Research Education News
20242023202220212020
Rahman Article Chosen as One of URMC's Top 10 News Stories of 2016
Saturday, December 31, 2016
Irfan Rahman's study, published in Oncotarget in November, has been chosen by the URMC as one of the top news stories of 2016.
The study is the first-ever showing that E-cigarettes cause damage to gum tissue. Rahman's research suggests that electronic cigarettes are as equally damaging to gums and teeth as conventional cigarettes.
GDSC Student to join the Steven’s Laboratory at Harvard Medical
Tuesday, December 27, 2016

Nicole Scott-Hewett
Nicole Scott-Hewett, a recent graduate of the GDSC program will be joining Beth Steven's laboratory at the Boston Children's Hospital F.M. Kirby Neurobiology Center. There Nicole will be involved in projects related to understanding mechanisms of complement and microglia-mediated pruning in development and in disease models. With her paper in this month's issue of PLoS Biology on lysosomal dysfunction, Nicole leaves us with a fanfare. We wish her all the best for her new beginnings in Boston!
Repurposed drugs may offer improved treatments for fatal genetic disorders
Wednesday, December 21, 2016
Department of Biomedical Genetics researchers believe they have identified a new means of treating some of the most severe genetic diseases of childhood, according to a new study in PLOS Biology. The diseases, called lysosomal storage disorders (LSDs), are caused by disruptions in the functioning of the stomach of the cell, known as the lysosome. LSDs include Krabbe disease, Gaucher disease , metachromatic leukodystrophy and about 40 related conditions. In their most aggressive forms, they cause death of affected children within a few years after birth.

Nicole Scott-Hewett

Christopher Folts
The research was spear-headed by Nicole Scott-Hewett and Chris Folts, two recent graduates of the program in Genetics, Development and Stem Cells. Led by the article's corresponding author Mark Noble, Ph.D., the team discovered for the first time how specific toxic waste products that accumulate in LSDs cause multiple dysfunctions in affected cells. They also found that several drugs already approved for other uses have the unexpected ability of overcoming the cellular toxic build-up, providing new opportunities for treatment. Key to this discovery was the finding that these drugs can help restore normal acidification of the lysosome.
In a mouse model of Krabbe disease (one of the most severe LSDs), Drs. Folts and Scott-Hewett found that their lead study drug, colforsin, increased survival as effectively as in studies where disease-causing mutations were corrected by gene therapy. Colforsin is approved in Japan to treat cardiac disease, which provides information to investigators about its use in humans.
Increased survival in mice occurred even though treatment was started later than is necessary for gene therapy. The research treatment also decreased damage to the brain and improved the quality of life in the diseased mice. All of these outcomes are critical goals in the treatment of children with Krabbe disease or related illnesses, said Noble, who is the Martha M. Freeman, M.D., Professor in Biomedical Genetics at URMC.
"One of the great challenges in these diseases is that they are both rare and come in many different varieties, and advances have tended to focus on single diseases," Noble said. "In contrast, our findings suggest our treatments will be relevant to multiple disorders. Also, we saw benefits of our treatment even without needing to correct the underlying genetic defects. That gives us great hope that we could combine our treatments with other candidate approaches to gain additional benefits."
If the results can be translated into humans, Noble said, the repurposed drugs might improve the quality of life for afflicted children while more difficult experimental genetic treatments are pursued. The complete study can be found at: PLoS Biology
Meng Wang, a former graduate student in the laboratory of Dr. Bohmann, has been named a Howard Hughes Medical Institute (HHMI) Faculty Scholar
Tuesday, December 20, 2016

Meng Weng, PhD
Dr. Meng Wang a former graduate student in the laboratory of Dr. Bohmann, has been named a Howard Hughes Medical Institute (HHMI) Faculty Scholar, a grant awarded to outstanding young scientists and researchers who have made impressive accomplishments and have a bright future in making groundbreaking contributions.
Dr. Wang is currently an associate professor at Baylor College of Medicine, where she studies the influence of endocrine and metabolic functions on aging, using C. elegans as a model system.
Research Led by Hucky Land Points to Prostate Cancer Tool
Tuesday, December 20, 2016
Researchers from Wilmot Cancer Institute and Roswell Park Cancer Institute in Buffalo reported in the journal Oncotarget that they have discovered a possible new tool for predicting whether prostate cancer will reoccur following surgery based on the expression patterns of four genes.
The Wilmot/Roswell Park tool was able to predict recurrence, based on human tissue samples and known patient outcomes, with 83 percent accuracy. Currently the only other way to estimate tumor aggressiveness is with a Gleason score, a grading system for prostate tumors that has limited power in most cases, researchers said.
Some prostate cancers grow very slowly, and when the disease is detected early, the five-year survival rates are nearly 100 percent. However, some men are diagnosed with more aggressive localized disease and, even after having a radical prostatectomy, cancer will return in one-third of patients.
“Our study sought to improve upon the prediction tools used in these types of cases so that oncologists would know with more certainty when to recommend additional treatment, such as radiotherapy, immediately after surgery,” said Hucky Land, Ph.D., director of research at Wilmot and the Robert and Dorothy Markin Chair of the Department of Biomedical Genetics, who led the research. (Most patients receive no further treatment after surgery.)
