Using AI and Ultrasound to Evaluate Flexor Tendon Injuries
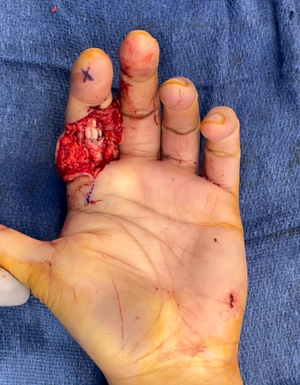 University of Rochester researchers are collaborating on a new approach to evaluate flexor tendon injuries: longitudinal 3D quantification of tendon scar tissue as a predictor of outcome. They’re also employing novel tools to aid their research efforts: artificial intelligence to automate analysis of thousands of diagnostic images, and a 3D-printed device that minimizes preventable variation in the collection of ultrasound images.
University of Rochester researchers are collaborating on a new approach to evaluate flexor tendon injuries: longitudinal 3D quantification of tendon scar tissue as a predictor of outcome. They’re also employing novel tools to aid their research efforts: artificial intelligence to automate analysis of thousands of diagnostic images, and a 3D-printed device that minimizes preventable variation in the collection of ultrasound images.
Near-term, musculoskeletal researcher Alayna Loiselle, PhD, and clinician-scientist and orthopaedic surgeon Constantinos Ketonis, MD, PhD aim to improve the way physicians evaluate post-op patients to optimize their physical therapy and recovery; long-term, they are working to expedite research on the next frontier – biologic therapies to minimize scar formation.
Using 3D ultrasound images, the physician and researcher have already demonstrated that the amount of scar formation is a predictor of physical function in animal models and now they’re now translating their findings to human studies.
What are the advantages of ultrasound evaluation over current methods?
Ketonis: We can non-invasively monitor healing progress and tailor therapy to each patient’s physiology. We believe the amount of scar tissue that’s forming post-operatively is predictive of a patient’s function. Traditional evaluation of post-op progress focuses on physical examination of patients’ functional performance. Employing 3D ultrasound imagery to actually evaluate the quality of the repair and see the amount of scar formation a patient developed could enable more precise rehabilitation regimens – tailored to each patient – for the best possible recovery.
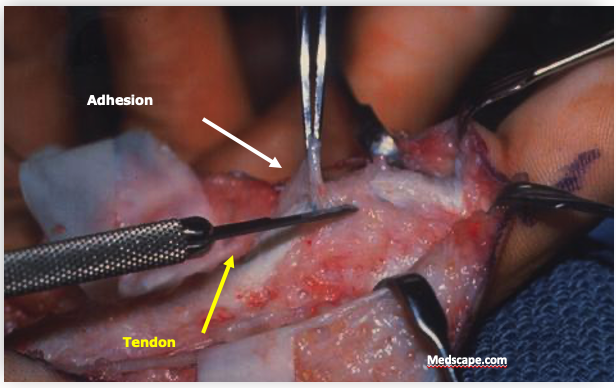
Scar tissue that formed in a sutured tendon is shown during a second procedure to lyse the adhesions
Loiselle: Ultrasound of scar tissue is more data-driven than evaluation of a patient’s function. Without this imaging there’s no way to visualize injury site healing, so physicians have to subjectively determine how well a patient is progressing and make therapy decisions, with less information than an ultrasound can provide.
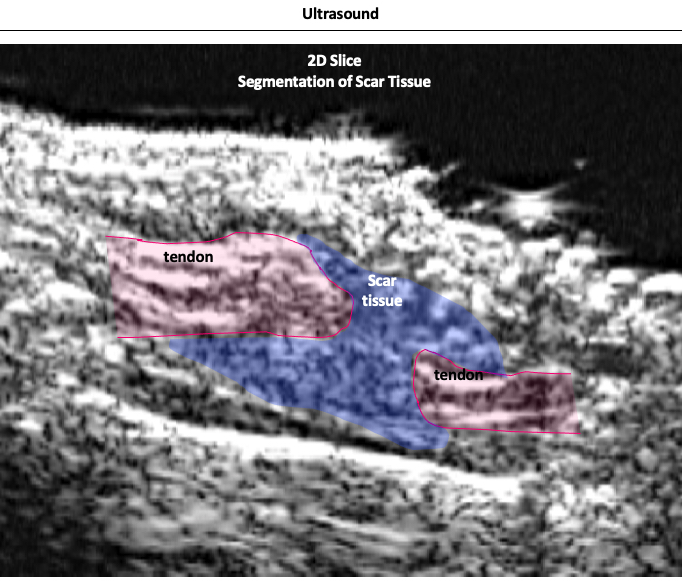
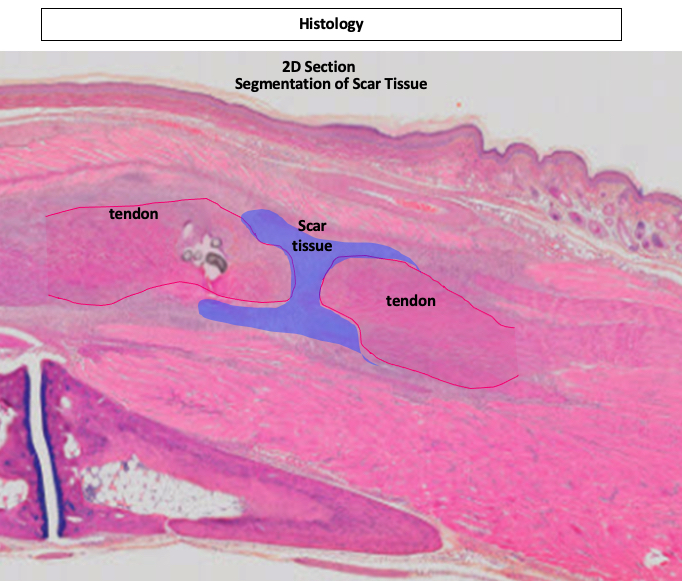
Ultrasound imaging, aided by artificial intelligence, helps the researchers quantify the amount of scar tissue non-invasively and more efficiently than histology.
