News
20242023202220212020
Research: How the Immune System Fails as Cancer Arises
Wednesday, September 11, 2024
Cancer has been described as “a wound that does not heal,” implying that the immune system is unable to wipe out invading tumor cells. A new discovery confirms that a key molecule can reprogram immune cells that normally protect against infection and cancer, turning them into bad guys that promote cancer growth.
Studying the behavior of these “pro-tumor” immune cells is important because they could be targets for therapies that block their harmful activity, said Minsoo Kim, PhD, corresponding author of the study and a research leader at the Wilmot Cancer Institute.
The Proceedings of the National Academy of Sciences (PNAS) published the discovery.
Kim led a team of scientists investigating the dynamic interactions that occur between cells in the tumor environment, and the underlying factors that cause the harmful transformation of immune cells from good to bad.
They found that PAF (platelet-activating factor) is the key molecule that controls the destiny of the immune cells. PAF not only recruits cancer-promoting cells, but it also suppresses the immune system’s ability to fight back. In addition, they found that multiple cancers rely on the same PAF signals.
Read More: Research: How the Immune System Fails as Cancer ArisesWhat Is H5N1 Bird Flu, How it Spreads to Humans, and Safety Concerns
Thursday, June 20, 2024
 The H5N1 virus, also known as avian or bird flu, is once again in the news and on the radar of scientists and public health officials as a growing number of infections have been found in dairy cattle and farm workers.
The H5N1 virus, also known as avian or bird flu, is once again in the news and on the radar of scientists and public health officials as a growing number of infections have been found in dairy cattle and farm workers.
David Topham, PhD, a professor of Microbiology and Immunology and director of the University of Rochester Translational Immunology and Infectious Disease Institute, has been studying the H5N1 and other influenza viruses for 27 years.
Read More: What Is H5N1 Bird Flu, How it Spreads to Humans, and Safety ConcernsA Game Plan for Timing Your Flu, Covid and RSV Shots This Fall with Commentary by David Topham
Tuesday, September 19, 2023
Trying to figure out when a virus will peak is a guessing game, says David Topham, the Marie Curran Wilson and Joseph Chamberlain Wilson Professor in the Department of Microbiology and Immunology. While respiratory viruses often pick up in the fall and peak in the winter, in recent years the COVID-19 pandemic disrupted the usual patterns. RSV and influenza activity started earlier than usual last year. “COVID-19 has really thrown everything into turmoil,” says Topham.
Read More: A Game Plan for Timing Your Flu, Covid and RSV Shots This Fall with Commentary by David TophamWilmot Researchers Discover How to Steer Army of Immune Cells toward Cancer
Monday, April 17, 2023
Findings will Guide Improvements to CAR T-Cell Therapy for Breast, Lung, Brain Cancer
Immunotherapy, particularly CAR T-Cell treatment for cancer, is extending the lives of many patients. But sometimes the therapy randomly migrates to places it shouldn’t go, tucking into the lungs or other noncancerous tissue and causing toxic side effects. A Wilmot Cancer Institute team discovered the molecule responsible for guiding T cells toward tumors, setting the stage for scientists to improve upon the groundbreaking treatment.
The next step is to find a drug that can manipulate the key T-cell protein, ST3GAL1, said Minsoo Kim, Ph.D., corresponding author of an article in Nature Immunology that describes the research. If the investigation evolves as planned, such a drug could be added to the CAR T-cell regimen to ensure that T cells hit their targets, Kim said.
His lab is collaborating with other Wilmot investigators to screen for drugs that will accomplish that feat, while also minimizing the risk of life-threatening side effects.
“You can create very powerful treatments,” said Kim, co-leader of Wilmot’s Cancer Microenvironment research program at the University of Rochester, “but if they can’t get to their targets or they go to the wrong place, it does not provide the outcome you intended.”
Read More: Wilmot Researchers Discover How to Steer Army of Immune Cells toward CancerWho Should Get a Second Covid-19 Booster Shot?
Friday, April 1, 2022
The FDA authorized a fourth shot for Americans age 50 and older. Here’s what scientists say you should know.
 Dr. Topham was quoted in the Wall Street Journal article.
Dr. Topham was quoted in the Wall Street Journal article.
Americans are once again wondering whether they need another Covid-19 shot, with many growing weary of vaccinations.
The U.S. Food and Drug Administration this week authorized a second Covid-19 booster for anyone 50 years and older who had their first booster four or more months ago.
Experts are split on how important it is to get a second booster in light of the low Covid-19 rates in the U.S. and evidence of high protection against death and hospitalization following the first booster. But other countries are experiencing a surge in cases from the Omicron subvariant BA.2, and there are signs that rates are increasing in some parts of the Northeast.
Read More: Who Should Get a Second Covid-19 Booster Shot?Faculty Highlighted as "Inspiring Women in Science"
Friday, February 11, 2022
Juilee Thakar, PhD, Associate Professor of Microbiology and Immunology, and Paige Lawrence, PhD, Professor and Chair of Environmental Medicine, and Professor of Microbiology and Immunology were both highlighted in URMC's reflection on International Day of Girls and Women in Science. Read more about their stories entering their respective fields and their current work in the article, "Meet Five Inspiring Women in Science".
Edith Lord Recognized by American Association of Immunologists
Thursday, February 3, 2022
The professor of Microbiology & Immunology and Oncology was recently honored with the AAI Distinguished Service Award and named as a Distinguished Fellow of AAI – Class of 2022. Lord, who studies the microenvironment of solid tumors and has an appointment at the Wilmot Cancer Institute, joined AAI in 1978 and served as Secretary-Treasurer from 2015–2021. Both honors pay tribute to her outstanding scientific contributions and her service to AAI and the immunology community.
Checking out the Ingredient Label on Vaccines
Sunday, November 28, 2021
Dr. David Topham, PhD was featured in Bloomberg's Corona Virus daily.
Nearly a year into the rollout, when shot talk has become part of daily life, it can be easy to take for granted some of the most basic (and confusing) things about vaccines. Much like reading the ingredients on your favorite candy bar or soda, looking at the fine print can invite more questions than answers.
“This is a boring way to answer, but I'd refer the reader to this CDC website,” says David Topham, an immunologist at the University of Rochester Medical Center, referring to the U.S. Centers for Disease Control and Prevention.
“The vaccines each have a number of components with the two mRNA vaccines being mostly lipid (fat) and mRNA coding for the spike protein of the virus,” says Topham. That’s right — mRNA, or messenger ribonucleic acid, is actually one of the ingredients in both the Pfizer and Moderna vaccines. It’s what tells the body how to create a (harmless) piece of viral protein to build up an immune response against SARS-CoV-2. Otherwise, those vaccines are composed of fats that help the mRNA enter the body’s cells, and sugars and acid stabilizers to help keep the vaccine stable as it’s manufactured and shipped around the world.
Read More: Checking out the Ingredient Label on VaccinesResearchers Awarded Grant to Combat Vaccine Hesitancy in Adolescents
Tuesday, November 16, 2021
The Environmental Medicine team, including Dr. Danielle Alcéna-Stiner, IMV graduate and Assistant Professor at UR School of Nursing, received an NIH award to address hesitancy and improve health literacy among middle- and high-school students. The team will work with Rochester-area teachers and health professionals to teach students how COVID-19 spreads, how COVID testing works, what RNA is, and how the vaccine works.
