News
20242023202220212020
Subscribe to Neuroscience LISTSERV
Changes in the brain hinder addiction recovery in people who are HIV-positive
Monday, December 20, 2021
Researchers with the Del Monte Institute for Neuroscience at the University of Rochester are studying how the brain puts the 'brakes' on behavior. That may be different in individuals recovering from cocaine addiction and who are also HIV-positive.
“Scientists have long known that drug abuse can cause damage to the brain. We also know HIV infection can cause brain changes,” said John Foxe, Ph.D., director of the Del Monte Institute for Neurosciences and senior author of the study published in Neuropharmacology. “Since drug use is common in individuals with HIV, an important question is how brain deficits associated with both conditions might add up.”
Researchers used functional magnetic resonance imaging (fMRI) to measure the brain responses of cocaine addicts and patients with HIV while participants played a game that involved purposefully withholding responses to target stimuli. “The challenging thing about the game we asked participants to play isn’t exactly playing it, per se,” said Kathryn Mary Wakim, Ph.D., a recent graduate from the Neuroscience Graduate Program at the University of Rochester and the first author of the study. “What’s really hard is not playing the game. What we wanted to measure was how the brain holds back a response under certain task conditions.”
Difficulty withholding inappropriate responses are thought to be a central issue in addiction. Researchers found that participants diagnosed with both HIV and cocaine dependence had a difficult time holding back behavioral responses while playing the game, changes which were also reflected in brain activity. A companion study also published in Neuropharmacology found similar results while brain activity was measured using electroencephalography (EEG). Brain activity during response withholding in HIV+ participants in recovery from cocaine dependence was different than brain activity in HIV- participants in recovery.
“Currently, when someone who is HIV positive enters a recovery program they are treated in much the same fashion as any other person with an addiction issue,” said Foxe. “But our findings show that they, very likely, need to be treated differently or more intensively. HIV and drug addiction should be a dual diagnosis when it comes to recovery, and we will need to design specific targeted intervention approaches for this population, given their unique vulnerability.”
“When people with HIV relapse, it's a big deal. The majority of participants in our study contracted HIV – not by injecting cocaine – but by having unprotected sex,” Wakim said. “Relapse is an outcome that should be minimized in this population because cocaine use is strongly associated with risky sexual behavior, which makes the spread of HIV more likely when a relapse occurs.”
Additional co-authors are Edward Freedman, Ciara Molloy, Madalina Tivarus, Nicole Vieyto, and Zhewei Cao with the University of Rochester, and Armin Heinecke at Maastricht University in the Netherlands. The research was supported with funding from Eunice Kennedy Shriver National Institute of Child Health and Human Development and by the University of Rochester Center for AIDS Research, which is funded through the National Institute of Allergy and Infectious Diseases.
URMC researchers reveal how the brain understands one voice in a noisy crowd
Wednesday, December 15, 2021
Previous work is honored for its impact on improving hearing devices
In a crowded room where many people are talking, such as a family birthday party or busy restaurant, our brains have the ability to focus our attention on a single speaker. Understanding this scenario and how the brain processes stimuli like speech, language, and music has been the research focus of Edmund Lalor, Ph.D., associate professor of Neuroscience and Biomedical Engineering at the University of Rochester Medical Center.
Recently, his lab found a new clue into how the brain is able to unpack this information and intentionally hear one speaker, while weaning out or ignoring a different speaker. The brain is actually taking an extra step to understand the words coming from the speaker being listened to, and not taking that step with the other words swirling around the conversation. “Our findings suggest that the acoustics of both the attended story and the unattended or ignored story are processed similarly,” said Lalor. “But we found there was a clear distinction between what happened next in the brain.”
For this study, recently published in The Journal of Neuroscience, participants simultaneously listened to two stories, but were asked to focus their attention on only one. Using EEG brainwave recordings, the researchers found the story that participants were instructed to pay attention to was converted into linguistic units known as phonemes – these are units of sound that can distinguish one word from another – while the other story was not. “That conversion is the first step towards understanding the attended story,” Lalor said. “Sounds need to be recognized as corresponding to specific linguistic categories like phonemes and syllables, so that we can ultimately determine what words are being spoken – even if they sound different -- for example, spoken by people with different accents or different voice pitches.” Co-authors on this paper include Rochester graduate student Farhin Ahmed, and Emily Teoh of Trinity College, University of Dublin. The research was funded by the Science Foundation of Ireland, Irish Research Council Government of Ireland, Del Monte Institute for Neuroscience Pilot Program, and the National Institute on Deafness and Other Communication Disorders.
Read More: URMC researchers reveal how the brain understands one voice in a noisy crowdURMC & RIT faculty awarded patent for gene transfer technology that could transform cancer therapies
Monday, December 13, 2021
The carbon nanotube device could streamline some cancer therapies like CAR T-cell therapy.
Researchers at the University of Rochester Del Monte Institute for Neuroscience and Rochester Institute of Technology have received a U.S. patent for technology designed to accelerate development of cell therapies for cancer and other bio-therapies. The technique provides a less toxic alternative to standard gene transfer techniques by using an array of carbon nanotubes to deliver DNA into primary neurons, immune cells, and stem cells.
“Our goal is to provide a technology that can lower the cost and increase speed and the range of cell types that can be adapted for therapeutic use,” said Ian Dickerson, Ph.D., associate professor of Neuroscience. “Many new cell-based therapies depend on changing the gene expression of primary cells. These approaches range from stem cells for production of patient-specific repair tissues, to CAR T-cells used for focused cancer therapy.”
