PD-L1 Testing for Lung Cancer
Lung Cancer Detection
About the Test
URMC is one of the only institutions in Western New York to offer this testing to medical oncology professionals using an FDA-approved kit. Add PD-L1 IHC 22C3 pharmDx to your lung cancer panel to inform your treatment options for metastatic NSCLC patients. In patients with advanced NSCLC and PDL1 expression in at least 50% of tumor cells, pembrolizumab was associated with significantly longer progression-free and overall survival and with fewer adverse events than platinum-based chemotherapy.*
How it Works
- Assay performed on formalin-fixed, paraffin-embedded (FFPE) tissue specimen of at least 100 cells taken from NSCLC patient.
- Assay used to determine PD-L1 expression by IHC.
- Pathologist determines tumor proportion score (TPS) category and numerical TPS.
- Oncologist determines treatment based on TPS number and treatment history.
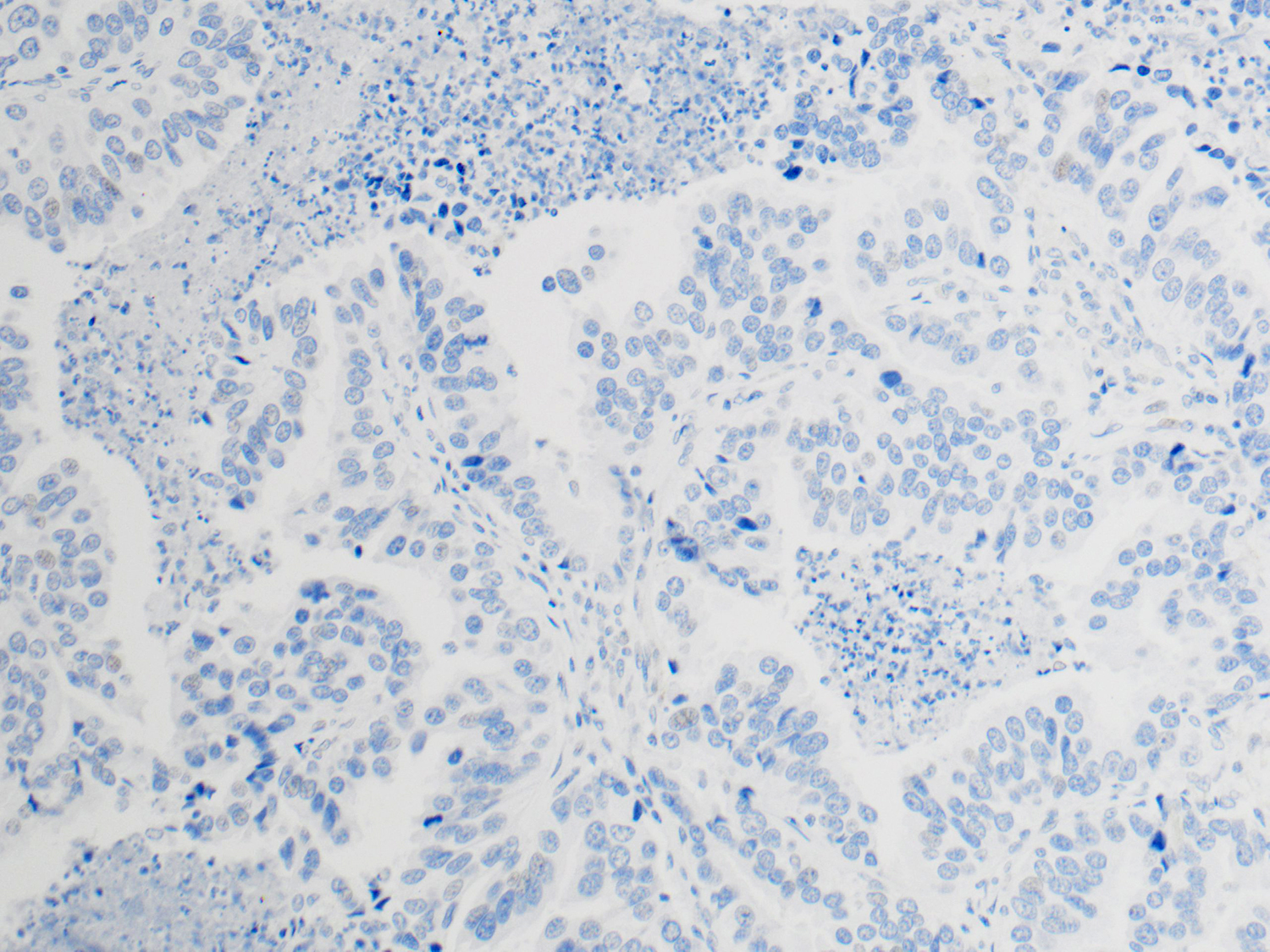
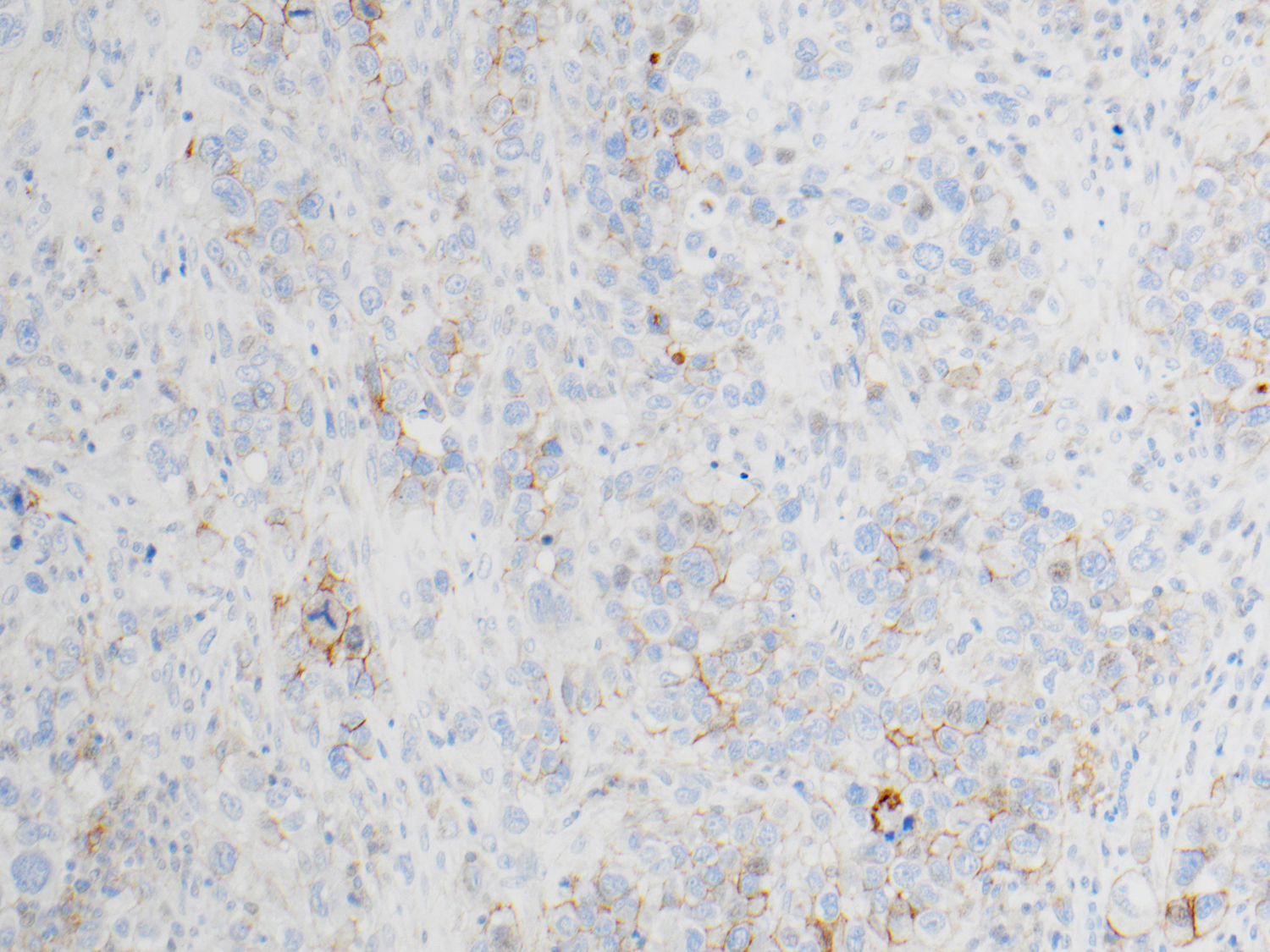