Earlier, Land’s lab discovered a large group of non-mutated genes that are actively involved in cancer development. After analyzing expression of this gene set in frozen prostate cancer tissue samples, researchers discovered the four-gene signature, which was expressed differently in prostate cancer that later returned. Justin Komisarof, an M.D./Ph.D. student in the Land lab, developed the various algorithms and methods to evaluate the gene signature. The research team concluded that their tool outperformed other scientific methods, and they have applied for a U.S. patent.
The National Institutes of Health and Wilmot Cancer Institute/Roswell Park Cancer Institute Collaboration Pilot Funds supported the research. Chief collaborators from Roswell Park include Carl Morrison, M.D., executive director of the Center for Personalized Medicine, and James Mohler, M.D., associate director and senior vice president for translational research at Roswell.
Jimmy Zhang, Awarded Two-year American Heart Association Predoctoral Fellowship
Friday, December 16, 2016
Jimmy Zhang, graduate student in the laboratory of Dr. Paul Brookes was awarded a two-year American Heart Association predoctoral fellowship entitled,
“The Development of Novel Acute Myocardial Infarction Therapeutics Using Metabolomics and High-Throughput Screening” beginning January 1, 2017.
Project Summary:
Paradoxically, current AMI therapies have the common goal of promoting reperfusion and, in doing so, trigger events that lead to cell death. As a result, there is a need for new therapeutics that limit reperfusion-induced injury.
Many of the pathologic cellular events of reperfusion-induced injury can be attributed to maladaptive metabolic remodeling. One particular metabolite of interest is succinate, which accumulates during ischemia. Upon reperfusion, succinate is consumed in the electron transport chain by Complex II, generating reactive oxygen species at Complex I. This reverse electron transport (RET) appears to be a major contributor to IR injury. Yet, despite the relevance of RET to IR injury, the pathway of succinate accumulation has yet to be elucidated. Additionally, succinate accumulation during ischemia might contribute to the generation of the mitochondrial membrane potential by permitting Complex I activity. This membrane potential can then be used for functions such as membrane transport and maintenance of redox status.
In our preliminary data, nornicotine was identified as a potentially cardioprotective candidate, and was shown to inhibit Complex I activity. Inhibition of RET could be the mechanism of protection by nornicotine. Using high-throughput screening and metabolomic approaches, this project will determine whether inhibition of RET is a rapid metabolic adaptation that is conserved across cardioprotective strategies (nornicotine treatment, ischemic preconditioning, and ischemic postconditioning). Finally, the pathway and function of succinate accumulation will be investigated by measuring membrane potential and redox status in isolated mitochondria.
Overall, this project aims to investigate RET in IR injury with the goal of developing novel therapeutics for AMI.
Dr. Robert Block awarded a 2-year research grant from the international Atherosclerosis Society and Pfizer Pharmaceutical Corporation
Thursday, December 15, 2016
Robert Block, MD, MPH, was awarded a 2-year research grant from the international Atherosclerosis Society and Pfizer Pharmaceutical Corporation. The overall goal is to partner with patients with familial hypercholesterolemia (a genetic disease that causes very premature heart attacks and strokes) and physicians in order to build and test educational/motivational information about this disease within the University of Rochester's electronic health record.
Researchers Identify Brain Region as Possible Target for Dementia Prevention
Tuesday, December 13, 2016
A University of Rochester study has found that older adults with excellent memories have more efficient connections between specific areas of the brain — findings that could hold promise for the prevention of dementia and cognitive decline.
Although researchers have historically viewed memory deterioration as an inevitable part of the aging process, a small group of older adults — called “supernormals” — are able to maintain their memory capacities much better than their peers. Feng (Vankee) Lin, PhD, an assistant professor in the University of Rochester School of Nursing, is spearheading a new approach to the study of Alzheimer’s disease by exploring what can be learned from these individuals.
In a study on the topic published in Cortex, an international journal devoted to the study of cognition and the relationship between the nervous system and mental processes, Lin and her team explored differences in brain function among three groups of older adults: supernormals, who were defined as having higher than average memory scores for their age, older adults diagnosed with amnestic mild cognitive impairment who are at high risk for developing Alzheimer’s disease, and a healthy control group. The study is the first to compare the brain function of supernormals to those who are at risk for developing Alzheimer’s.
Mitchell O'Connell Lab To Open
Monday, December 12, 2016
Current Postdoctoral Berkeley Fellow, Mitchell O'Connell, Ph.D. is set to open his new lab in April 2017, in the department of Biochemistry & Biophysics, at URMC. Currently Mitch is working in Jennifer Doudna's lab and his research aims to understand the mechanisms of RNA-mediated gene regulation through the development of new RNA-targeting tools based on CRISPR/Cas technology.
Welcome Mitch!
University Research Award helps team explore regeneration in a critical layer of the cornea
Friday, December 9, 2016
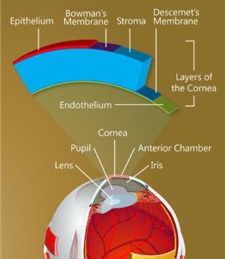
The structure of the cornea.
(Keratomania.com eye diagram by
Chabacano,via Wikimedia Commons.)