Read More: Researchers Awarded Grant to Combat Vaccine Hesitancy in AdolescentsUR Graduate Women in Science (GWIS) 2021 Mentoring-Up Resolution Challenge CONTEST RESULTS
Monday, May 24, 2021
Congratulations, Mentoring-Up Resolution Challenge Winners!
Full-time UR grad students (gender-inclusive) in biomedical, biological, or chemical sciences took charge of their futures by setting and, through mentoring-up, achieving professional and personal goals for Spring 2021. Participants submitted their goals in a January write-up and progress reports on their professional goals in May. The anonymous faculty evaluation committee selected finalists to present on May 20th. All four presenters were selected to win $1500 ea. in technology-related (hardware, software, and/or peripherals) prizes. Of note, submissions for this contest were received from graduate students of 12 different programs!
Read More: UR Graduate Women in Science (GWIS) 2021 Mentoring-Up Resolution Challenge CONTEST RESULTSYou Got the COVID Vaccine. How Long Are You Protected?
Wednesday, April 14, 2021
As more states expand eligibility and vaccination rates continue to rise, pre-pandemic normalcy is on the horizon. The question now on many people's minds is how long vaccines will provide protection. While the short answer is it's too soon to know with certainty, experience with other infectious diseases can help scientists make an educated guess. And it is good news -- your immune system will probably be able to detect and quickly respond to the coronavirus for many years.
"Long-term data from vaccinated individuals will give us the definitive answer, but all signs point to the fact that a level of protection from vaccines should last for a significant period of time, potentially two to three years or longer," said David Topham, Ph.D., the director of the University of Rochester Medical Center Translational Immunology and Infectious Diseases Institute. Topham and his colleagues at URMC have decades of experience studying the immune response to respiratory infections. His work on this subject has been featured recently in The Washington Post and The Wall Street Journal.
One of the sources of uncertainty is the simple fact that currently approved COVID vaccines are new, so researchers don't have hard data on the durability of immunity to the virus. While around 100 million Americans have received at least one dose, the bulk of these vaccinations have only occurred in the last couple of months. However, data from a smaller group who participated in clinical trials provide some evidence that vaccines are producing a long-lasting immune response. Both Moderna and Pfizer-BioNTech recently announced that their vaccines remain 91% effective in preventing infection, and 100% effective in preventing severe disease six months after the second dose.
How Long Do Covid-19 Vaccines Provide Immunity?
Monday, April 12, 2021
How long does protection from Covid vaccines last? It's a question that's becoming more important as some of the first people to be vaccinated approach four months post-inoculation.
The short answer is: We don't fully know yet. But more data is coming in that provides clues. Here's what we know so far.
How long are we protected from getting Covid-19? Does the efficacy decline over time?
Recent data from Pfizer, the manufacturer of one of the three vaccines available in the U.S., indicates that protection lasts at least six months. The results showed minimal antibody decline. Recipients of the Moderna vaccine also had robust levels of antibodies more than six months later, according to a recent study published in NEJM.
Some people have incorrectly concluded that means that those vaccines offer only six months of protection, says Scott Hensley, a professor of microbiology at the University of Pennsylvania. "That's false," says Dr. Hensley. "We only have six months of data...Six months from now it's likely we'll learn we have one year of protection."
Boosters will likely be needed for at least a few years "out of an abundance of caution, knowing that immunity does wane in some individuals more than others," says David Topham, the Marie Curran Wilson and Joseph Chamberlain Wilson Professor in the Department of Microbiology and Immunology and the founding director of the Translational Immunology and Infectious Disease Institute. The article also appeared on MSN News.
Should I Take Tylenol After My COVID Shot? Can I Drink a Beer?
Friday, April 9, 2021
Here we are, finally on the cusp of our long-awaited Hot Vaxx Summer. About 112 million people in the U.S. have received at least one dose of a COVID-19 vaccine, and if you haven't gotten one yet, chances are you will be able to soon: Starting April 19, the general public over 16 will become eligible. Among people who are enthusiastic about vaccination, there's a mix of excitement—after more than a year of living through the pandemic, some light at the end of the tunnel!—along with some nervousness about side effects. Some who have been lucky enough to be vaccinated already have reported chills, fevers, fatigue, or even vomiting after receiving a vaccine dose. (Side effects are more common after the second dose of the Pfizer or Moderna vaccines.)
As a result, people are getting all sorts of advice about what to do—or not to do—in the hours leading up to and following a COVID-19 vaccination. Slate staffers report receiving general advice, like staying hydrated or avoiding alcohol. Meanwhile, my parents were advised to adhere to a very specific routine: Drink one glass of water before the shot with three days' worth of Vitamin C supplements, then one glass of water after.
Other advice, especially around taking over-the-counter painkillers like ibuprofen or acetaminophen has been contradictory.* One friend told me the nurse who administered her shot told her to take ibuprofen afterward; meanwhile, on social media, people have posted about taking pain meds even before their vaccination appointment, in hopes it could head off soreness at the injection site or muscle aches. But others tell me they've heard to specifically avoid ibuprofen and use acetaminophen instead, or advised to avoid pain killers entirely. What does the science say?
To understand that, it's worth reviewing what vaccines actually do. While the Pfizer and Moderna vaccines use a different method from Johnson & Johnson's vaccine, the result is the same: The vaccine teaches your immune system to recognize coronavirus. In that process, your body recruits specific types of cells to identify and clean up invader cells. One type of cell, called helper T-cells, aids another, called B-cells, which release antibodies that target coronavirus. Essentially, the vaccines serve as a rehearsal for your immune system to practice how to spot and clear coronavirus cells, so that if it encounters the real thing, it's ready.
But if you do end up taking over-the-counter painkillers with your vaccine—or already did—it's probably not a huge deal. The overall effect of NSAIDs is likely pretty small. Among the small body of existing studies looking at the effect of NSAIDs on post-vaccine antibody production in children, researchers did find some evidence of reduced antibody responses, but that did not affect the efficacy of vaccines. A 2009 paper examining the effect of OTC painkillers in mice and human cells in a lab found a similar result: Ibuprofen, but not acetaminophen, dampens antibody responses. But, of course, humans are not mice, and our cells may behave differently when they're actually inside of us rather than in a petri dish. "This is not an FDA-sanctioned clinical trial that shows [NSAIDs] have an effect on vaccinations, which is what you really need to do," says David Topham, a co-author of that study and a professor of microbiology and immunology at the University of Rochester. "I think if they did the trial, the effect would be modest." Still, based on his results, Topham says that if he had a choice, he'd take acetaminophen and not ibuprofen. And, if it makes you feel any better, Shresta told me she actually caved and took some ibuprofen the day after her second vaccine dose. "I had a major deadline the next day and with the headache, there was no way that was going to happen," she says. Life happens.
In defence of Canada’s unprecedented decision for a four-month vaccine interval
Saturday, April 3, 2021
Seniors across the country are rightly enraged that they were promised a 21-day interval between vaccine doses only to have that appointment pushed into July due to new recommendations by the National Advisory Committee on Immunization prescribing a four-month interval between doses. Seniors, after all, were the most vulnerable to COVID-19.
Throwing thousands of Canadian seniors into three extra months of vaccine limbo was never going to be popular, particularly when Canada is the only jurisdiction on Earth to use a four-month interval between doses.
That controversy is not going to abate anytime soon, but below find the best arguments as to why Canada's decision to stretch its booster shot gap could end up saving lives.
