Monoclonal antibody therapy of lymphoid malignancies is dependent on effective macrophage phagocytosis
The addition of anti-CD20 monoclonal antibody (mAb) to chemotherapies for the treatment of CLL was the first real significant improvement in patient outcomes and survival. Anti-CD20 mAb therapy targets this molecule that is expressed on the surface of CLL cells. CLL cells coated with with this mAb are subsequently eliminated by the body’s innate immune system. Current data by our lab and others indicate that the major mechanism of removal of anti-CD20 coated CLL cells is via antibody-dependent cell phagocytosis (ADCP) by tissue macrophages. Initial clearance of anti-CD20 mAb treated CLL cells from the blood of patients is dramatic, but subsequently patient CLL cells develop resistance to anti-CD20 mAb therapy. Thus, despite the significant advance in treatment of CLL patients with the addition of anti-CD20 mAb to chemotherapies, it is still not curative, and most CLL patients will die from progressive disease or its complications. Therefore, our laboratory is interested in understanding the basis for mAb therapy resistance.
In collaboration with the laboratories of Clive S. Zent, M.D. in the Wilmot Cancer Institute, and Michael R. Elliott, Ph.D. in the Center for Vaccine Biology and Immunology, we are studying the regulation of macrophage ADCP, with a focus on understanding the inhibition of ADCP relating to mAb therapy resistance. Towards this end, we are developing time-lapse image techniques to measure the kinetics of ADCP in detail.
Our long-term goal is to apply these new findings toward the development of more effective mAb therapies for CLL and other types of cancer.
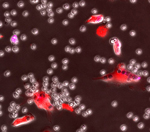
Above: Time-lapse video image of bone marrow derived macrophages (labeled with red dye) engulfing mAb coated CLL cells (labeled with pH sensitive purple dye).