Dickerson and Michael Schrlau, Ph.D., associate professor of mechanical engineering in RIT’s Kate Gleason College of Engineering, were recently awarded a patent for this technology. It delivers biomolecules into cells through carbon nanotube arrays. Their “honeycomb” of nanotubes device was first described in a 2016 study published in the journal Small.
Read More: URMC & RIT faculty awarded patent for gene transfer technology that could transform cancer therapiesEven in a safe environment, people with anxiety may feel threatened
Wednesday, November 24, 2021
“These findings tell us that anxiety disorders might be more than a lack of awareness of the environment or ignorance of safety, but rather that individuals suffering from an anxiety disorder cannot control their feelings and behavior even if they wanted to,” says Benjamin Suarez-Jimenez, an assistant professor of neuroscience. “The patients with an anxiety disorder could rationally say—I’m in a safe space—but we found their brain was behaving as if it was not.”
Read More: Even in a safe environment, people with anxiety may feel threatenedAnxiety cues found in the brain despite safe environment
Friday, November 12, 2021
Imagine you are in a meadow picking flowers. You know that some flowers are safe, while others have a bee inside that will sting you. How would you react to this environment and, more importantly, how would your brain react? This is the scene in a virtual-reality environment used by researchers to understand the impact anxiety has on the brain and how brain regions interact with one another to shape behavior.
“These findings tell us that anxiety disorders might be more than a lack of awareness of the environment or ignorance of safety, but rather that individuals suffering from an anxiety disorder cannot control their feelings and behavior even if they wanted to,” said Benjamin Suarez-Jimenez, Ph.D., assistant professor in the Del Monte Institute for Neuroscience at the University of Rochester and first author of the study published in Communications Biology. “The patients with an anxiety disorder could rationally say – I’m in a safe space – but we found their brain was behaving as if it was not.”
Read More: Anxiety cues found in the brain despite safe environmentMultiple sclerosis drug improves memory in mice modeling Alzheimer’s disease
Friday, November 5, 2021
Losing memory is a hallmark of Alzheimer’s, a symptom of the disease that depletes a patient’s quality of life. Improving memory and slowing cognitive changes caused by the disease is an ongoing challenge for researchers seeking to develop novel therapies. In a newly published paper in Frontiers in Neuroscience, researchers at the Del Monte Institute for Neuroscience at the University of Rochester found that glatiramer acetate, a prescription drug currently used to treat patients with multiple sclerosis (MS), improved memory in a mouse model of Alzheimer’s disease.
“This research extends our information about glatiramer acetate’s potential use in Alzheimer’s disease,” said M. Kerry O’Banion, M.D., Ph.D., professor of Neuroscience and senior author of the study. “This isn’t a cure, but it could be a step in the right direction for a treatment to slow the symptoms of this debilitating disease.”
Using a mouse model, researchers found changes in microglia – part of the brain’s immune system – and improvements in cognitive behavior when glatiramer acetate was used. These changes were associated with less amyloid plaques and modifications to tau pathology – a protein found in neurodegenerative diseases – in the brain, indicating that molecular hallmarks of Alzheimer’s disease had been impacted. Previous studies have found that glatiramer acetate can alter brain pathology in Alzheimer’s disease mouse models, but the exact mechanisms that are impacted in the brain are still unknown.
“Overall, these findings provide further evidence that therapies that modify the immune system could be effective in the treatment of Alzheimer’s disease,” said Dawling Dionisio-Santos, Ph.D., a first-year resident in Neurology and graduate of the Medical Scientist Training Program and co-first author on the paper. “It adds evidence to support trials that test the use of glatiramer acetate in patients at risk for developing Alzheimer’s.”
Co-authors on this paper include Berke Karaahmet, Elizabeth K. Belcher, Ph.D., Laura D. Owlett, Ph.D., Lee A. Trojanczyk, and John A. Olschowka, Ph.D. The research was funded by the National Institute on Aging.
Read More: Multiple sclerosis drug improves memory in mice modeling Alzheimer’s diseaseURMC to Lead First Gene Therapy Study for Batten Disease
Wednesday, November 3, 2021
The University of Rochester Medical Center (URMC) will serve as the lead study site in the U.S. for an experimental treatment being developed by Neurogene for CLN5 Batten disease, a rare and fatal neurodegenerative disorder.
“CLN5 is a devastating and rapidly progressive neurodegenerative disease in children that leads to vision loss, cognitive and motor impairment, seizures and, ultimately, premature death,” said Jonathan W. Mink, M.D., Ph.D., the Frederick A. Horner MD Distinguished Professor in Pediatric Neurology, Chief of Child Neurology at URMC. “This trial will move research forward in developing a potentially disease-modifying treatment for CLN5 disease, providing hope to individuals and families where currently none exists.”
Batten disease is a group of rare, inherited neurodegenerative diseases also called neuronal ceroid lipofuscinoses (NCLs). The CLN5 subtype is caused by a variants in the CLN5 gene, which leads to disruption of normal protein function. The disease will often first appear and be diagnosed in childhood. There are currently no approved treatments that can reverse the symptoms of this disease.
The Medical Center is home to the University of Rochester Batten Center (URBC), which is led by Mink and is one of the nation’s premier centers dedicated to the study and treatment of this condition.
This clinical trial is possible because of the work started back in 2001 by Mink, URMC neurologist Frederick Marshall, and others to understand the natural history of these complex diseases. Working with patients from across the U.S., the team created the Unified Batten Disease Rating Scale, which tracks and quantifies how the different forms of disease progress over time. This tool is employed by researchers across the world to evaluate patients and develop and test new potential therapies.