In a pandemic that has been defined by near-constant government missteps, this may be one of the few times where Canada has prioritized science over political expediency.
The 21-day gap between doses was always somewhat arbitrary
There is no spreadsheet at Pfizer headquarters saying that 21 days is the optimum, experiment-tested gap to maximize the effectiveness of their vaccine. In fact, it's a number largely pulled out of thin air.
"I know this sounds crazy, but it's arbitrary. If you look for literature that documents why that's the best time point, there isn't any," David Topham, a University of Rochester immunologist, said in a February interview.
In fact, the typical rule of thumb is that booster shots have the best potency when administered at least two months after the initial dose. Vaccinations for HPV, Hepatitis A and Hepatitis B, among others, all have their boosters at five months or more . Boosters shots for avian flu, meanwhile, have been found to be more effective at six months than if given only 28 days after the first shot, according to a 2009 study .
How long will the coronavirus vaccines protect you? Experts weigh in.
Monday, March 29, 2021
You may be among the more than 95 million people in the United States who have taken at least one dose of a coronavirus vaccine. Or you may still be awaiting your turn. Regardless, there's a crucial question on most of our minds: How long will the vaccine really protect us?
As with most aspects of the virus, the answer is not completely clear. Why? Because although we have been battling the pandemic for more than a year, the vaccines were granted emergency use authorization relatively recently. So experts have not had time to observe their long-term effectiveness.
However, that research is underway, and in the meantime, experts say we can make an educated guess.
Can we extrapolate from what we know about natural immunity?
In fact, much of this hypothesizing comes from extrapolating data examining immune responses in people who have had covid-19 and illnesses from other coronaviruses, rather than in people who have been vaccinated, said Dbeibo, who is director of vaccine initiatives for Indiana University.
"But vaccine responses should not be less reliable than in natural infection," she added.
Current research shows that people who have been infected with covid-19, the illness caused by the coronavirus, retained immunity that was robust after eight months. That gives researchers a starting point in predicting how long immunity may last after vaccination, Dbeibo explained.
But research also shows people who had more severe cases developed a stronger immune reaction than those with milder forms of the disease. And because vaccine-induced immunity appears to be more similar to natural immunity that is derived from severe covid-19 infections, researchers say they believe people who take a coronavirus vaccine will be protected better than most people with natural immunity, said David Topham, a professor of microbiology and immunology at the University of Rochester.
All of that said, antibodies will wane. And although it is a gradual process, once antibodies decline to a level that is no longer protective, reinfection is possible. Still, the infection is likely to be milder, experts said.
Topham, founding director of the Translational Immunology and Infectious Disease Institute at the University of Rochester, has been studying the coronavirus and the role of memory B cells — immune cells that persist for a lifetime and produce antibodies when re-exposed to a pathogen that they have been programmed to fight. He said some people who were hospitalized with severe covid-19 infections still had high frequencies of memory B cells, as well as antibodies, up to nine months after infection.
He said memory B cells can even adapt quickly to a new variant, usually within days.
Getting a COVID Vaccine? Try Tylenol but Skip the Advil for Mild Discomfort
Thursday, March 4, 2021
It's best to avoid some common pain relievers after the COVID shot, because they can dilute the power of the vaccine, according to research at the University of Rochester Medical Center.
Ibuprofen (Advil) and naproxen (Aleve) dampen the production of necessary antibodies that protect against illnesses such as COVID, scientists said.
Over-the-counter pain and fever-reducers that are classified as nonsteroidal anti-inflammatories (NSAIDs) are the ones to avoid. They act in part by blocking the cyclooxygenase-2 (cox-2) enzyme. But blocking the cox-2 enzyme is not a good idea in the context of vaccination, because the cox-2 enzyme is necessary for high production of B-lymphocytes. When people take medications like Advil for discomfort at the injection site they're also inadvertently reducing the ability of B cells to make antibodies that protect against COVID and other viruses.
"Unless your health care provider tells you otherwise, it's best not to take pain relievers one or two days before the coronavirus vaccine and for a week afterward," said David J. Topham, Ph.D., a professor in the Center for Vaccine Biology and Immunology at URMC. Abstaining from NSAIDs for 14 days afterward would be even better, he added.
Taking acetaminophen (Tylenol) is probably okay after the vaccine, Topham said, because it targets pain and fever in a different way.
Topham bases his opinion on peer-reviewed, published research he co-authored on this topic, including the first description of how NSAIDs and other pain relievers impact cox-2 and blunt the body's production of anti-viral antibodies.
The CDC also cautions against using pain relievers prior to vaccination for coronavirus — but states that individuals should talk to their doctors about using them afterward.
According to the CDC website: "If you have pain or discomfort, talk to your doctor about taking over-the-counter medicine, such as ibuprofen, aspirin, antihistamines, or acetaminophen, for any pain and discomfort you may experience after getting vaccinated. You can take these medications to relieve post-vaccination side effects if you have no other medical reasons that prevent you from taking these medications normally. It is not recommended you take these medicines before vaccination for the purpose of trying to prevent side effects, because it is not known how these medications may impact how well the vaccine works."
Individuals who take aspirin for cardiovascular or vascular disease should talk to their doctors before stopping even low-dose aspirin. And people who take medications such as Celebrex for arthritis or other chronic pain also should consult their physicians.
Medical Center faculty, Anthony Fauci to discuss coronaviruses on TV program
Thursday, February 4, 2021
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, will join David Topham, the Marie Curran Wilson and Joseph Chamberlain Wilson Professor in the Department of Microbiology and Immunology, and Lou Papa, a professor of clinical medicine, for a discussion of coronaviruses, a family of viruses that cause illnesses such as the common cold and COVID-19. Tune in to the health care program "Second Opinion" at 8 p.m. tonight, February 4, on WXXI-TV.
"Experience Rochester: Rochester's Quest to Beat COVID-19" from January 28, 2021
Tuesday, February 2, 2021
A number of faculty participated in an "Experience Rochester" webinar to discuss groundbreaking work on treating, tracking, and preventing COVID-19 from spreading. Topics discussed include latest information on vaccine distribution, vaccine trials for children, and research on new coronavirus variants. The session was moderated by Stephen Dewhurst, PhD, Professor and Chair of Microbiology and Immunology and Vice Dean for Research.
Participants Panel:
Mary Caserta, MD, Professor, Department of Pediatrics (Infectious Diseases)
David Topham, PhD, Marie Curran Wilson and Joseph Chamberlain Wilson Professor, Center for Vaccine Biology and Immunology
Ann Falsey, MD, Professor, Department of Medicine (Infectious Diseases)
Nana Bennett, MD, Professor, Department of Medicine and Public Health Sciences
Angela Branche, MD, Assistant Professor, Department of Medicine (Infectious Diseases)
Will the Covid-19 vaccine work on new strains of the virus?
Monday, January 25, 2021
Dave Topham was recently quoted on MSN.com as to whether the current COVID-19 vaccines will be effective for the variants.
As researchers become more aware of strains of SARS-CoV-2, the virus that causes Covid-19, public health officials have one question: Will vaccines offer protection against them?
At the moment, there are two broad kinds of mutations scientists are keeping an eye on: Some that make the virus more infectious, and others that appear to make it capable of evading antibodies generated by vaccines.
