URMC / Labs / Mosmann Lab / Projects / Effector Functions of Influenza-Specific T Cells Induced by Immunization and Infection
Effector Functions of Influenza-Specific T Cells Induced by Immunization and Infection
Diversity of the CD4 T Cell Response to the 2009 Pandemic H1N1 Influenza
CD4 T cells are typically classified into different subsets based upon cytokine expression, and hence functionality. However, the diversity of the cytokine patterns within a given subset, Th1 for example, is still quite heterogeneous. There are several mechanisms, not mutually exclusive, that could lead to the observed heterogeneity in an otherwise pre-committed subset within a T helper population. Cells having the greatest diversity in effector molecules correlate well with more effective responses in many immunological challenges. Therefore understanding the quality of the anti-influenza T cell response is an important parameter in assessing the robustness of natural infection and vaccination. The emergence of the 2009 pandemic H1N1 exhibited the pathogenic threat of a novel quadruple-reassortant strain of influenza and provides the ability to assess a 'new' T cell response prior to and after natural infection. Analyzing the pre-committed population of T helper cells will not only enlighten us to the quality of the T cells, but also lead to a broader understanding of the mechanisms that may control the regulation of heterogeneity in cytokine patterns displayed during a pathogen challenge.
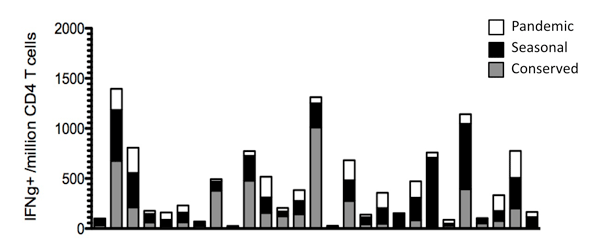
Assessing the history of different influenza specificities in the general population.
Pregnancy and Influenza Immunity
There is substantial evidence that the immune response is altered during pregnancy. Many passive and active mechanisms protect the fetus from immune attack, and ensure the success of normal semi-allogeneic pregnancy. However, modification of immune responses to aid pregnancy can increase susceptibility to some pathogens, particularly those requiring strong inflammatory Th1-like responses, and ameliorate some autoimmune diseases. Conversely, pregnancy can increase resistance to pathogens cleared by allergic, Th2-like responses. The current 2009 pandemic influenza H1N1 strain appears to cause unusually severe infection in pregnant versus non-pregnant women. Here we are testing whether these immunological manifestations during pregnancy correlate with increased susceptibility in pregnant and non-pregnant women following seasonal influenza vaccination.
Aging and Influenza Immunity
As individuals age their ability to respond and clear pathogens declines, which leads to a greater incidence of severe disease. This increased frequency of illness is a reflection of the age-associated decline in the immune system. Influenza is one pathogen that is a major cause of morbidity and mortality in the elderly and even more so due to the recent novel H1N1 pandemic and potential threat of avian H5N1. Despite the recommendation that high-risk groups, such as the elderly, receive influenza vaccination yearly, there is no direct evidence that supports an immunological protective benefit. Moreover, even in healthy adults, the observation that T cell boosting occurs after vaccination is difficult to visualize. CD4 T cells are key components of the adaptive immune response that provide the necessary help for antibody production and long-lived killer T cells to eliminate an influenza infection. CD4 T cell function is characterized by the cytokines and chemokines produced in response to a pathogen or vaccine. In particular, chemokines are functionally important in the guidance and recruitment of immune cells to lymph nodes and peripheral tissue sites. Importantly, the consequences aging may have on the regulation of chemokine expression by antigen-reactive CD4 T cells are unknown. In this study, we are seeking to understand the role chemokine expression by T cells may have on immune senescence and how this may effect the diversity of the influenza-specific CD4 T cell response after TIV vaccination in the elderly.
Analysis of Cytokine and Transcription Factor Expression in Single Cells
Although current intracellular cytokine secretion (ICS) panels can simultaneously measure 8-10 cytokines in single cells, this is still less than a full description of the effector functions of CD4 T cells. It is also difficult to measure the expression of molecules that may regulate different cytokine expression patterns, such as transcription factors and signaling molecules. Recent advances in cDNA sequencing have opened the possibility of sequencing all mRNA molecules from individual, sorted influenza-specific T cells. We are actively pursuing the description of the effector diversity and functional stability of single-cell, influenza-specific CD4 T cells.