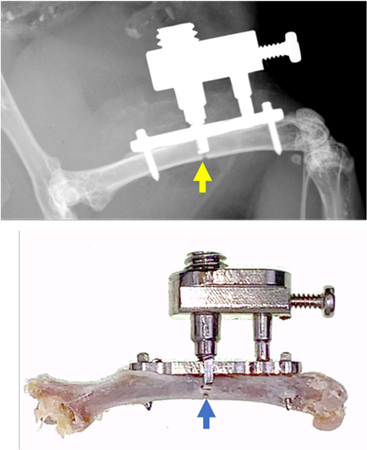
How did you make it possible to see this?
We used intravital multiphoton laser scanning microscopy, a technology available at virtually any medical center. Research using a microscope in a living mouse to capture images of things happening in real time is a very exciting field right now; many researchers are taking advantage of this technology. What didn’t exist is, how to put that microscope into the mouse femur so that the microscope lens is perfectly aligned with the area of study on the implant within the bone, so you can visually capture what is happening in a living animal at the cellular level.
To do this, our team built a metal jig that’s small enough to insert in the femur of a mouse model and stable enough to hold the microscope lens at a precise 90-degree angle for the duration of the study. We also used a pin infected with MRSA to penetrate the bone so we could measure the infection inside the bone marrow. The microscope captures a series of images of the study area – similar to the way a CT gets “slices” of an area in the human body and reconstructs them into a 3D image. We ended up with an animated video of the surgical area as the infection progresses.
What did you see?
We have 2-hour, 4-hour and 6-hour studies that are time-lapse-compressed into 12-minute videos. In each, we can see healthy white blood cells and the MRSA very clearly. As the infection takes hold, we see the white blood cells attacking and absorbing some of the bacteria, the bacteria reproducing, and the eventual outcome in an animal that has no antibiotic protection: MRSA overtakes the surface of the implant and becomes an uncontrolled infection.
What’s next?
We’re enthusiastic about this first result and we want to validate our findings with further research. In addition to repeating this test with additional mouse models, we’re also interested in quantifying the movement of immune cells and bacteria. We’d like to compare their presence in an infected vs. a sterile implant, test the effect of different antibiotics at various dosages and timing of use, etc.