The planned open-label phase 1/2 clinical trial will evaluate a single intraventricular dose delivered directly into cerebrospinal fluid in the brain. The treatment, called NGN-101, uses an adeno-associated virus to deliver a healthy version of CLN5 gene directly to the central nervous system. Animal studies have shown that the treatment has the potential to halt the key features of disease progression, including vision, motor, cognitive, and behavioral declines.
“At this point for CLN5 disease, what we can offer patients is limited to managing some of their symptoms, and even standard treatment for symptoms like seizures are not consistently effective,” said Mink. “This new study also has the potential to inform development of gene therapies for other forms of the disease. It is a moment that the research community and patients and families have been waiting a long time to come.”
Neurogene’s NGN-101 was recently cleared by the Food and Drug Administration to begin clinical trials and has received Orphan Drug Designation by the U.S. and European regulatory agencies. URMC anticipates enrolling its first study participants in early 2022. Due to the rare nature of CNL5, Mink anticipates that patients from across the U.S. will travel to Rochester to participate in the study. Other URMC researchers involved in the clinical trial include Amy Vierhile, D.N.P. Jennifer Vermilion, M.D., Heather Adams, Ph.D., and Erika Augustine, M.D.
Research continues to rule out brain’s immune system as key to fetal alcohol spectrum disorders
Tuesday, November 2, 2021
Researchers at the Del Monte Institute for Neuroscience at the University of Rochester continue to find evidence that the brain’s immune system may not be the culprit behind fetal alcohol spectrum disorders (FASD). It has long been thought that cells in the brain’s immune system called microglia are responsible for the neurological damage that occurs when this type of exposure happens.
“We looked for more subtle changes in microglia function this time,” said Ania Majewska, Ph.D., professor of neuroscience at the Del Monte Institute for Neuroscience, and lead author on the study published in Developmental Neurobiology. “We were not able to detect a difference in function leading us to believe that the defect may exist in other cell types. However, given the heterogeneity and complexity of microglial function, we are also exploring how different populations of microglia in different parts of the brain respond to alcohol.”
One out of every 100 babies born in the U.S. is diagnosed with FASD, which occurs when a child is exposed to alcohol in the womb. FASD can cause lifelong physical and cognitive impairments, such as poor impulse control and attention, learning disabilities, compromised fine motor skills, and delays in the ability of the brain to process visual and auditory information. There is no available treatment for FASD.
Majewska’s research initially poked holes in the idea that FASD is driven by damage caused in the brain by impaired microglia when her lab found no difference in brain activity in the area among mice exposed to alcohol early in development when compared to those who were not exposed.
In this recent work, researchers exposed mice to alcohol early in development and looked beyond the baseline of microglial movement. Using advanced imaging technology called two-photon microscopy the scientists observed how the microglia interacted with neuronal dendrites – part of the connection between neurons that enables neighboring nerves cells to communicate. They found no difference in this connection when comparing brains exposed to alcohol to brains that were not.
“The brain is made up of many neuronal types so it is possible other microglia-neuron interactions are changing, but it is not a general phenomenon,” said Majewska. “But just because these are immune cells doesn’t mean that they will be involved in every disorder or developmental perturbation. Narrowing down their exact roles is important in terms of targeting these cells for therapies for neurodevelopmental disorders.”
Additional co-authors of the study include Elissa Wong, Alexandra Strohm, Jason Atlas, and Cassandra Lamantia with the University of Rochester Medical Center. The research was funded by National Institute on Alcohol Abuse and Alcoholism.
Read More: Research continues to rule out brain’s immune system as key to fetal alcohol spectrum disordersURMC Convocation 2021: Congratulations to NGP Students Receiving Awards
Monday, September 20, 2021
Congratulations to all the Neuroscience Graduate Students who were presented awards at the 2021 URMC Opening Convocation.
- Alexis Feidler: Graduate Alumni Fellowship in recognition of an incoming first-year student with promise for exceptional accomplishment in graduate study.
- Lia Calcines Rodriguez: Irving L. Spar Fellowship in recognition of an incoming first-year student with promise for exceptional accomplishment in graduate study.
- Catalina Guzman: Merritt & Marjorie Cleveland Fellowship supporting a deserving first-year student with interest in developing a neuroscience-related research career.
- Julia Granato: J. Newell Stannard Graduate Student Scholarship recognizing a deserving incoming graduate student for their commendable academic achievements.
- Keshov Sharma: Outstanding Student Mentor Award recognizing a graduate student appointed in, funded by, or primarily housed in a research area in SMD for outstanding mentoring of undergraduate or graduate students by guiding, supporting and promoting the training and career development of others.
- Katherine Andersh: Graduate Student Award for Excellence in Equity and Inclusion for outstanding efforts in advancing equity and inclusion.
- Rianne Stowell, PhD: Outstanding Postdoctoral Research Award in recognition for outstanding research contributions by originality, creativity, and significance of research accomplishments. Rianne is now working in Kuan Wang's lab.
Give them all a hardy congratulations when you see them.
Anne Luebke co-authors: Intermittent broadband sound may be key to slowing hearing loss
Friday, August 27, 2021
A combination of sound and silence may be a key in helping slow the progression of permanent hearing loss. Medical Center researchers found that intermittent broadband sound played over an extended time preserved sensory cells in the ear, while also rewiring some of the central auditory system in the brain, helping preserve the ability to sense the timing of sounds.
“By showing that the detection of sound is being preserved in the brain, we’re saying it’s not just the hair cells in the ear, there’s some connectivity in the central auditory system in the brain that is vital to hearing too,” says Adam Dziorny, assistant professor of pediatrics and biomedical engineering, and first author of the study which appears in the journal eNeuro. “This isn’t a cure, but it could be a step that may lead to a treatment in the future.”