These new strains are somewhat expected—viruses mutate constantly. Their only evolutionary goal is to be a glorified genetic copy-and-paste machine; destroyed cells and illness are just collateral damage. It's understandable that sometimes, viruses make copying mistakes in their genetic code along the way—and sometimes, those bugs end up being perks for them instead.
"What's going on now is the virus is adapting to a new host," says David Topham, a microbiologist and immunologist at the University of Rochester in New York. SARS-CoV-2 didn't start out as a virus infectious to humans—it began as an animal virus. Now that it's had over a year of practice copying itself in people, it's not surprising that it's gotten better at replicating and surviving among us.
WXXI Live Forum addresses COVID-19 vaccines:
Thursday, December 17, 2020
 WXXI hosts a live, televised forum addressing COVID-19 vaccines.
WXXI hosts a live, televised forum addressing COVID-19 vaccines.
Local experts on the latest COVID-19 vaccine research gathered for a live forum on WXXI-TV and radio Thursday night. Those on the panel said they see reason for hope, but they also caution it will take some time for the community to build up immunity to the virus.
Researchers on the program from the University of Rochester Medical Center included Dr. Angela Branche, co--director of the URMC Vaccine Trials and Evaluation Unit. Branche talked about the challenge to get past the mistrust that some people in the Black community have regarding medical research, when the vaccine becomes more widely available.
"And we're really going to have to work with our community partners, leaders and activists in the community who already have established trust with these groups that we're talking about and partnering with them and giving them educational tools and helping them deliver the message is really what's going to be the most effective," Branche said.
Monroe County's commissioner of Public Health, Dr. Michael Mendoza also sees a need to provide as much outreach and education to the community as possible about the vaccine. And he emphasized the need to continue following guidelines about wearing masks and physical distancing, because it will take a while to get most people inoculated.
"We have a vaccine on the ground, there are people who are getting the vaccine now, but the reality is that there is still so much time between now and when this is over."
Mendoza said that right now he is concerned about the rapidly increasing numbers of people being hospitalized and how that could impact health care in general in the Finger Lakes region.
$18 Million Grant Supports HIV/AIDS Research, Outreach to Rochester Community
Wednesday, December 2, 2020
The University of Rochester Medical Center's Infectious Diseases Division has partnered with the National Institutes of Health (NIH) to study HIV/AIDS for more than 30 years. Their efforts have been recognized with a new $18 million grant to continue conducting vaccine and treatment trials and engaging with communities affected by HIV.
The award also allows Rochester researchers to study other high-priority infectious diseases, including COVID-19. The team (pictured above) pivoted to study coronavirus vaccines and treatments over the past six months, contributing to the worldwide effort to bring safe and effective vaccines and therapies to market as quickly as possible.
"While creating a preventive HIV vaccine has been complex, our work has established approaches that are used for HIV and also contributed to the unprecedented speed at which coronavirus vaccines are being developed," said Michael C. Keefer, M.D., professor in the department of Medicine and interim chief of the Infectious Diseases Division. For example, vaccines using adenoviruses as carriers of HIV proteins have been used in HIV research for years, and that is one of the approaches being used in two of the actively enrolling COVID-19 vaccine efficacy trials (AstraZeneca and Janssen trials). Additionally, anti-HIV monoclonal neutralizing antibodies have been shown to be effective in preventing some HIV infections, and anti-coronavirus monoclonal antibodies have become an important strategy to treat patients with early-stage COVID-19.
The division received its first grant and became a part of the National Institute of Allergy and Infectious Diseases (NIAID) HIV/AIDS Clinical Research Networks in 1987. Led by Keefer and Stephen Dewhurst, Ph.D., chair of the department of Microbiology and Immunology, as well as program director Catherine Bunce, the new grant will run for seven years and will be used to:
- Coordinate and execute high-quality HIV/AIDS vaccine and treatment trials that enroll participants from diverse populations.
- Engage with local communities to assess attitudes to clinical research and conduct educational outreach around research participation.
- Mentor and train the next generation of HIV/AIDS researchers.
- Advance HIV/AIDS research by providing scientific leadership and supporting NIAID's AIDS Clinical Trials Group (ACTG) and HIV Vaccine Trials Network (HVTN).
- Provide data and quality management, regulatory support, laboratory, pharmacy and other resources to effectively support HIV/AIDS clinical trials.
- Rapidly respond to emerging infectious diseases that require national attention and coordination.
Congratulations Dr. Kim
Friday, November 20, 2020
 Minsoo Kim, Ph.D was recognized recently with the Davey Award, an honor bestowed on University of Rochester/Wilmot Cancer Institute faculty members who have made outstanding contributions to cancer research.
Minsoo Kim, Ph.D was recognized recently with the Davey Award, an honor bestowed on University of Rochester/Wilmot Cancer Institute faculty members who have made outstanding contributions to cancer research.
New Institute Takes Aim at Infectious Pathogens, Builds Pipeline of Clinician Researchers
Wednesday, November 11, 2020
The University of Rochester Medical Center (URMC) has created a new institute that will strengthen and accelerate the study of viral, bacterial, and fungal pathogens. The Translational Immunology and Infectious Diseases Institute (TIIDI) will build upon decades of scientific research leadership in the human immune system, respiratory viruses, and vaccine development, and, more recently, the Medical Center's role in the national response to the coronavirus pandemic.
The new Institute -- which is led by David J. Topham, Ph.D., and was approved by the University of Rochester Board of Trustees last month -- will bring together multi-disciplinary teams of scientists and clinicians that will take the knowledge gained from basic research in immunology, microbiology, and virology and apply it to problems in human diseases caused by infectious pathogens. TIIDI will also strengthen education and training programs to build a pipeline that produces the next generation of adult and pediatric clinician-researchers in the field.
Researchers will study infectious diseases across several areas of medicine, including immunodeficiency, cancer, transplant, orthopedics, ophthalmology, dermatology, gastroenterology, autoimmunity, neonatology, and public health. TIIDI will focus on the development of new treatments and vaccines, and strategies to better control the spread of infectious diseases, such as HIV, respiratory pathogens, and hospital acquired infections. Researchers will also study the pathology of infectious diseases to better assess risk factors for severe outcomes and develop more effective early interventions.
Special Department of Microbiology and Immunology Seminar – Dr. Malika Grayson – November 9th at NOON
Friday, October 23, 2020
How do you make an impact when you are the only person in the room that looks like you? We hear the terms diversity and inclusion but forget that the term representation should be a reflection of diversity and inclusion combined. This isn't always the case. Dr. Grayson discusses her views on what it means to increase diversity and representation as an Individual Contributor. Learn more about her journey as the 2nd Black Woman to graduate with a PhD in Mechanical Engineering from her graduate institution. Hear about her current work as both an engineer, a STEM Advocate, and her most recent success as author of 'HOODED: A Black Girl's Guide to the PhD' where she highlights her time and lessons learned during her PhD Program.
Can the Common Cold Help Protect You from COVID-19?
Thursday, October 1, 2020
Seasonal colds are by all accounts no fun, but new research suggests the colds you've had in the past may provide some protection from COVID-19. The study, authored by infectious disease experts at the University of Rochester Medical Center, also suggests that immunity to COVID-19 is likely to last a long time -- maybe even a lifetime.
The study, published in mBio, is the first to show that the COVID-19-causing virus, SARS-CoV-2, induces memory B cells, long-lived immune cells that detect pathogens, create antibodies to destroy them and remember them for the future. The next time that pathogen tries to enter the body, those memory B cells can hop into action even faster to clear the infection before it starts.