Tell us about the technology/tools that have advanced this research.
Loiselle: In our animal models, we have been using deep learning to automate scar detection in large numbers of ultrasound images. We’ve segmented thousands of images to train the machine to “see” scar tissue vs. surrounding normal tissue. Because hand-held ultrasound is subject to a lot of variation between operators, we’ve developed a device that holds the patient’s hand steady during the ultrasound. That consistent image collection enhances the ability of AI to identify and quantify the scar tissue.
Ketonis: We created a motorized 3D-printed jig that assists in reproducible automated ultrasound scanning of a finger that is then reconstructed into a 3D model of the tendon and scar. The goal is to train an AI algorithm to automatically segment and quantify the scar volume, and by using predictive analytics, to provide insights into the individual patient’s future path. This would then inform decision-making for therapists and clinicians to modify treatment, whether that is more aggressive physical therapy regimens or perhaps earlier surgical interventions.
Using this technology, the process is: surgical repair of the tendon injury; ultrasound imaging post-op; AI quantification of the scar; evaluation of the scar and its potential impact on function; then tailoring the patient’s therapy based on those findings to deliver effective rehabilitation that preserves as much long-term function as possible.
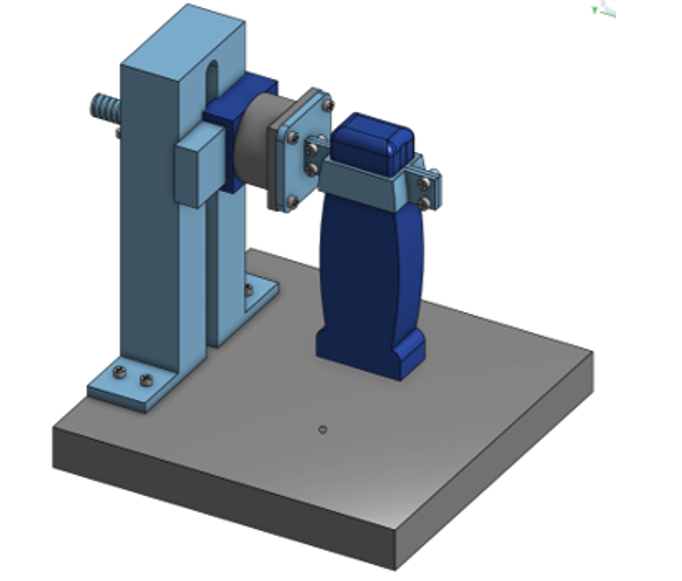

A motorized jig invented by the researchers automates and standardizes capture of tendon-healing images.
What are the findings from your human trial?
Loiselle: We have recently completed our first longitudinal pilot study, which completed ultrasound scans of 9 patients that underwent a primary flexor tendon repair in the finger, with scans beginning 3-4 weeks post-op and follow-up scans through 12 months. We are now in the process of finalizing the data analysis process, but early results are promising in terms of potentially being able to use the 3D volumetric quantification of scar tissue to predict which patients will go on to have satisfactory outcomes (good restoration of finger range of motion).
Ketonis: We also have been collaborating with an investigator at Rochester Institute of Technology (RIT) to develop additional ultrasound-based metrics that provide information about the strength of the repair, with the goal of understanding what level of physical therapy the tendon repair may be able to withstand.
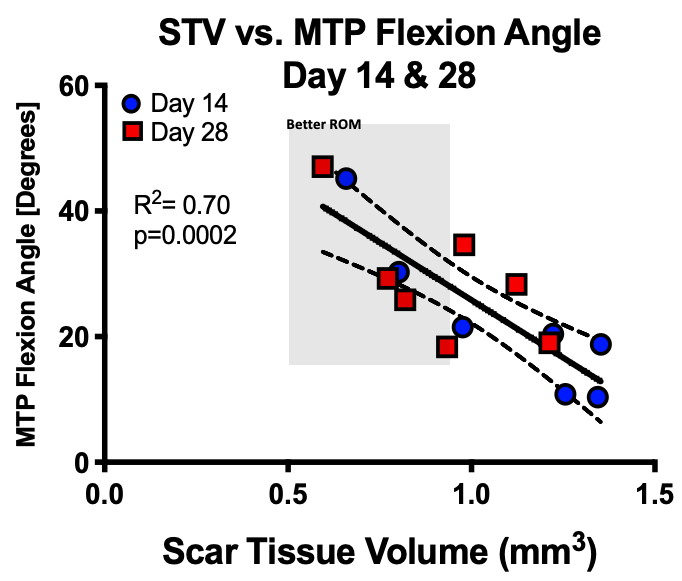
The researchers are analyzing the relationship between quantity of scar tissue in a healing tendon and function of the hand
What is the end goal of your research? How will it benefit patients?
Ketonis: The ultimate goal is to improve patient outcomes. At 300,000 injuries and $20 billion in U.S. health care costs annually, flexor tendon injuries are a significant health issue with potential lifelong disability for many patients. As a surgeon, my concern is that flexor tendon healing is complicated by range of motion-limiting scar formation in up to 40 percent of surgical repairs. We often have to perform a secondary surgical procedure to lyse the adhesions that form.
Loiselle: The primary goal of my lab is to understand the cellular and molecular drivers of tendon scar formation so we can better identify promising biological therapies to improve the healing process. However, physical therapy also has great potential to improve patient outcomes, so we feel like pursuing both the fundamental science and translational studies in parallel will give us the greatest chance for success.