On the backside of the cornea is a single layer of cells that plays an all-important role, maintaining just the right fluid balance to keep the cornea transparent so that light can enter the eye. Until recently, it was believed this layer, called the corneal endothelium, is incapable of replacing its damaged cells. As more cells become damaged, the cornea becomes opaque, leading to loss of vision and, ultimately, to as many as 30,000 endothelium transplants a year in the United States alone.
A team of University researchers is exploring the possibility that stem cells on the outer edges of the cornea, given the right stimulation, can migrate into the endothelium to replace damaged cells. (Undifferentiated stem cells develop into specialized cells.) The work raises the possibility of restoring vision without the need for transplants.
The team is led by Amy Kiernan, associate professor of ophthalmology, and includes Jannick Rolland, the Brian J. Thompson Professor of Optical Engineering; Patrice Tankam, a senior scientist in the Center for Visual Science; Changsik Yoon, a graduate student in Rolland's lab; Rebecca Rausch, a graduate student in Kiernan's lab; and Holly Hindman, former associate professor of ophthalmology, now in private practice but still consulting on the project. They are supported with a $75,000 University Research Award. The URA program is designed to help researchers develop preliminary data or proof of concept needed to leverage larger federal or foundation awards to carry a promising project to completion.
There have been tantalizing clinical hints that the corneal endothelium may have regenerative capabilities, Kiernan says. For example, there have been cases in which endothelial transplants failed to engraft, but the cornea cleared up anyway, with regeneration of the endothelium occurring on its own. "So it seems that if something is done that stimulates a progenitor or stem cell population, most likely those in the periphery of the cornea, there is some regenerative capacity in the endothelium -- just based on clinical studies," Kiernan says.
Her team will attempt to identify the potential stem cells that might be stimulated to migrate to the endothelium to repair damage. They will use mouse models from Kiernan's lab in which adult stem cells can be permanently tagged with fluorescent biomarkers and tracked even after they differentiate into other cells. The identification and tracking of those cells will be done by refining a novel imaging approach developed in Rolland's lab. Called Gabor domain optical coherence microscopy, the technology allows rapid, noninvasive imaging of cellular structures beneath the surface of the skin or within the human eye -- in greater detail than traditional imaging with optical coherence tomography.
"Think of it as a high-definition, volumetric imaging," Rolland says. "But we also want to know what kind of cells we are looking at, so we are integrating fluorescence imaging with the high-definition volumetric microscopy so we can do both." The team represents a combination of pertinent expertise: cell development and regeneration (Kiernan and Rausch), imaging (Rolland, Tankam, and Yoon), and the biological basis for corneal and ocular surface diseases in humans (Hindman). The University Research Award funding is helping support graduate students and technicians working on the project, and the cost of mice and supplies. "Pilot funding like this is so important, especially with NIH grants shrinking," Kiernan says.
"It's really helpful to be able to bridge this kind of interdisciplinary effort," says Rolland. "You need to work together a little bit to understand the challenges involved and what you need to do to secure preliminary data, to show we have a pathway. "It takes time to get data, so even a small grant that provides a bridge for a year or two can make a huge difference."
Nina Schor to Step Down as Children's Hospital Pediatrician-in-Chief
Wednesday, December 7, 2016
Nina Schor, M.D., Ph.D., William H. Eilinger Chair of Pediatrics at the University of Rochester Medical Center (URMC), will step down as chair in June 2017. Schor served 11 years as pediatrician-in-chief at UR Medicine’s Golisano Children’s Hospital, and under her leadership, the Department of Pediatrics fulfilled a decades-long dream of building a standalone children’s hospital in Rochester; the new facility opened its doors to patients in July 2015. "I don’t want to downplay the significance of the new hospital, but it’s really what we do inside of it and because of it that’s so important,” said Schor. “I look at the academic physicians and physician scientists who came to Rochester with just a dream and a fire in their belly and how they’ve now brought those dreams to fruition — that’s what I’m most proud of.”
The Department of Pediatrics grew from 110 faculty members to over 170 during Schor’s tenure, creating new divisions in palliative care, sleep medicine, allergy, and hospitalist medicine. Research centers focused on premature infants, translational molecular programs, and red blood cell development also developed under Schor’s leadership.
“Not only was the new hospital built under Nina’s leadership, but she truly championed the project, ensuring that every detail was designed with patients and families in mind,” said Mark Taubman, M.D., URMC CEO and Dean of the School of Medicine and Dentistry. “She has been the face of the children’s hospital and inspired trust in our families, physicians, and donors at a time when we very much needed the community’s support.”
Study Challenges Autism Brain Response Theory
Tuesday, December 6, 2016
A new study challenges the hypothesis that nerve cells in the brains of individuals with Autism Spectrum Disorders do not reliably and consistently respond to external stimuli. “Our findings show there is no measurable variation in how individuals with autism respond to repeated visual and tactile stimuli,” says senior author John Foxe, the Kilian J. and Caroline F. Schmitt Professor in Neuroscience.
