Welcome to the Electron Microscopy Resource
The goal of the Electron Microscopy Shared Resource Laboratory (EM-SRL) is to support investigators in their pursuit of studies involving electron microscopy with state-of-the-art technology, technical expertise, and staff to streamline their projects. The EM-SRL supports your projects from initial project design through imaging interpretation utilizing either in house transmission electron microscopes (TEM) (Hitachi 7650 or FEI Talos 120C) or a Zeiss Auriga scanning electron microscope (SEM) on the River Campus, including Cryo-EM utilizing the FEI Talos 120C. To arrange a consultation, please get in touch!
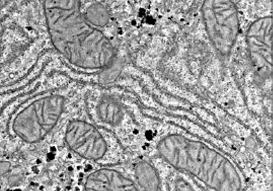
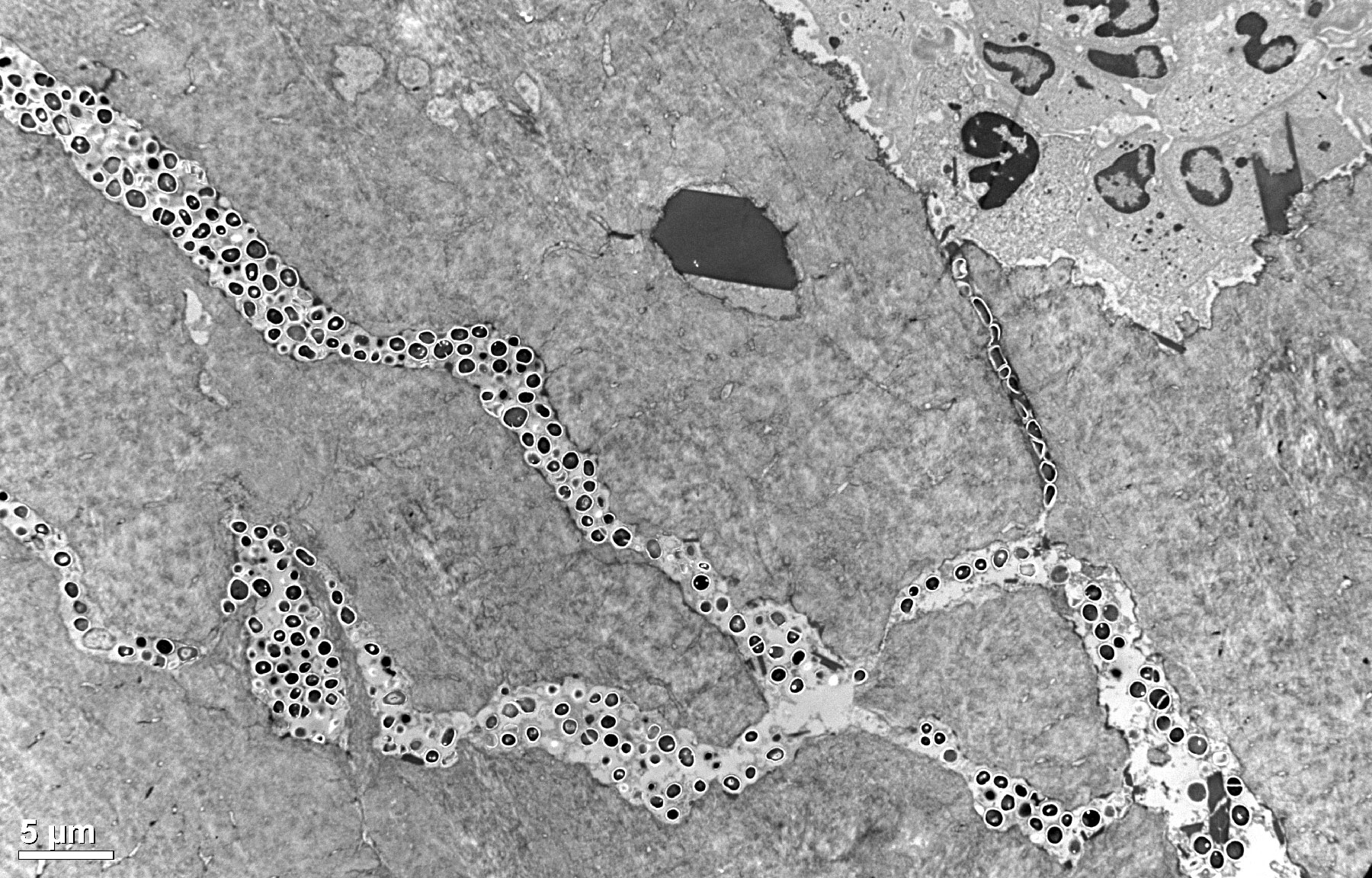
TEM
Transmission EM to examine thin-sectioned tissue ultrastructure and changes in experimental and pathologic conditions.

Cryo TEM
Visualization of vitrified protein, macromolecular complexes and biologic solutions at cryogenic temperatures.

SEM
Scanning EM to examine the external surface structure of biologic or inorganic specimen.
Microscopic bacterial abscesses are key aspects of orthopaedic infections. We utilize scanning electron microscopy to assess bacterial biofilm on implants, and transmission electron microscopy to study bone marrow abscesses and bacteria colonization of sub-micron channels in bone, to help URMC researchers elucidate how virulent microbes like methicillin-resistant Staphylococcus aureus (MRSA) establish chronic skeletal infections and evaluate the safety and efficacy of novel bone-targeted antibiotics and immunizations." - Eddie Schwarz, Ph.D.
Contact Information
Electron Microscopy Resource
University of Rochester Medical Center
601 Elmwood Ave.
Rochester, NY 14642
B.7811
Email: Karen_Bentley@URMC.rochester.edu
Email: Chad_Galloway@URMC.rochester.edu
Phone: (585) 275-1954