Because memory B cells can survive for decades, they could protect COVID-19 survivors from subsequent infections for a long time, but further research will have to bear that out.
The study is also the first to report cross-reactivity of memory B cells -- meaning B cells that once attacked cold-causing coronaviruses appeared to also recognize SARS-CoV-2. Study authors believe this could mean that anyone who has been infected by a common coronavirus -- which is nearly everyone -- may have some degree of pre-existing immunity to COVID-19.
"When we looked at blood samples from people who were recovering from COVID-19, it looked like many of them had a pre-existing pool of memory B cells that could recognize SARS-CoV-2 and rapidly produce antibodies that could attack it," said lead study author Mark Sangster, Ph.D., a research professor of Microbiology and Immunology at URMC.
Sangster's findings are based on a comparison of blood samples from 26 people who were recovering from mild to moderate COVID-19 and 21 healthy donors whose samples were collected six to 10 years ago -- long before they could have been exposed to COVID-19. From those samples, study authors measured levels of memory B cells and antibodies that target specific parts of the Spike protein, which exists in all coronaviruses and is crucial for helping the viruses infect cells.
Cells Sacrifice Themselves to Boost Immune Response to Viruses
Wednesday, September 30, 2020
Whether flu or coronavirus, it can take several days for the body to ramp up an effective response to a viral infection. New research appearing in the journal Nature Immunology describes how different cells in the immune system work together, communicate, and -- in the case of cells called neutrophils -- bring about their own death to help fight off infections. The findings could have important implications for the development of vaccines and anti-viral therapies.
"The immune system consists of several different types of cells, all acting in coordination," said Minsoo Kim, Ph.D., a professor of Microbiology and Immunology at the University of Rochester Medical Center (URMC) and senior author of the study. "These findings show that cells called neutrophils play an important altruistic role that benefits other immune cells by providing key resources for their survival and, in the process, enhancing the body's immune response against a virus."
Neutrophils are a key component of the innate immune system, the part of the body's defenses that is always switched on and alert for bacterial and viral invaders. The vast majority of white cells circulating in blood are neutrophils and, as a result, these cells are the first on the scene to respond to an infection.
However, neutrophils are not fully equipped to eliminate a viral threat by themselves. Instead, when the respiratory tract is infected with a virus like influenza or COVID-19, a large number of neutrophils rush to the infection site and release chemical signals. This triggers the production of specialized T cells, which are part of the body's adaptive immune system, which is activated to produce a more direct response to specific infections. Once mobilized in sufficient quantities, a process that typically takes several days, these T cells target and ultimately destroy the infected cells.
The new study, which was conducted in mice infected with the flu virus, shows that in addition to jump-starting the adaptive immune response, neutrophils have one more important mission that requires that they sacrifice themselves. As T cells arrive at the infection site, the neutrophils initiate a process called apoptosis, or controlled death, which releases large quantities of a molecule called epidermal growth factor (EGF). EGF provides T cells with the extra boost in energy necessary to finish the job.
"This study represents an important paradigm shift and shows that the adaptive immune system doesn't generate a successful response without instruction and help from the innate immune system," said Kim. "The findings reveal, for the first time, how different immune cells work together, and even sacrifice themselves, to accomplish the same goal of protecting the host from the viral infection."
RNA Essay Contest Results and Congratulations
Wednesday, August 26, 2020
The UR Center for RNA Biology offered an exercise during the time when COVID-19 became a sufficient threat to largely shut-down our research enterprise. We're pleased to announce the winners of the UR's Center for RNA Biology Essay Contest on "The role of RNA research in community health". These awards are sponsored by a grant from the RNA Society & Lexogen to the UR Center for RNA Biology, and funds from UR RNA Structure & Function Cluster.
Our Gold prize (~$1,000 value) award goes to Sydney Simpson, an Immunology, Microbiology & Virology graduate student in Steve Dewhurst's lab in the Department of Microbiology & Immunology, for her essay: "Nucleoside Analog Inhibitors: Timeless & Timely Beacons of Hope".
The Silver Prize (~$250 value) award goes to Omar Hedaya, a Biochemistry & Molecular Biology graduate student in Peng Yao's lab in the Department of Medicine/Department of Biochemistry & Biophysics, for his essay: "Know the Fundamentals when Seeking the Future".

Omar Hedaya

Sydney Simpson
Both awardees have become members of the RNA Society and will use their winnings toward technology needs for the upcoming semester.
The RNA Society now features our contest results, including the winning essays, in its latest RNA Salon update: https://www.rnasociety.org/featured-salons, see bullet #3.
We would like to acknowledge Honorable Mentions for the following applicants:
- Sai Shashank Chavali -- Graduate student; Biophysics, Structural & Computational Biology; Wedekind Lab
- Lily Cisco -- Graduate student; Cellular and Molecular Pharmacology & Physiology; Lueck Lab
- Gabrielle Kosoy -- Graduate student; Biophysics, Structural & Computational Biology; Miller Lab
- Ashwin Kumar -- Graduate student; Biophysics, Structural & Computational Biology; Topham Lab
- Li Xie -- Graduate student; Genetics, Development & Stem Cells; Pröschel Lab
We thank all who participated -- and our judges, too!
Thakar Lab publishes article on the Impact of vaccine type on HIV-1 vaccine elicited antibody durability and B cell gene signature
Monday, August 10, 2020
Abstract
Efficacious HIV-1 vaccination requires elicitation of long-lived antibody responses. However, our understanding of how different vaccine types elicit durable antibody responses is lacking. To assess the impact of vaccine type on antibody responses, we measured IgG isotypes against four consensus HIV antigens from 2 weeks to 10 years post HIV-1 vaccination and used mixed effects models to estimate half-life of responses in four human clinical trials. Compared to protein-boosted regimens, half-lives of gp120-specific antibodies were longer but peak magnitudes were lower in Modified Vaccinia Ankara (MVA)-boosted regimens. Furthermore, gp120-specific B cell transcriptomics from MVA-boosted and protein-boosted vaccines revealed a distinct signature at a peak (2 weeks after last vaccination) including CD19, CD40, and FCRL2-5 activation along with increased B cell receptor signaling. Additional analysis revealed contributions of RIG-I-like receptor pathway and genes such as SMAD5 and IL-32 to antibody durability. Thus, this study provides novel insights into vaccine induced antibody durability and B-cell receptor signaling.
Sangster Weighs in on COVID-19 Vaccine, Antibodies
Wednesday, August 5, 2020
With millions of lives on the line, researchers have been working at an unprecedented pace to develop a COVID-19 vaccine.
But that speed—and some widely touted breakthroughs—belie the enormous complexity and potential risks involved. Researchers have an incomplete understanding of the coronavirus and are using technology that's largely unproven.
Among many worries: A handful of studies on COVID-19 survivors suggest that antibodies—key immune system proteins that fight infection—begin to disappear within months. That's led scientists to worry that the protection provided by vaccines could fade quickly as well. Some even question whether vaccines will really end the pandemic. If vaccines produce limited protection against infection, experts note, people will need to continue wearing masks and social distancing even after vaccines roll out.
People with severe symptoms from COVID-19 tend to have higher antibody levels than those with milder cases.
Some people fail to generate antibodies because they have compromised immune systems, said Mark Sangster, a research professor at the University of Rochester Medical Center.
Even when people do generate antibodies against the novel coronavirus, studies suggest the antibodies may not last long.