New Biophysical Research Service Available
Friday, December 2, 2016
The University has purchased a J-1100 circular dichroism (CD) spectrometer from JASCO Inc. The shared-use instrument will be housed and maintained as part of the Structural Biology and Biophysics facility. Manager Jermaine Jenkins will maintain the instrument, as well as manage user time, train users, and assist with data collection and analysis as needed. Email Jermaine Jenkins, Ph.D. to plan your experiments.
Helena Temkin-Greener, PhD was awarded a two-year research grant from the Donaghue Foundation
Thursday, December 1, 2016
Helena Temkin-Greener, PhD was awarded a two-year research grant from the Donaghue Foundation. The overall goal of the research project is to develop process and outcome-based measures of care quality for nursing home residents with mental health and behavioral disorders, and to explain variations in these measures across facilities and regions/states. Locally, findings will provide a benchmark performance measure for nursing homes participating in the NYS Delivery System Reform Incentive Payment (DSRIP) Program.
NIH Pre-Doctoral Fellowship Award
Tuesday, November 29, 2016
Matt Cavanaugh, a fifth year Neuroscience Graduate Program student in Dr. Krystel Huxlin’s lab was awarded an NIH Individual Pre-doctoral Fellowship from the National Eye Institute for his project entitled: Properties of training-induced visual recovery in cortical blindness (2016-2019).
Congratulations Matt!
Brain training video games help low-vision kids see better
Monday, November 28, 2016
Studies going back several years have shown that playing action video games (AVG) can help improve visual acuity. A new study by vision scientists at the University of Rochester and Vanderbilt University found that children with poor vision see vast improvement in their peripheral vision after only eight hours of training via kid-friendly video games. Most surprising to the scientists was the range of visual gains the children made, and that the gains were quickly acquired and stable when tested a year later.
"Children who have profound visual deficits often expend a disproportionate amount of effort trying to see straight ahead, and as a consequence they neglect their peripheral vision," said Duje Tadin, associate professor of brain and cognitive sciences at Rochester. "This is problematic because visual periphery—which plays a critical role in mobility and other key visual functions—is often less affected by visual impairments."
"We know that action video games (AVG) can improve visual perception, so we isolated the AVG components that we thought would have the strongest effect on perception and devised a kid-friendly game that compels players to pay attention to the entire visual field, not just where their vision is most impaired," said Tadin, who is also a professor in the Center for Visual Science. "As a result, we've seen up to 50 percent improvement in visual perception tasks."
Children’s Hospital Pediatrician-in-Chief Named Fellow of the AAAS
Monday, November 21, 2016
Nina Schor, M.D., Ph.D., William H. Eilinger Chair of Pediatrics and the pediatrician-in-chief at UR Medicine’s Golisano Children’s Hospital, has been named a Fellow of the American Association for the Advancement of Science (AAAS), the world’s largest general scientific society.
AAAS, which will publish the announcement on Nov. 25 in its journal Science, selects Fellows based on their scientifically and socially distinguished efforts to advance science or its applications. Schor has spent much of her career investigating neuroblastoma — which is among the most common childhood cancers — and was recognized for “her distinguished contributions to developmental neuroscience and neuropharmacology, particularly using molecular neuroscientific discoveries to design innovative therapies for tumors of the developing nervous system.”
‘Antisense’ compounds offer new weapon against influenza A
Thursday, November 17, 2016
Challenging a long-held convention, University researchers have shown they can inhibit the influenza A virus by targeting its genomic RNA with “antisense” compounds.
Their findings, highlighted on the cover of Nucleic Acid Therapeutics, offer scientists a new way to attack an increasingly drug-resistant pathogen that causes an estimated 250,000 to 500,000 deaths a year.
“Antisense” compounds are synthesized with nucleotides, the building blocks of nucleic acid, often shown as various combinations of A, U, G and C. When the compounds – called antisense oligonucleotides (ASOs) – bind to the targeted genomic RNA, they block its ability to replicate.
The collaboration, involving the labs of Douglas Turner, professor of chemistry; Luis Martinez-Sobrido, associate professor of microbiology and immunology; and two researchers in Poland, reported that “antisense” compounds targeting one of the virus’ eight genomic RNA segments caused a five- to 25-fold reduction of influenza A virus in cell cultures.
“That’s a big difference,” Martinez-Sobrido says. “When mice are infected with 10,000 viruses, they all die. However, with 25 times less virus, all animals can survive infection and they don’t even develop symptoms.”
Wilmot Co-directors Honored with Davey Award
Tuesday, November 15, 2016

Hartmut “Hucky” Land, Ph.D. (left) and David C. Linehan, M.D.
Wilmot Cancer Institute’s co-directors Hartmut “Hucky” Land, Ph.D., and David C. Linehan, M.D., were recognized recently with the Davey Award, an honor bestowed on University of Rochester faculty members who have made outstanding contributions to cancer research.
They received their awards at the 21st annual Wilmot Scientific Symposium Nov. 10. The award for Land, who organizes the annual symposium, was a surprise orchestrated by Jonathan W. Friedberg, M.D., M.M.Sc., director of Wilmot Cancer Institute.
At the symposium, Land presented the planned Davey Award to Linehan, who is also Wilmot’s director of clinical operations and the Seymour I. Schwartz Professor and Chairman of Surgery. Linehan was recognized for his work studying the role of the tumor microenvironment in promoting treatment resistance in pancreatic cancer.
