Clinical Trials Coordination Center (CTCC)

Specializing in the development, management, and conduct of clinical research studies
CTCC provides a full range of research and clinical trial management support services that facilitate the conduct of clinical research from study concept through data analysis, publication, and FDA approval. Over the past 30 years, the CTCC has managed the conduct of more than 135 clinical research studies with 50 sponsors (government, industry, & private) that enrolled over 40,000 research participants in the US, Canada, Europe, New Zealand and Australia.
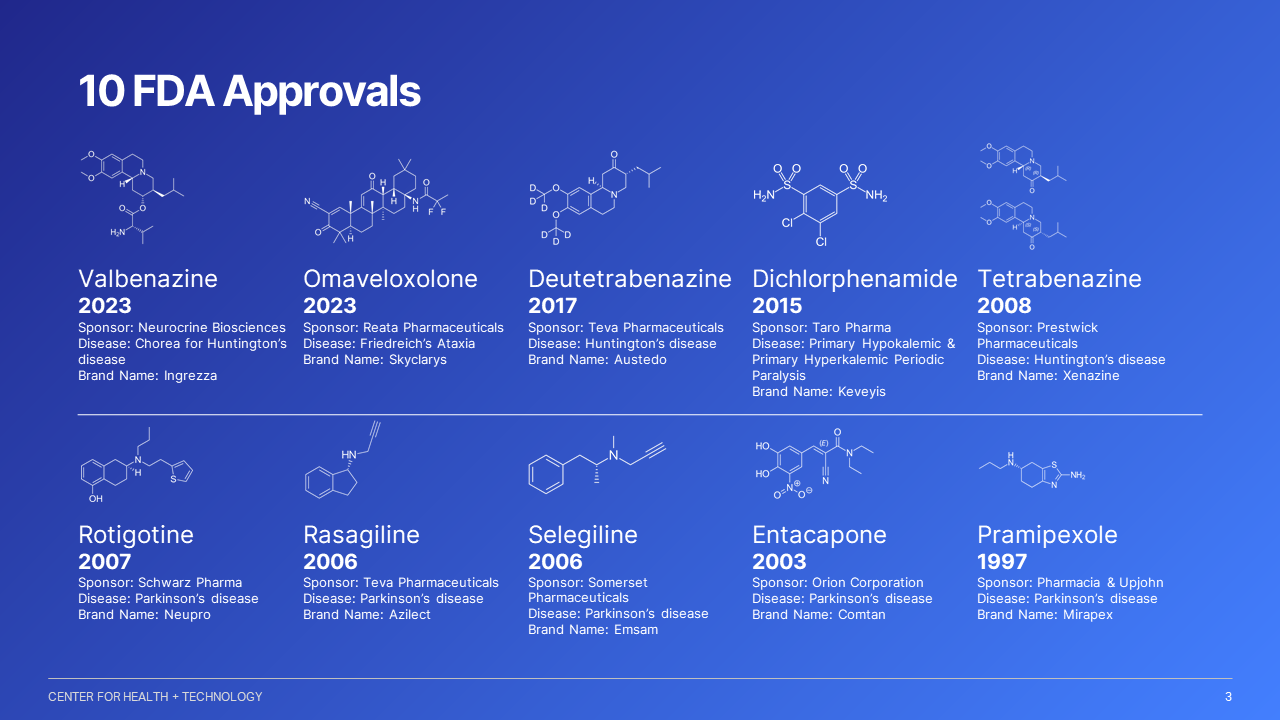
The CTCC has completed over 25 IND submissions and participated in the development of ten FDA approved compounds. The CTCC houses the largest repository of clinical trial data in the world for Parkinson disease and Huntington disease.
Our clinical research expertise includes:
- Study Startup (including but not limited to the following): Novel and adaptive trial design; Protocol development and training; Contract facilitation and negotiation; Site selection based on key performance indications and research study datasets
- Monitoring: Remote, risk-based quality management, and on-site
- Data Management: Clinical Data Management System (21 CFR part 11 compliant); Data sharing, Visualization, and Data standards (CDISC, STDM, CDASH, CDE)
- Clinical Trial Rescue and Recovery: Provide services to revamp, refocus, and revitalize your clinical trial
- Investigational New Drug/Investigational Device Exemption support
- Statistical analysis, modeling, and data mining
CTCC has the infrastructure to conduct worldwide, high quality, regulatory compliant multi-center clinical research:
- 200+ credentialed investigators and coordinators
- Direct web-based data entry and ePRO
- Access to 100+ research study datasets
- Data visualization tools and templates
- Clinical Trial Management Systems (21 CFR part 11 compliant)
- 60+ SOPs and guidelines for audit readiness
CTCC achieves study start-up, enrollment, database lock and regulatory submission at an accelerated pace due to long-standing-relationships with clinical sites, competitive site start-up, disease specific expertise and many other unique experiences that will benefit your trial.