IMV Grad Student Megan Ulbrich Wins the Melville A. Hare Award for Excellence in Teaching
Friday, June 5, 2020
Please join the department in congratulating Megan Ulbrich, this year's winner of the Melville A. Hare Award for Excellence in Teaching. This award is given annually by the Department of Microbiology and Immunology to a graduate student who has displayed outstanding qualities of mentoring and teaching in one or more MBI courses. The award will be officially conferred at URMC Convocation in the late summer.
Megan is currently an IMV graduate student in Michelle Dziejman's lab working on multidisciplinary approaches to uncover novel effector protein functions. Megan grew up in Buffalo, NY and received her B.S. in Microbiology from the University of Pittsburgh.
Dr. Deborah Fowell appointed next Chair of the Department of Microbiology and Immunology
Tuesday, May 12, 2020
 We congratulate Dr. Deborah Fowell on her new position as Chair of the Department of Microbiology and Immunology at Cornell University College of Veterinary Medicine, where she will start this fall. As a valued faculty member and Dean's Professor in the Department of Microbiology and Immunology, Center for Vaccine Biology and Immunology, this is a bittersweet goodbye -- but a wonderful new opportunity for Deb!
We congratulate Dr. Deborah Fowell on her new position as Chair of the Department of Microbiology and Immunology at Cornell University College of Veterinary Medicine, where she will start this fall. As a valued faculty member and Dean's Professor in the Department of Microbiology and Immunology, Center for Vaccine Biology and Immunology, this is a bittersweet goodbye -- but a wonderful new opportunity for Deb!
Since joining UR in April 2000, Dr. Fowell has achieved great success in her research program. One example is her organization and leadership of an accomplished, interdisciplinary team of investigators in an NIH funded program project to visualize the immune system in action, which was recently renewed. Her efforts have led to tremendous advancements in the field and will continue to enhance our understanding of tissue inflammation and immune responses, while also providing new therapeutic targets for mitigation of a wide variety of inflammatory diseases.
Not only is Dr. Fowell an outstanding researcher, she is also deeply committed to excellence in graduate education. She recently took over as Program Director and successfully renewed the University's Predoctoral Training Grant in Immunology. In addition, she has received multiple mentoring awards at the UR, including the 2015 Graduate Alumni Award, which is the most prestigious student teaching and mentoring award here at the School of Medicine and Dentistry.
Dr. Fowell has a strong reputation among her peers as a highly innovative researcher with rigorous intellectual standards, and as a consistent advocate of the highest quality science and long-term success for academic research. Her collaborative approach to research, commitment to enriching learning and intellectual discourse, coupled with her drive and energy, will ensure her success at Cornell.
It's with pride, warm wishes and anticipation of exciting future collaborations that we wish Deb every success in her new role!
Cassandra Houser Receives AAI Young Investigator Award at the 22nd Annual Upstate New York Immunology Conference
Wednesday, November 6, 2019
IMV Ph.D. Graduate Student, Cassandra Houser (Paige Lawrence's Lab) received one of ten AAI Young Investigator Awards at the 22nd Annual Upstate New York Immunology Conference. The conference this year took place October 28th at Albany Medical Center. The award is based on the top poster abstracts submitted based on scientific rigor and significance to the field of immunology.
Congrats Cassandra!
URMC-099 Combats Surgery-Induced Delirium, Cognitive Dysfunction in Preclinical Model of Orthopedic Surgery
Wednesday, November 6, 2019
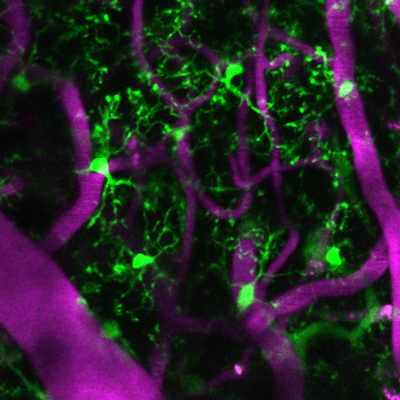
Living microglia, genetically marked to glow green, in the cerebral cortex with magenta colored blood vessels from a mouse treated with URMC-099.
A new study published in the Journal of Neuroinflammation found that prophylactic treatment with URMC-099 -- a "broad spectrum" mixed-lineage kinase 3 inhibitor -- prevents neuroinflammation-associated cognitive impairment in a mouse model of orthopedic surgery-induced perioperative neurocognitive disorders (PND).
PND, a new term that encompasses postoperative delirium, delayed neurocognitive recovery, and postoperative neurocognitive disorder, is the most common complication after routine surgical procedures, particularly in the elderly. Following surgery, such as hip replacement or fracture repair, up to 50 percent of patients experience cognitive disturbances like anxiety, irritability, hallucinations, or panic attacks, which can lead to more serious complications down the line. Currently, there are no FDA-approved therapies to treat it.
Developed in the laboratory of Harris A. "Handy" Gelbard, M.D., Ph.D., director of the Center for Neurotherapeutics Discovery at the University of Rochester Medical Center, URMC-099 inhibits damaging innate immune responses that lead to inflammation in the brain and accompanying cognitive problems. Using animal models of diseases like HIV-1-associated neurocognitive disorders, Alzheimer's disease and multiple sclerosis, Gelbard has shown that the compound blocks enzymes called kinases (such as mixed lineage kinase type 3, or MLK3) that respond to inflammatory stressors inside and outside cells.
Gelbard and Niccolò Terrando, Ph.D., director of the Neuroinflammation and Cognitive Outcomes laboratory in the Department of Anesthesiology at Duke University Medical Center, used an orthopedic surgery mouse model that recapitulates features of clinical procedures such as a fracture repair or hip replacement, which are often associated with PND in frail subjects. In a pilot experiment, they treated one group of these mice with URMC-099 before and after surgery, and another group prior to surgery only. Gelbard and Terrando's teams, including first author Patrick Miller-Rhodes, a senior pre-doctoral student in the Neuroscience Graduate Program working in the Gelbard lab at URMC, measured the following:
- How the surgery affected the central nervous system and the immune cells (microglia) that reside there was evaluated using stereology and microscopy.
- Surgery-induced memory impairment was assessed using the "What-Where-When" and Memory Load Object Discrimination tasks.
- The acute peripheral immune response to surgery was assessed by cytokine/chemokine profiling and flow cytometry.
- Long-term fracture healing was assessed in fracture callouses using micro-computerized tomography and histomorphometry analyses.
- For additional details see the "Materials and Methods" section of the study
The team found that the surgery disrupted the blood brain barrier and activated microglia (a first line immune responder present in the inflamed brain), which led to impaired object place and identity discrimination when the mice were subject to the "What-Where-When" and Memory Load Object Discrimination tasks. Both URMC-099 dosing methods prevented the surgery-induced microgliosis (increase in the number of activated microglia) and cognitive impairment without affecting fracture healing.
"A major concern regarding the use of anti-inflammatory drugs for PND is whether they will affect fracture healing. We found that our preventive, time-limited treatment with URMC-099 didn't influence bone healing or long-term bone repair," said Gelbard and Terrando, professor of Neurology, Neuroscience, Microbiology and Immunology, and Pediatrics at URMC and associate professor of Anesthesiology at Duke University Medical Center, respectively. "These findings of improvement in cognition and normal fracture healing provide compelling evidence for the advancement of URMC-099 as a therapeutic option for PND."