Before his lecture, Linehan presented Land the surprise award with a recorded video message from Friedberg. Land, who is also Wilmot’s director of research and the Robert and Dorothy Markin Professor of Biomedical Genetics, was recognized for his body of work and for his work studying the genetic programs that control all of cancer’s worst shared features — such as a cancer cell’s ability to quickly divide and survive despite aggressive treatment.
Repurposed Drug May Offer Diagnosis, Treatment for Traumatic Nerve Damage
Monday, November 14, 2016
Researchers at the University of Rochester Medical Center believe they have identified a new means of enhancing the body's ability to repair its own cells, which they hope will lead to better diagnosis and treatment of traumatic nerve injuries, like those sustained in car accidents, sports injuries, or in combat. In a study published today, the team showed that a drug previously approved for other purposes can 'wake up' damaged peripheral nerves and speed repair and functional recovery after injury.
The study appearing in EMBO Molecular Medicine, demonstrates for the first time that 4-aminopyridine (4AP), a drug currently used to treat patients with the chronic nerve disease, multiple sclerosis, has the unexpected property of promoting recovery from acute nerve damage. Although this drug has been studied for over 30 years for its ability to treat chronic diseases, this is the first demonstration of 4AP's benefit in treating acute nerve injury and the first time those benefits were shown to persist after treatment was stopped.
Study authors, Dr. John Elfar, associate professor of Orthopaedics, and Mark Noble, Ph.D., Martha M. Freeman, M.D., Professor in Biomedical Genetics, and their laboratory team, found that daily treatment with 4AP promotes repair of myelin, the insulating material that normally surrounds nerve fibers, in mice. When this insulation is damaged, as occurs in traumatic peripheral nerve injury, nerve cell function is impaired. These researchers found that 4AP treatment accelerates repair of myelin damage and improvement in nerve function.
Catherine Ovitt Featured in D&C's Hot Jobs
Sunday, November 13, 2016
Dr. Catherine Ovitt
Salivary glands, which make as much as a quart of saliva each day, don’t pose a life-threatening risk if they stop working properly. But given their roles — they are important for swallowing, keep the inside of your mouth moist so your cheeks can move around, and have both anti-fungal and anti-bacterial properties — a malfunction would greatly impact quality of life.
Medical scientist Catherine Ovitt has dedicated her career to the study of salivary glands, in particular to establishing therapeutic strategies for their repair or regeneration after damage from radiation treatment due to head and neck cancers, or because of cellular damage from autoimmune diseases.
“A long-term goal would be to develop some sort of cell therapy treatment, some kind of transplantation or artificial salivary gland,” said Ovitt, who lives in Pittsford and is an associate professor in the Center for Oral Biology, part of UR Medicine’s Eastman Institute for Oral Health. Without the glands, she added, “you end up losing all your teeth.”
Karl Smith Featured as Part of MAG Hidden Passions Series
Tuesday, November 8, 2016
Biophysics PhD candidate, Karl Smith, is giving a talk at the Memorial Art Gallery, this Thursday at 7pm as part of their Hidden Passions speaking series.
At URMC, Smith studies glass filters 10,000 times thinner than a human hair as part of the Nanomembranes Research Group. It’s because of his rigorous academic schedule that he began the 10-cent project.
The Pittsburgh native has written more than 900 stories, each roughly 500 words, on half sheets of paper. Strangers give him a prompt, and he pecks away. He’s crafted stories about lost loves, lost dogs, sea lions, flying princesses, and frogs who jump over the moon. Stories about babies, treehouses, aardvarks, and dancing polar bears. Stories about murder.
Read more about Karl and his passion.
Thomas Mariani Authors Study on Infant Nose, Lung Cells
Thursday, October 13, 2016
Cells from an infant's nose are remarkably similar to those found in the lungs, a discovery that could lead to much more precise diagnosis of respiratory syncytial virus (RSV) and other infant lung diseases, according to new URMC research.
The study, published in Scientific Reports, provides a potential avenue for diagnosis that has challenged physicians for years, as infants with respiratory disease are usually so fragile that attempting to obtain lung samples is unsafe. Nasal cells, however, can be captured through a simple swab of the nostril, and their similarity to lung cells on an RNA level would allow physicians to get an accurate representation of how the lung is responding during disease states, without the need for more invasive tests.
"An infant with RSV could potentially have their nasal cells tested to determine if they are among the small group that will develop a severe response that might require hospitalization," said Thomas Mariani, Ph.D., professor of Pediatrics and the study's lead author. "Additionally, we could potentially use this method to examine other at-risk infants, such as those born prematurely who face a greater risk for lung disease throughout life — and identify which of those children should be treated more aggressively."
The research also carries tremendous promise for future studies. While scientists have made significant progress over the past several decades to better understand adult lung diseases — such as chronic obstructive pulmonary disease and lung fibrosis — discovery has not been nearly as robust for infant diseases, due to the risks involved in securing lung tissue.
But the relative ease of obtaining nasal cells could accelerate understanding of how infant lungs respond to RSV and other diseases. While this study examined 53 healthy infants as a means of establishing a benchmark for normal cell structure, researchers at URMC have already begun studying the nasal tissue of diseased infants. Early results are promising.