For 12-hour periods, researchers played a mix of broadband sound with gaps of silence for mice with sensorineural hearing loss – the most common type of hearing loss in humans caused by damage to hair cells or the nerve that connects the ear to the brain. Previous studies have shown the presence of broadband sound preserves structure and function of sensory cells in the ear. In this study, Dziorny explains, the gaps of silence allowed researchers to presume that rewiring was occurring in the brain itself within the central auditory system. These gaps are what preserved the mouse’s ability to sense the timing of sound.
“Past research has shown that the brain is capable of modifying its signaling to both the frequency and loudness of sounds. This study shows that the brain can also be trained to modify temporal aspects as well,” says Anne Luebke, associate professor of neuroscience and biomedical engineering and co-author of this study. “This is important for speech comprehension since not only do you need to hear the speech sounds, you also need to be able to hear them in normal timing. We know a speech recording played too fast or too slow is incomprehensible.”
Researchers see these findings as an important step toward demonstrating that children with sensorineural hearing loss have the potential to preserve function using an augmented acoustic stimulus–as long as the stimulus changes the way neurons encode temporal elements of sound, and preserves outer hair cells. “This will not preserve long term hearing. The underlying biological process will still exist so there will be a drop off in hearing over time,” Dziorny says. “But preserving hearing for even a period of time will allow someone to develop speech and language processing.”
Read More: Anne Luebke co-authors: Intermittent broadband sound may be key to slowing hearing lossAppetite for survival: Brain signal alerts roundworms to changing food supply
Wednesday, August 25, 2021
Microscopic roundworms may hold the key to understanding what is happening in the brain when the instinct of an animal changes in order to survive. In a newly published paper in the journal Current Biology University of Rochester Medical Center researchers found that a signaling system in the brain changes to redirect the behavior of an animal when their survival is at risk because there is not enough food.
The experiments were conducted in C. elegans – a microscopic roundworm that has been used by scientists for decades to understand the basic organization and function of the central nervous system and how it impacts behavior. Researchers found C. elegans hermaphrodites (the equivalent of females in this species) produce a pheromone that allows worms to monitor how crowded their environment is and how much food there is to go around. When food becomes scarce the aversion circuit is trigged in the animal and it becomes repelled by the pheromone.
“The key thing we identified is a molecular mechanism whereby an instinctive response can be suppressed under particular environmental conditions, namely, abundant food,” said Douglas Portman, Ph.D., lead author of the study and professor of Biomedical Genetics. “Adaptively it makes sense that an animal’s instinctive response would have this kind of flexibility.”
This underlying repulsive mechanism to the pheromone is present in both hermaphrodites and males, but researchers found that in males, the mechanism is overridden by another circuit that causes males to be attracted to the pheromone. A subtlety that could provide an understanding of how the neural circuits work that cause this change in behavior.
Understanding how basic decision-making mechanisms work gives insight into the inner workings of a more complex brain. “These findings lend important insight into the mechanisms by which animals detect and integrate multiple sensory cues to make adaptive behavioral decisions. Understanding how things like this work at the molecular level, provides a framework for understanding how much more complex brains work, and how genetic and environmental insults can ‘break’ things and lead to behavioral and psychiatric disorders.”
Postdoctoral fellow Jintao Luo, Ph.D., with the University of Rochester, co-authored this study. The research was supported with funding from the National Institute of General Medical Sciences.
Read More: Appetite for survival: Brain signal alerts roundworms to changing food supplyUnderlying instincts: An appetite for survival
Wednesday, August 25, 2021
Microscopic roundworms may hold the key to understanding what is happening in the brain when the instinct of an animal changes in order to survive. In a newly published paper in the journal Current Biology University of Rochester Medical Center researchers found that a signaling system in the brain changes to redirect the behavior of an animal when their survival is at risk because there is not enough food.
The experiments were conducted in C. elegans – a microscopic roundworm that has been used by scientists for decades to understand the basic organization and function of the central nervous system and how it impacts behavior. Researchers found C. elegans hermaphrodites (the equivalent of females in this species) produce a pheromone that allows worms to monitor how crowded their environment is and how much food there is to go around. When food becomes scarce the aversion circuit is trigged in the animal and it becomes repelled by the pheromone.
“The key thing we identified is a molecular mechanism whereby an instinctive response can be suppressed under particular environmental conditions, namely, abundant food,” said Douglas Portman, Ph.D., lead author of the study and professor of Biomedical Genetics. “Adaptively it makes sense that an animal’s instinctive response would have this kind of flexibility.”
Read More: Underlying instincts: An appetite for survivalTom O'Connor Study Says Sensitive Parenting Saves Money
Thursday, July 15, 2021
The director of the Wynne Center for Family Research found that sensitive, responsive parenting correlates directly with financial benefits for families and society. "We show for the first time that early parenting quality predicted financial costs associated with health, education and welfare approximately a decade later," he says.
Read More: Tom O'Connor Study Says Sensitive Parenting Saves MoneyShraddha Shah publishes first author paper in the Journal of Comparative Neurology
Wednesday, June 2, 2021
Congratulate Shraddha when you see her. Her paper, "Attention differentially modulates multiunit activity in the lateral geniculate nucleus and V1 of macaque monkeys" has just been published in the Journal of Comparative Neurology. The work used rigorous data processing & statistical techniques to offer some clues to why the effects of attention differ vastly in magnitude in humans & macaques.