"Right now we have nothing to treat this condition," said Mark A. Oldham, M.D., assistant professor in the department of Psychiatry at URMC who treats patients with PND. "We work hard to provide good medical care, including helping people sleep at night and making sure they are walking, eating and drinking, but it isn't clear that these efforts have any meaningful long-term impact."
According to Oldham, recent studies that track patients following an episode of PND show that many of them don't resolve completely, and that they have a new cognitive baseline after delirium.
"It is increasingly an accepted fact that after delirium, people have suffered some kind of neurological insult, which leaves them cognitively or functionally worse off than before the incident," he noted.
Next steps for the research include identifying definitive mechanisms for pain modulation, immune cell trafficking and neuro-immune characterization in PND. Gelbard and Terrando are tackling some of these questions with funds from the National Institutes of Health (RO1 AG057525). The current study was also funded by multiple grants from the NIH (P01MH64570, RO1 MH104147, RO1 AG057525 and F31 MH113504). The University of Rochester has four issued U.S. patents and multiple issued patents in foreign countries covering URMC-099.
Rochester Researchers Receive $12 Million Award to Visualize the Immune System in Action
Wednesday, October 16, 2019
NIH grant will help identify new strategies to enhance immune response
 Cutting-edge imaging technologies that allow scientists to watch the immune system work in real time are leading to a greater understanding of how we combat infection and disease. With a new $12 million grant, researchers will use this knowledge to explore strategies to better fight infections like the flu and beat back overactive immune responses in disorders like rheumatoid arthritis and lupus.
Cutting-edge imaging technologies that allow scientists to watch the immune system work in real time are leading to a greater understanding of how we combat infection and disease. With a new $12 million grant, researchers will use this knowledge to explore strategies to better fight infections like the flu and beat back overactive immune responses in disorders like rheumatoid arthritis and lupus.
The five-year project, led by Deborah J. Fowell, Ph.D., Dean's Professor in the department of Microbiology and Immunology at the University of Rochester Medical Center, builds on a $9 million grant that her team received in 2014. Both program project grants were awarded by the National Institute of Allergy and Infectious Diseases at the National Institutes of Health.
"When we take cells out of their natural environment and study them in a dish we're missing out on a lot of biology," said David J. Topham, Ph.D., a study project leader and professor of Microbiology and Immunology at URMC. "Imaging and tracking live cells is an emerging theme in immunology and one that I think is going to move forward meaningful discoveries in the field."
In addition to Fowell and Topham, project leaders include Minsoo Kim, Ph.D.,James F. Miller, Ph.D., and Patrick Oakes, Ph.D. Scientists from Cornell and Loyola University will collaborate with the team, as well.
Read More: Rochester Researchers Receive $12 Million Award to Visualize the Immune System in ActionInnovative Pancreatic Cancer Treatment May Rev up Immune System
Tuesday, October 8, 2019
New Wilmot Cancer Institute research shows that combining a type of radiation therapy with immunotherapy not only cures pancreatic cancer in mice, but appears to reprogram the immune system to create an "immune memory" in the same way that a vaccine keeps the flu away. The combination treatment also destroyed pancreatic cells that had spread to the liver, a common site for metastatic disease.
The result is that the combination treatment also destroyed pancreatic cells that had spread to the liver, a common site for metastatic disease. The journal Cell Reports published the study, with Scott Gerber, Ph.D., as corresponding author, and Bradley Mills, Ph.D., a post-doctoral fellow in the Gerber Lab, as first author. They led a large team of collaborators from the University of Rochester departments of Surgery, Microbiology and Immunology, Pathology and Laboratory Medicine, Radiation Oncology, and Environmental Medicine.
Deb Fowell Authors Study on Immune Cell Navigation Systems
Tuesday, August 13, 2019
When immune cells get recruited to infections, tumors, or other sites of inflammation they exit the blood stream and begin searching for the damage. But how they effectively traverse the body's tissue and home in on their targets is unclear. A new study led by Deborah Fowell, Ph.D. suggests that T cells have distinct navigation systems that help them pinpoint their targets.
Fowell's research team, based in the David H. Smith Center for Vaccine Biology and Immunology in the Department of Microbiology and Immunology made the discovery by visualizing the immune system in real time using intravital multiphoton microscopy. The technology allows you to look directly into the skin and observe the dynamic behavior of immune cells 'live.' Their findings were published earlier this month in the journal Immunity.
"We thought that locating the infection foci was a passive event for immune cells; that they used the tissue as a scaffold to weave their way through this complex matrix to get to their target," said Fowell, Dean's professor in the Department of Microbiology and Immunology. "We discovered that they are pre-programmed to respond to certain cues within the tissue microenvironment that help them find their targets more efficiently."
The team hopes that discovering these specialized programs for migration in tissues will provide new therapeutic targets that enable manipulation of the immune response in a disease-specific or tissue-specific fashion, rather than globally suppressing the immune system. Possibilities include boosting protective immunity in diseases where the immune system is inefficient, such as chronic infections and tumors, and limiting immunity in diseases that are exacerbated by the immune system, like autoimmunity and heart disease.
Hen Prizant, Ph.D., a postdoctoral fellow in Fowell's lab and Alison Gaylo-Moynihan, M.D., Ph.D., a former student in the lab are co-first authors. Graduate students Ninoshka R.J. Fernandes, Hannah Bell, Dillon C. Schrock, Tara Capece, Brandon Walling, and Christopher Anderson contributed to the study. Faculty members David Topham, Minsoo Kim, Alan Smrcka and James Miller are also authors.
Fowell credits the new finding to the power of NIH Program Project Grants (P01), which allow faculty, trainees and students to explore uncharted scientific territory and branch out among different disciplines. For example, the team reached across Elmwood Avenue to have conversations with astrophysicists and engineers on River Campus about how objects move through and are found in space. The P01 that funded the research was awarded to Fowell (PI) and Kim, Topham and Miller in 2014.
Department of Microbiology Retreat Coming Up!
Monday, July 22, 2019
After an extended hiatus, the Microbiology & Immunology department retreat will return on Monday, August 26, 2019 at the Memorial Art Gallery, located at 500 University Avenue.
Some agenda highlights include:
- We are hosting three "gurus", one for each branch of the department. They will be giving individual seminars, as well as participate together in an audience guided panel discussion.
- Dr. John Harty, University of Iowa (Immunology)
- Dr. Jacques Ravel, University of Maryland School of Medicine (Microbiology)
- Dr. Luis Schang, Cornell University College of Veterinary Medicine (Virology)
- Lunch will include trivia hosted by Geeks Who Drink! One round will be questions pulled from poster abstracts -- hope you paid attention!
- The day will conclude with a happy hour to celebrate the achievements of the past year in the department.
If you have any questions, please see Jennifer Brennan in the Microbiology Administration Office.
Barbara H. Iglewski Receives The Eastman Medal at 2019 Commencement
Wednesday, May 22, 2019
Barbara H. Iglewski, professor and chair emerita in the Department of Microbiology and Immunology at the School of Medicine and Dentistry, has contributed landmark research on how bacteria cause infections. Her laboratory was the first to discover that bacteria use a communication system to coordinate attacks on human cells and initiate disease, and her work launched an entire field of study into how the system works in many types of bacteria. Several drugs that interrupt the bacterial communication process, thereby preventing infections, have been developed based on her work.