"We're actively working on studies in infants with lung diseases, and we're showing quite clearly that we can identify differences between those with mild disease and those with more severe outcomes," Mariani said.
The research is conducted by URMC's Respiratory Pathogens Research Center, which coordinates its work with the national Respiratory Pathogens Research Center established by the National Institute of Allergy and Infectious Diseases. The Center, under the direction of David Topham, Ph.D., focuses on research that will lead to a better understanding of the interactions between respiratory pathogens, the immune system, and other genetic and environmental factors.
ChinYi Chu, M.S., Xing Qiu, Ph.D., Lu Wang, M.S., Soumyaroop Bhattacharya, M.S., M.Ed., Gerry Lofthus, Ph.D., Anthony Corbett, M.S., Jeanne Holden-Wiltse, M.S., Alex Grier, M.S., Brenda Tesini, M.D., Steve Gill, Ph.D., Ann Falsey, M.D., Mary Caserta, M.D., and Ed Walsh, M.D., from the University of Rochester, contributed to these studies.
Research Will Explore New Therapies for Huntington's Disease
Wednesday, October 12, 2016
A new award from the CHDI Foundation will advance promising research that aims to slow the progression of Huntington's disease. The funding, anticipated to total more than $10.5 million over next five years, will help University of Rochester Medical Center (URMC) scientists develop a stem cell-based therapy that swaps sick brain cells for healthy ones.
The new award will go to the lab of Steve Goldman, M.D., Ph.D., the co-director of the URMC Center for Translational Neuromedicine, which has research operations in both Rochester and at the University of Copenhagen.
Huntington's is a hereditary neurodegenerative disease characterized by the loss of medium spiny neurons, a nerve cell in the brain that plays a critical role in motor control. As the disease progresses over time and more of these cells die, the result is involuntary movements, problems with coordination, and cognitive decline, depression, and often psychosis. There is currently no way to slow or modify this fatal disease.
The new award will support research that builds upon findings published by Goldman earlier this year in the journal Nature Communications showing that researchers were able to slow the progression of the disease in mice by transplanting healthy human support cells, called glial progenitor cells, into the animals' brains.
Wilmot Scientists Exploit Cell Metabolism to Attack Cancer
Wednesday, October 12, 2016
Cancer cells have their own unique ways of reproducing, involving a shrewd metabolic reprograming that has been observed in virtually all types of cancer but not in normal cells. Now, University of Rochester Medical Center scientists have pinpointed one key source of the problem, which could lead to new treatment opportunities.
In an article published by Cell Reports, the scientific team shows for the first time how cancer-causing mutations control and alter the way cancer cells biosynthesize and replicate.
The discovery is the result of a close collaboration between the laboratories of Joshua Munger, Ph.D., associate professor of Biochemistry and Biophysics, and Hucky Land, Ph.D., the Robert and Dorothy Markin Professor and Chair of Biomedical Genetics and director of research at the URMC’s Wilmot Cancer Institute.
“Every tissue or cell type in the body has different metabolic needs but as cells become cancerous their metabolism shifts in ways that are very different from normal cells,” Munger said. “Being able to identify those differences is critical for developing treatment targets.”
Armond Collins Presents his work from Fudge Lab
Tuesday, October 11, 2016
Armond Collins, a second year medical student, presents the study he conducted with us this summer under the auspices of a Babigian Fellowship. Armond studied changes in myelination in amygdala and cortex of adult rats that had been exposed to 3 day bout of a repeated variable stressor during adolescence. His works follows up studies by Michele Saul, PhD that indicate that adolescent stress results in decreased oligodendrocyte precursors in the amygdala in the week following the stress.
NIH Director Visits URMC, Says it’s an Exciting Time to be a Researcher
Monday, October 10, 2016
 Collins' first stop was lunch with 15 graduate students and postdocs who came prepared with a wide range of questions. The discussion covered the importance of communicating science to the public and policymakers, increasing diversity in biomedical research and new mechanisms to support young scientists at the start of their careers. Postdoctoral fellow Sarah Latchney and Ph.D. graduate student Solomon Abiola attended the lunch with Collins and describe the experience here.
Collins' first stop was lunch with 15 graduate students and postdocs who came prepared with a wide range of questions. The discussion covered the importance of communicating science to the public and policymakers, increasing diversity in biomedical research and new mechanisms to support young scientists at the start of their careers. Postdoctoral fellow Sarah Latchney and Ph.D. graduate student Solomon Abiola attended the lunch with Collins and describe the experience here.
Members of the Center for RNA Biology highlighted their most promising work for Collins and Center director Lynne E. Maquat, Ph.D., gave Collins a tour of her lab, where he met more trainees and junior researchers (admittedly, Collins' favorite part of visits like these).
 In his keynote address at the end of the day, Collins delivered an uplifting message to a packed house in the Class of '62 auditorium: it is an extremely exciting time to be in biomedical research, and after many lean years we are turning a corner, with the NIH budget finally increasing in real terms. He detailed several of the NIH's new programs, like the Human Microbiome Project, Big Data to Knowledge (BD2K), the Precision Medicine Initiative and the Cancer Moonshot.