Attention promotes the selection of behaviorally relevant sensory signals from the barrage of sensory information available. Visual attention modulates the gain of neuronal activity in all visual brain areas examined, although magnitudes of gain modulations vary across areas. For example, attention gain magnitudes in the dorsal lateral geniculate nucleus (LGN) and primary visual cortex (V1) vary tremendously across fMRI measurements in humans and electrophysiological recordings in behaving monkeys. We sought to determine whether these discrepancies are due simply to differences in species or measurement, or more nuanced properties unique to each visual brain area. We also explored whether robust and consistent attention effects, comparable to those measured in humans with fMRI, are observable in the LGN or V1 of monkeys. We measured attentional modulation of multiunit activity in the LGN and V1 of macaque monkeys engaged in a contrast change detection task requiring shifts in covert visual spatial attention. Rigorous analyses of LGN and V1 multiunit activity revealed robust and consistent attentional facilitation throughout V1, with magnitudes comparable to those observed with fMRI. Interestingly, attentional modulation in the LGN was consistently negligible. These findings demonstrate that discrepancies in attention effects are not simply due to species or measurement differences. We also examined whether attention effects correlated with the feature selectivity of recorded multiunits. Distinct relationships suggest that attentional modulation of multiunit activity depends upon the unique structure and function of visual brain areas.
Kathryn-Mary Wakim's Thesis Paper Accepted in Neuropharmacology
Tuesday, June 1, 2021
When you see her, congratulate Kamy on her first authorship of "Assessing combinatorial effects of HIV infection and former cocaine dependence on cognitive control processes: A high-density electrical mapping study of response inhibition" which was accepted to the journal Neuropharmacology.
Stimulant drug use in HIV+ patients is associated with poor personal and public health outcomes, including high-risk sexual behavior and faster progression from HIV to AIDS. Inhibitory control--the ability to withhold a thought, feeling, or action--is a central construct involved in the minimization of risk-taking behaviors. Recent neuroimaging and behavioral evidence indicate normalization of inhibitory control processes in former cocaine users as a function of the duration of drug abstinence, but it is unknown whether this recovery trajectory persists in former users with comorbid HIV. Here, we investigate the neural correlates of inhibitory control in 103 human subjects using highdensity EEG recording as participants performed a Go/NoGo response inhibition task. Four groups of participants were recruited, varying on HIV and cocaine-dependence status. Electrophysiological responses to successful inhibitions and behavioral task performance were compared among groups. Results indicate persistent behavioral and neurophysiological impairment in HIV+ patients' response inhibition despite current abstinence from cocaine. Analysis of task performance showed that HIV+ abstinent cocaine-dependent participants demonstrate the lowest performance of all groups across all metrics of task accuracy. Planned comparisons of electrophysiological components revealed a main effect of scalp site and an interaction between HIV-status and scalp site on N2 amplitudes during successful inhibitions. Analysis of the P3 time region showed a main effect of scalp site and an interaction between HIV-status and cocaine dependence. These results suggest synergistic alterations in the neurophysiology of response inhibition and indicate that abstinence-related recovery of inhibitory control may be attenuated in patients with HIV.
UR Graduate Women in Science (GWIS) 2021 Mentoring-Up Resolution Challenge CONTEST RESULTS
Monday, May 24, 2021
Congratulations, Mentoring-Up Resolution Challenge Winners!
Full-time UR grad students (gender-inclusive) in biomedical, biological, or chemical sciences took charge of their futures by setting and, through mentoring-up, achieving professional and personal goals for Spring 2021. Participants submitted their goals in a January write-up and progress reports on their professional goals in May. The anonymous faculty evaluation committee selected finalists to present on May 20th. All four presenters were selected to win $1500 ea. in technology-related (hardware, software, and/or peripherals) prizes. Of note, submissions for this contest were received from graduate students of 12 different programs!
Read More: UR Graduate Women in Science (GWIS) 2021 Mentoring-Up Resolution Challenge CONTEST RESULTSAndrew Wojtovich is the University’s Student Supervisor of the Year
Friday, May 7, 2021
Andrew Wojtovich, an assistant professor of anesthesiology, pharmacology, and physiology, is the University's Student Supervisor of the Year. The award is selected by a committee of students and staff members and goes to a person who integrates students into their roles, provides guidance and mentoring, and supports the University's Vision and Values.
Congrats Andrew!
Researchers find breastfeeding linked to higher neurocognitive testing scores
Monday, April 26, 2021
New research finds that children who were breastfed scored higher on neurocognitive tests. Researchers in the Del Monte Institute for Neuroscience at the University of Rochester Medical Center (URMC) analyzed thousands of cognitive tests taken by nine and ten-year-olds whose mothers reported they were breastfed, and compared those results to scores of children who were not.
"Our findings suggest that any amount of breastfeeding has a positive cognitive impact, even after just a few months." Daniel Adan Lopez, Ph.D. candidate in the Epidemiology program who is first author on the study recently published in the journal Frontiers in Public Health. "That's what's exciting about these results. Hopefully from a policy standpoint, this can help improve the motivation to breastfeed."
Hayley Martin, Ph.D., a fourth year medical student in the Medical Scientist Training Program and co-author of the study, focuses her research on breastfeeding. "There's already established research showing the numerous benefits breastfeeding has for both mother and child. This study's findings are important for families particularly before and soon after birth when breastfeeding decisions are made. It may encourage breastfeeding goals of one year or more. It also highlights the critical importance of continued work to provide equity focused access to breastfeeding support, prenatal education, and practices to eliminate structural barriers to breastfeeding."