Barbara Iglewski is a trailblazer who paved the way for many other female scientists and leaders in Rochester and across the country.
Iglewski was the first female department chair at the School of Medicine and Dentistry and a trailblazer who paved the way for many other female scientists and leaders in Rochester and across the country. She pursued a career in science after accompanying her father, a country physician, on house calls. She received her Ph.D. in microbiology from Penn State University, and was recruited to the University of Rochester in 1986 to serve as chair of the Department of Microbiology and Immunology.
She holds seven patents, has published more than 180 papers and book chapters, and has received many awards and honors, including from the National Institutes of Health and the American Society for Microbiology. In 2015, she was inducted into the National Women's Hall of Fame, and has been recognized with the School of Medicine and Dentistry's Lifetime Mentoring Award (2009), the Susan B. Anthony Lifetime Achievement Award (2001), and the Arthur Kornberg Research Award (1999). She served as president of the American Society for Microbiology (ASM) from 1987 to 1988, and chaired its publications board from 1990 to 1999.
URMC and MBI awards for Excellence in Teaching and Research Winners
Tuesday, May 14, 2019
Please join our department in congratulating this year's winners of the URMC and MBI awards for Excellence in Teaching and Research.
- Maureen Banach - Co-recipient, Melville A. Hare Award for Excellence in Graduate Research
- Maxime Jean - Co-recipient, Melville A. Hare Award for Excellence in Graduate Research
- Zanah Francis - Melville A. Hare Award for Excellence in Graduate Teaching
BMB Graduates Receive College Prizes
Thursday, May 2, 2019
2019 College Prize Recipients
- Katherine Woo: Ayman Amin-Salem Memorial Prize
- Fayth Kim: the Janet Howell Clark Prize
- Nicholas Lim: Irene Bush Steinbock Award
- Kavya Bana: Helen S. Jones Memorial Fund
Research Roundup: Stephen Dewhurst Explores the Latest Bench-to-Bedside Projects
Monday, April 8, 2019
Transitions and Trials
Stephen Dewhurst, Ph.D., Vice Dean for Research
Almost 10 years ago, Brad Berk had the idea that the Medical Center should position itself to take a lead in the new field of cell-based therapies by constructing a manufacturing facility that could produce those cells under the highly regulated conditions that are required by the FDA. Brad's vision was that, by doing this, we would enable UR to deliver first-in-human therapies to patients.
Fast forward, and the facility we built -- the Upstate Stem Cell cGMP Facility (USCGF) -- is working in coordination with Torque Therapeutics (Cambridge, MA) to produce modified T cells that are being infused into cancer patients as part of a clinical trial that started earlier this month.
As with most research partnerships, our relationship with Torque is fundamentally a relationship between people, and an expression of trust in the team led by USCGF Director Luisa Caetano-Davies. It's worth noting that only two years ago, Luisa was a postdoctoral fellow in Chris Proschel's lab. Her subsequent success and growth are the combined result of a lot of hard work, intelligence and -- in no small measure -- opportunities created by our URBEST program.
The Torque trial is a huge step for the USCGF because it represents the first time that a cell-based product produced by our facility has been administered to human subjects. But it's also an important step for our Medical Center, when viewed in the broader context of our evolving approach to clinical trials.
Pat Ames is heading up a new Office of Clinical Research, working with Martin Zand, Steven Wormsley and many others to lead the implementation of a clinical trial management system to improve our clinical trials infrastructure. This system will streamline and automate many cumbersome clinical research processes and reduce administrative burden on our research teams, helping us conduct more clinical trials and offer more treatments to our patients and community members.
At the same time, Paul Barr in the Wilmot Cancer Institute (WCI) was just awarded a major new grant to support WCI involvement in National Cancer Institute (NCI) cooperative group clinical trials. This award establishes URMC as one of 30 lead academic sites within the NCI consortium, a designation rarely given to an institution that (currently) does not have an NCI-designated cancer center.
Perhaps most exciting of all, Mark Noble and Nimish Mohile recently received a highly encouraging score for a proposal that would (if funded, as we hope it will be!) launch a first-in-human trial of a new cancer treatment that is the result of fundamental research conducted in the Noble laboratory. Based on a new tumor-specific vulnerability, and discovery of existing drugs with the unexpected property of attacking this vulnerability, the new therapy eliminates cancer stem cells in glioblastoma (one of the most deadly human cancers).
This is exactly the kind of bench-to-bedside science that Brad envisaged ten years ago. We've made lots of progress, and there's more to come. It's an exciting time to be involved in research at the Medical Center.
These less common proteins may help fend off the flu
Tuesday, March 12, 2019
Influenza type B, though generally less widespread than type A, poses a formidable threat for vulnerable populations like the elderly and the young. In the 2012-2013 flu season, for example, influenza type B caused the majority of deaths due to flu among children, according to data from the Centers for Disease Control and Prevention. Findings published this week in mBio, ASM's open access journal, suggest that an efficient way to boost the efficacy of vaccines against influenza type B might be hiding in plain sight.
The researchers report that neuraminidase (NA), a protein found in small amounts in current vaccines, prompts the immune system to produce antibodies that may mount a broad protective response against influenza B viruses. Previous studies have connected NA antibodies to protection against the flu -- likely by preventing the spread of infection -- but this new study is among the first to show how that mechanism might be exploited for future, broad-acting flu vaccines.
"Targeting this type of vaccine response can help us develop a universal vaccine," said influenza virologist Luis Martinez-Sobrido, PhD, at the University of Rochester in New York. He co-led the study with immunologist James Kobie, PhD, also at Rochester.
Research Roundup: Dealing with Failure and an Unfunded Grant Application
Wednesday, January 9, 2019
Stephen Dewhurst, Ph.D., Vice Dean for Research
It's something we rarely talk about: how it feels when a grant application isn't funded. And yet, it's by far the most common outcome for any such submission -- an unavoidable consequence of paylines that are in the low teens or single digits.
The months between the submission of a grant and its review pass surprisingly quickly. And then time slows to a crawl. The self-doubt and self-criticism become more insistent. And hope flickers -- such a fragile thing, in the end.
Recently, after submitting a grant application, I found myself logging onto the NIH website every day after the review panel had met, to see if the scores had been posted. Eventually, they appeared.
This particular grant isn't going to be funded.
It's a horrible feeling. A private hurt that's immeasurably hard to share with colleagues, family and friends. That's because the narrative is one of failure.
But, I've chosen to write about it anyway -- because we've all been here. Because shame thrives in secrecy and loses its power when we talk about it (something I learned from Brené Brown).
What has helped is input from friends. One wrote: "Thank you for sharing this. I'm glad you did. As Directors etc., we don't share enough of the worries, the worthiness/unworthiness and the vulnerabilities that things like grants.... bring to the work and to our sense of ourselves as 'good' researchers, colleagues, leaders and people."
She went on to say: "I wish I had great advice. I have nothing. Except that you are a good person, a good mentor.... and whatever happens, you will still be those things. If you receive the grant, you know what your work will be; if you don't, you will have new and different work to do."
She's right.
It's also true that a life in science requires resilience -- the ability to pick oneself up after a fall and to learn and improve from failure. No one ever said that it would be easy.
In a few weeks, the summary statement will be released and I'll start thinking (with my colleagues) about ways to address the reviewers' concerns. Until then, I'll keep a space in my heart for these words of Samuel Beckett: "I can't go on. I'll go on."