In his keynote address at the end of the day, Collins delivered an uplifting message to a packed house in the Class of '62 auditorium: it is an extremely exciting time to be in biomedical research, and after many lean years we are turning a corner, with the NIH budget finally increasing in real terms. He detailed several of the NIH's new programs, like the Human Microbiome Project, Big Data to Knowledge (BD2K), the Precision Medicine Initiative and the Cancer Moonshot.
He applauded URMC on the renewal of the CTSI funding and cited the translational research conducted by Arthur J. Moss, M.D., which has led to new treatments for patients with Long QT syndrome (LQTS), and John J. Treanor, M.D., which is helping scientists in pursuit of a universal flu vaccine. Collins outlined several new funding initiatives, including the NIH Director's Early Independence Award, which is helping assistant professor Elaine L. Hill, Ph.D., study the impact of fracking on infant and child health.
Collins affirmed that the U.S. is the strongest biomedical research country in the world thanks to institutions like URMC. You can view his keynote, "Exceptional Opportunities in Biomedical Research," here.
New Grants Explore Role of Brain’s “Garbage Truck” in Mini-Stokes and Trauma
Wednesday, October 5, 2016

Maiken Nedergaard, M.D., D.M.Sc.
More than $4.5 million in new grants to the lab of University of Rochester Medical Center scientist Maiken Nedergaard, M.D., D.M.Sc., underscore the important role the brain's waste disposal system may play in a range of neurological disorders. The new awards will advance understanding of how small vessel disease and traumatic brain injury can give rise to cognitive and behavioral problems.
Nedergaard and her colleagues first unveiled the brain's unique method of removing waste -- dubbed the glymphatic system -- in a paper in Science Translational Medicine in 2012. The research revealed that the brain possesses a circulation network that piggybacks on blood vessels and uses cerebral spinal fluid to flush away waste products from brain tissue. Since that time, the team has gone on to show that the glymphatic system works primarily while we sleep, could be a key player in diseases like Alzheimer's, and is disrupted after traumatic brain injury.
NGP Student Awarded NIH Fellowship
Monday, October 3, 2016
Rebecca Rausch, a fifth year neuroscience graduate student in Dr. Richard Libby's lab was awarded an NIH Individual Pre-doctoral Fellowship from the National Eye Institute for her project entitled: The Role of Notch and BMP Signaling in Anterior Segment Dysgenesis (2016-2019).
Congratulations Becca!
Nuclear Protein Causes Neuroblastoma to Become More Aggressive
Wednesday, September 28, 2016
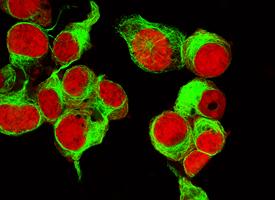
Aggressive forms of neuroblastoma contain a specific protein in their cells’ nuclei that is not found in the nuclei of more benign forms of the cancer, and the discovery, made through research from the University of Rochester Medical Center (URMC), could lead to new forms of targeted therapy.
EYA1, a protein that contributes to ear development, is present in the cytoplasm of many neuroblastoma tumors, but this protein migrates to the nucleus in the cells of more aggressive forms of the disease. The research, recently published in two medical research journals, allows for the development of targeted drugs that will work to prevent the neuroblastoma from reaching this more aggressive stage; researchers at URMC and elsewhere have already begun testing some of these potential treatments in a laboratory setting.
“Neuroblastoma is one of the most common and deadly forms of childhood cancer, and this discovery allows us to identify drugs that prevent the change in EYA1 structure and potentially minimize the danger to a child who has this disease,” said Nina Schor, M.D., Ph.D., professor of Pediatrics and Neuroscience and the William H. Eilinger Chair of Pediatrics at URMC.
URMC Researchers Discover Rare Flu-Thwarting Mutation
Wednesday, September 28, 2016
A rare and improbable mutation in a protein encoded by an influenza virus renders the virus defenseless against the body’s immune system. This University of Rochester Medical Center discovery could provide a new strategy for live influenza vaccines in the future.
A new approach to the live flu vaccine would be particularly advantageous right now after the Centers for Disease Control and Prevention stopped recommending use of the live attenuate flu vaccine, FluMist® earlier this year. Several studies found that the pain-free nasal spray, which was used in about one-third of young children in the U.S., offered no protection to that especially vulnerable population. The flu shot, on the other hand, performed well and the CDC recommends using this vaccine in place of FluMist®.
“There is a need to understand what's happening with the existing live vaccine and potentially a need to develop a new one,” said David Topham, Ph.D., Marie Curran Wilson and Joseph Chamberlain Wilson Professor of Microbiology and Immunology at URMC and author of the study. “We proposed that the mutation we found could be used to create a live vaccine.”
Jessica Cantlon Named One of 10 Scientists to Watch by Science News
Friday, September 23, 2016

Jessica Cantlon
Jessica Cantlon, associate professor of Brain and Cognitive Sciences, was selected by Science News as one of their 10 early- to mid-career scientists to watch. Cantlon’s work centers on how human and nonhuman primates distinguish between quantities. Understanding how the brain makes sense of concepts such as estimating quantities and counting might lead to better ways of teaching numerical concepts to children.