Researchers reviewed the test results of more than 9,000 nine and ten-year-old participants in the Adolescent Brain Cognitive Development (ABCD) study. Variations were found in the cumulative cognitive test scores of breastfed and non-breastfed children. There was also evidence that the longer a child was breastfed, the higher they scored.
"The strongest association was in children who were breastfed more than 12 months," said Lopez. "The scores of children breastfed until they were seven to 12 months were slightly less, and then the one to six month-old scores dips a little more. But all scores were higher when compared to children who didn't breastfeed at all." Previous studies found breastfeeding does not impact executive function or memory, findings in this study made similar findings.
"This supports the foundation of work already being done around lactation and breastfeeding and its impact on a child's health," said Ed Freedman, Ph.D., the principal investigator of the ABCD study in Rochester and lead author of the study. "These are findings that would have not been possible without the ABCD Study and the expansive data set it provides."
Research Funded to Study Efficacy of Early Visual Training after Occipital Stroke
Friday, April 16, 2021
Up to half-a-million people each year suffer occipital strokes that cause loss to some portion of their vision, permanently affecting how they navigate through life.
A team at the University of Rochester recently showed that visual rehabilitation can more effectively reverse some of this blindness if patients are treated in the first few months after their stroke. Such patients will now have the opportunity to become part of a research study at the Flaum Eye Institute of the University of Rochester, sponsored by the National Institutes of Health.
"In occipital strokes, there is a loss of conscious vision opposite the side of the brain where the stroke occurred," said James V. Aquavella Professor of Ophthalmology Krystel Huxlin, Ph.D. The occipital lobe of the brain contains the primary visual cortex, the first cerebral region responsible for complex visual processing and interpretation of signals received from the eye via the optic nerve.
"After two decades of discovery in our lab, we believe we have arrived at a critical point in our understanding of how to maximize vision restoration for cortically-blinded patients," she said. "Within the first few months of having an occipital stroke, retinal ganglion cells, which transmit signals from the eye to the brain, are still largely intact. After six months, these cells show signs of degeneration, making later-onset rehabilitation more difficult to achieve. And any vision recovered at later stages is grainy and limited to the border of the patients' blind fields. It's as if we are looking at a window of opportunity slowly closing."
The $2.5 million National Eye Institute-sponsored R01 grant, which includes funding for a small clinical trial, will support a collaborative team under Huxlin's leadership, which brings together cross-campus expertise from Duje Tadin, Ph.D. (Department of Brain & Cognitive Sciences, UR) and Brent Johnson, Ph.D. (Department of Biostatistics, UR). NYU's Dr. Marisa Carrasco (Department of Psychology and Neural Science) will also contribute to the research.
Patients who have recently suffered a visual stroke--within zero to five months--will be divided into groups to receive vision rehabilitation training. Their first week will be spent in Huxlin's laboratories at the Flaum Eye Institute and Center for Visual Science. Here, a team of graduate students, postdoctoral fellows and ophthalmic imaging specialists will measure each participant's baseline visual functions, and ocular and brain structures, before teaching them the complex rehabilitation routine they must perform. Each patient will then receive equipment to take home, where their therapy will be performed with remote monitoring. They will return to the Flaum laboratory at 6 and 12 months post-stroke for assessments of training efficacy, as well as to measure changes in their ocular and brain structures.
This study is designed to assess how visual restoration potential changes with time after occipital stroke in humans. It will first measure structural and mechanistic aspects of progressive degeneration along the early visual pathways induced by the stroke, correlating them with changes in visual performance in the blind field. It will then contrast the impact of visual training administered at different stages of degeneration, both on the magnitude of recovery and on the process of degeneration itself. These findings will be key to ascertain the degree to which visual training interventions administered early after stroke can prevent or slow retrograde degeneration, preserve the vision that is still present, and help recover some of the vision already lost.
In addition, knowing how long blind-field visual abilities are preserved after stroke and how this relates to the rate of structural degeneration of early visual pathways is critical to assess if interventions that promote neuronal survival and regeneration could be beneficial for this condition. The project is designed to advance scientific knowledge, technical capability and, ultimately, clinical practices for restoring vision and quality of life for people suffering from occipital strokes.
First-ever lab model of human eye offers hope for macular degeneration patients
Tuesday, March 30, 2021
Rochester researchers say their breakthrough could lead to patient-specific treatments.
Age-related macular degeneration (AMD), which leads to a loss of central vision, is the most frequent cause of blindness in adults 50 years of age or older, affecting an estimated 196 million people worldwide. There is no cure, though treatment can slow the onset and preserve some vision.
Recently, however, researchers at the University of Rochester have made an important breakthrough in the quest for an AMD cure. Their first three-dimensional (3D) lab model mimics the part of the human retina affected in macular degeneration.
Their model combines stem cell-derived retinal tissue and vascular networks from human patients with bioengineered synthetic materials in a three-dimensional "matrix." Notably, using patient-derived 3D retinal tissue allowed the researchers to investigate the underlying mechanisms involved in advanced neovascular macular degeneration, the wet form of macular degeneration, which is the more debilitating and blinding form of the disease.
The researchers have also demonstrated that wet-AMD-related changes in their human retina model could be targeted with drugs.
"Once we have validated this over a large sample, the next hope would be to develop rational drug therapies and potentially even test the efficacy of a specific drug to work for individual patients," says Ruchira Singh, an associate professor of ophthalmology at the University's Flaum Eye Institute.
The lab of Danielle Benoit, professor of biomedical engineering and director of the Materials Science Program, engineered the synthetic materials for the matrix and helped configure it, as described in a paper in Cell Stem Cell.