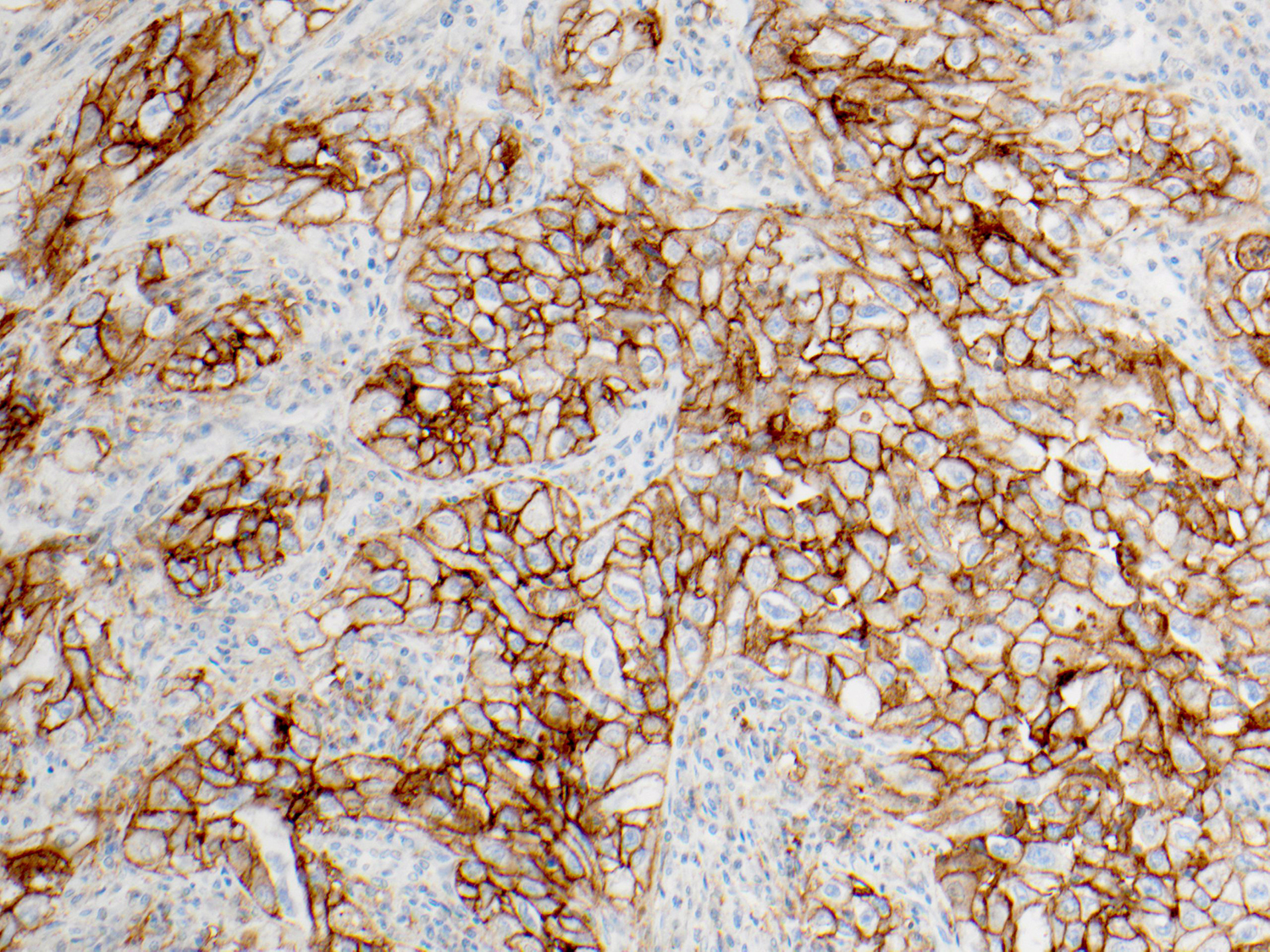
NSCLC: Analysis for PD-L1 Expression
Protein expression is determined using Tumor Proportion Score (TPS), the percentage of viable tumor cells showing partial or complete membrane staining at any intensity. The specimen is considered to have PD-L1 expression if TPS ≥ 1% and high PD-L1 expression if TPS ≥ 50%.
Contact Us
*References:
Reck M, Rodriguez-Abreu D., et. al; Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. NEJM 2016;375(19):1823.