NGP student honored with significant national achievement
Thursday, March 25, 2021
 Sarah Yablonski received an honorable mention on her NSF GRFP proposal!
Sarah Yablonski received an honorable mention on her NSF GRFP proposal!
The Graduate Research Fellowship Program (GRFP) is a National Science Foundation-wide program that provides Fellowships to individuals selected early in their graduate careers based on their demonstrated potential for significant research achievements in science, technology, engineering or mathematics (STEM) or in STEM education. The NSF accords Honorable Mention to meritorious applicants who do not receive Fellowship offers. This is considered a significant national academic achievement.
MRIs from Study by John Foxe & Ed Freedman Reveal More Incidental Findings in Children
Wednesday, March 24, 2021

 Published in JAMA Neurology, scan results from the Adolescent Brain Cognitive Development (ABCD) Study revealed one in 25 children needed further medical evaluation. The findings by the Del Monte faculty members and their colleagues could change our understanding of the prevalence of neurological problems in children and how neuroimaging is used to screen for these problems.
Published in JAMA Neurology, scan results from the Adolescent Brain Cognitive Development (ABCD) Study revealed one in 25 children needed further medical evaluation. The findings by the Del Monte faculty members and their colleagues could change our understanding of the prevalence of neurological problems in children and how neuroimaging is used to screen for these problems.
Read More: MRIs from Study by John Foxe & Ed Freedman Reveal More Incidental Findings in ChildrenAn experimental neurofeedback imaging treatment has emerged for psychiatric patients. Does it work?
Monday, March 22, 2021
Does neurofeedback imaging work for psychiatric patients?
University of Rochester psychologists have found that real-time functional magnetic resonance imaging neurofeedback (rtfMRI-NF) can help a person regulate neural activity in a way that might improve psychiatric illness.
 David Dodell-Feder, an assistant professor of psychology and neuroscience, together with Emily Dudek, who after graduating from Rochester became Dodell-Feder's full-time lab manager, looked at 17 relevant studies that included a total of 410 participants. The findings of their meta-analysis have been published in the journal Neuroscience and Biobehavioral Reviews.
David Dodell-Feder, an assistant professor of psychology and neuroscience, together with Emily Dudek, who after graduating from Rochester became Dodell-Feder's full-time lab manager, looked at 17 relevant studies that included a total of 410 participants. The findings of their meta-analysis have been published in the journal Neuroscience and Biobehavioral Reviews.
According to lead author Dudek, their review found that when people were shown their own brain patterns in real time, they were able to regulate activity in specific regions of the brain. "This training, known as neurofeedback, offers an exciting and novel treatment method for psychiatric illness."
Coauthor Dodell-Feder agrees that the findings are "very promising," especially because there are very few treatments, psychopharmacological included, that specifically target neural circuits known to contribute to psychopathology.
Yet, there are caveats: the data show less clear evidence that volitional control over the brain regions targeted by neurofeedback translates to an improvement in a person's symptoms or cognitive impairments. For example, when analyzing the impact of rtfMRI-NF on symptoms, the team found an approximately 60 percent chance that a randomly selected person who received rtfMRI-NF showed improvement in symptoms compared to a randomly selected person who received a placebo procedure.
Andrew Anderson & Edmund Lalor Explore How Brain Comprehends Sentences
Monday, March 22, 2021

 The Del Monte Institute faculty members and their colleagues studied and described the complex network within the brain that comprehends the meaning of a spoken sentence. The study, published in the Journal of Neuroscience, is an example of how the application of artificial neural networks, or A.I., is enabling researchers to unlock the extremely complex signaling in the brain that underlies functions such as processing language.
The Del Monte Institute faculty members and their colleagues studied and described the complex network within the brain that comprehends the meaning of a spoken sentence. The study, published in the Journal of Neuroscience, is an example of how the application of artificial neural networks, or A.I., is enabling researchers to unlock the extremely complex signaling in the brain that underlies functions such as processing language.
NGP well featured in recent publications
Friday, March 5, 2021
Drs. Ania Majewska and Monique Mendes in a podcast for NINDS on F applications
Monday, March 1, 2021
 Dr. Ania Majewska and NGP alum, Dr. Monique Mendes, will be featured in The National Institute of Neurological Disorders and Stroke's Building Up the Nerve podcast on 3/5/2021. In this episode, our grantee guests discuss how they involved their mentor(s)/sponsor(s) in the application process to ensure the training plan reflects their individual needs and the mentor is able to provide the appropriate level of support and expertise to achieve those training goals.
Dr. Ania Majewska and NGP alum, Dr. Monique Mendes, will be featured in The National Institute of Neurological Disorders and Stroke's Building Up the Nerve podcast on 3/5/2021. In this episode, our grantee guests discuss how they involved their mentor(s)/sponsor(s) in the application process to ensure the training plan reflects their individual needs and the mentor is able to provide the appropriate level of support and expertise to achieve those training goals.
The podcast features Jaroslaw Aronowski, PhD, Professor, UTHealth McGovern Med School; Alexis S. Mobley, MS, PhD Candidate, UTHealth McGovern Med School; Ania Majewska, PhD, Professor, University of Rochester; Monique Mendes, PhD, Postdoctoral Researcher, Stanford University; Mark Wu, MD, PhD, Professor, Johns Hopkins University; Margaret Ho, PhD, Postdoctoral Fellow, Johns Hopkins University School of Medicine.
Spread the word.
Two faculty members received Sloan Awards for research on how the brain perceives the world.
Wednesday, February 17, 2021
Two University of Rochester researchers in the Department of Brain and Cognitive Sciences are being honored with a celebrated award for their contributions to and leadership in the scientific community.
Martina Poletti and Manuel Gomez-Ramirez, both assistant professors of brain and cognitive sciences and of neuroscience, are among this year's recipients of Sloan Research Fellowships. Awarded annually by the Alfred P. Sloan Foundation since 1955, the fellowships recognize young scientists for their independent research accomplishments, creativity, and potential to become leaders in the scientific community. Each fellowship carries a $75,000, two-year award. This year, 128 scientists across the US and Canada were awarded fellowships. Gomez-Ramirez and Poletti are the University's fourth and filth Sloan fellows in the last three years.
Read More: Two faculty members received Sloan Awards for research on how the brain perceives the world.2020 in a Word
Tuesday, February 16, 2021
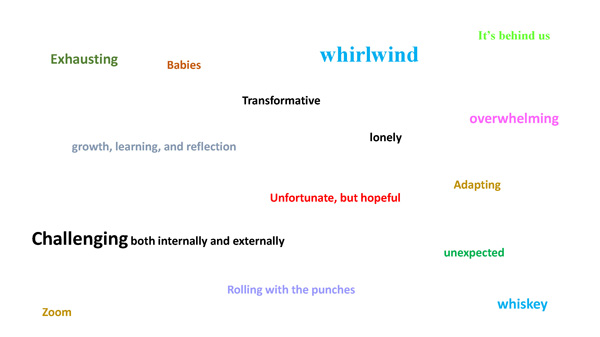
During the NGP 2020 Holiday Party, students were asked to provide a word to describe 2020.
The image below reflects the results. A larger font size indicates a word mentioned more than once.
Brain changed by caffeine in utero, study finds
Thursday, February 4, 2021
New research finds caffeine consumed during pregnancy can change important brain pathways in baby.
New research finds caffeine consumed during pregnancy can change important brain pathways that could lead to behavioral problems later in life. Researchers in the Del Monte Institute for Neuroscience at the University of Rochester Medical Center (URMC) analyzed thousands of brain scans of nine and ten-year-olds, and revealed changes in the brain structure in children who were exposed to caffeine in utero.
"These are sort of small effects and it's not causing horrendous psychiatric conditions, but it is causing minimal but noticeable behavioral issues that should make us consider long term effects of caffeine intake during pregnancy," said John Foxe, Ph.D., director of the Del Monte Institute for Neuroscience, and principal investigator of the Adolescent Brain Cognitive Development or ABCD Study at the University of Rochester. "I suppose the outcome of this study will be a recommendation that any caffeine during pregnancy is probably not such a good idea."
Elevated behavioral issues, attention difficulties, and hyperactivity are all symptoms that researchers observed in these children. "What makes this unique is that we have a biological pathway that looks different when you consume caffeine through pregnancy," said Zachary Christensen, a M.D/Ph.D. candidate in the Medical Science Training Program and first author on the paper published in the journal Neuropharmacology. "Previous studies have shown that children perform differently on IQ tests, or they have different psychopathology, but that could also be related to demographics, so it's hard to parse that out until you have something like a biomarker. This gives us a place to start future research to try to learn exactly when the change is occurring in the brain."
Investigators analyzed brain scans of more than 9,000 nine and ten-year-old participants in the ABCD study. They found clear changes in how the white matter tracks -- which form connections between brain regions -- were organized in children whose mothers reported they consumed caffeine during pregnancy.

Researchers analyzed the brain scans of more then 9,000 nine and ten-year-olds and found a change in important brain pathways in those whose mothers retrospectively reported consuming caffeine while pregnant.
URMC is one of 21-sites across the country collecting data for the ABCD study, the largest long-term study of brain development and child health. The study is funded by the National Institutes of Health. Ed Freedman, Ph.D., is the principal investigator of the ABCD study in Rochester and a co-author of the study.
"It is important to point out this is a retrospective study," said Foxe. "We are relying on mothers to remember how much caffeine they took in while they were pregnant."
Previous studies have found caffeine can have a negative effect on pregnancy. It is also known that a fetus does not have the enzyme necessary to breakdown caffeine when it crosses the placenta. This new study reveals that caffeine could also leave a lasting impact on neurodevelopment.
The researchers point out that it is unclear if the impact of the caffeine on the fetal brain varies from one trimester to the next, or when during gestation these structural changes occur.
"Current clinical guidelines already suggest limiting caffeine intake during pregnancy -- no more than two normal cups of coffee a day," Christensen said. "In the long term, we hope to develop better guidance for mothers, but in the meantime, they should ask their doctor as concerns arise."
Motion got you feeling queasy? It may be all in your head... or your ears
Friday, January 29, 2021
New research from the University of Rochester Medical Center has detailed a part of the nervous system by which the brain can modify our sense of balance. The current study expands our understanding of how balance stimuli are received by the brain while also offering insights into potential drug targets in the ear, which may be leveraged for treating motion sickness and balance disorders.
"In my opinion, these data are one of the first steps in beginning to unravel the functional significance of the efferent vestibular system," Joseph C. Holt, Ph.D., senior author of the study published in the journal Scientific Reports said. The efferent vestibular system (EVS) begins as a small collection of neurons that travel from the brainstem out to the ear where our sense of balance begins. While there is still little known about the function of the EVS, URMC researchers are uncovering more about the role these neurons may play in processing motion stimuli and maintaining our balance.
NGP student wins award at NE Regional APSA Conference
Wednesday, January 13, 2021
Congratulation Udaysankar Chockanathan whose talk at the American Physician Scientists Association won second place. Uday presented his exciting work on population coding in the APP/PS1 mouse model of Amyloid pathology.