American Health Council Names Dr. Harold Smith, Ph.D. to Education Board
Monday, September 19, 2016
Dr. Harold Smith, Professor at The University of Rochester, has been selected to join the Education Board at the American Health Council. Dr. Smith will be sharing his knowledge and expertise in the field of molecular biology, molecular virology, RNA biology, and drug discovery.
Dr. Harold Smith became involved in research after beginning his career as a professor in the Department of Biochemistry at The University of Rochester. As a biophysics professor, he utilized his knowledge and expertise in the areas of research and innovation of RNA, protein molecular biology, cell regulation, and drug discovery. Furthermore, Dr. Smith develops curriculum, teaches and mentors students from high school to postgraduate.
Dr. Harold Smith is also the Founder, President, and CEO of OyaGen, Inc. The objective of OyaGen, Inc. is to induce transient and beneficial changes in the protein expression and function in human tissues by involving the editing enzymes in targeting biomedically relevant pathways.
Dr. Harold Smith is a member of The American Heart Association, The Council on Atherosclerosis, The RNA Society, The American Society for Biochemistry and Molecular Biology and a fellow in the The Royal Society of Biology. In addition, Dr. Smith serves on the Scientific Advisor Board of Cannabis Sciences, Inc., IgxBio, Inc. and Trovita Health Sciences as well as the Editorial Board of the International Journal of Virology and AIDS, Frontiers in Microbiology, The Journal of Biological Chemistry, and The Journal of BioDiscovery.
Miller Receives Patent for Technology that Can Help Detect Flu
Thursday, September 15, 2016
Benjamin L. Miller, Ph.D., professor of Dermatology, recently received yet another patent for a new technology that can detect miniscule amounts of specific molecules in blood or other liquids. The patent focuses on using this technology to make detecting immune responses to the flu quicker and easier.
The AIR™ Platform, marketed by Adarza Biosystems, can detect immune responses to flu vaccines as well as the virus itself. With a small blood sample from a patient, doctors can confirm a flu infection, see if the patient mounts an appropriate immune response to a vaccine, or see if immune responses cross react with several different strains of flu. AIR™ can also be used for viral surveillance.
While Miller’s AIR™ system is not the first to make these things possible, it is a great improvement on previous technologies. Its silicon chip, which is only about the size of the end of a pencil eraser, allows scientists to detect hundreds of different target molecules in a single drop of fluid, and its “label-free” design requires fewer steps and reagents, thus reducing cost and opportunities for error.
“Label-free” systems suppress background noise to detect tiny signals, whereas conventional “labeled” systems use a more cumbersome design to amplify a tiny signal, often creating a lot of background noise in the process.
“It’s like walking through a city during the day and looking up at the buildings,” Miller said. “You have no idea what's going on in the offices because there's so much ambient light, but if you come back at night, it's easy to see.”
Miller suppresses background noise using a near-perfect anti-reflective coating on his silicon chips. For every 100 million photons of light that hit the surface of the chip, only one photon is reflected back. That coating also contains capture molecules meant to bind or “capture” specific target molecules, like antibodies produced in response to the flu virus. The more antibodies that bind to the chip, the more the anti-reflective coating is perturbed, and the more light is reflected and captured by a camera.
This simple and unconventional design and the ability to use capture molecules both big and small makes AIR™ extremely versatile. From cancer and infectious diseases, to agriculture and food safety, AIR™ is poised to expedite research and clinical testing across a wide range of applications.
Haber, Farrar Receive Awards
Tuesday, September 13, 2016
Suzanne N. Haber, Ph.D., has been awarded a NIH R13 Conference grant. On October 11-13, 2016, the University of Rochester Institute of Neuromedicine and the Silvio O. Conte Center will hold a meeting entitled “Persistent, maladaptive behaviors: why we make bad choices”. The program is designed to involve basic and clinical scientists with a specific focus on the fundamental elements that drive basic behaviors and action plans (reward, fear, and value assignment); circuit dysfunctions that underlie abnormalities in diseases with persistent, habit-like behaviors, despite some awareness that these behaviors are maladaptive; the circuit components that are common amongst diseases; computational approaches to understanding these circuits; and therapeutic approaches that effect these circuits.
Christopher Farrar, a Ph.D. candidate in the lab of Professor Denise Hocking, has been awarded a Ruth L. Kirschstein National Research Service Award (NIH-NRSA) Individual Predoctoral Fellowship (F31) from the National Heart, Lung, and Blood Institute for his project entitled “Influence of Extracellular Matrix Fibronectin on Platelet-Derived Growth Factor (PDGF) Signaling”. PDGF is produced by a variety of different cell types and stimulates mesenchymal cell proliferation, migration, and gene expression. Together with fibronectin, PDGF plays an important role in angiogenesis and wound repair. In contrast, excess PDGF and abnormal fibronectin matrix deposition are linked to several diseases, including pulmonary fibrosis, atherosclerosis, and certain cancers. The focus of Chris’ project is to determine how mesenchymal cell adhesion to extracellular matrix fibronectin fibrils influences the ability of these cells to respond to PDGF, with the long-term goal of developing new treatment approaches to effectively regulate the sensitivity of cells to growth factor stimulation.
Harris Gelbard Receives International Award for Neurovirology Research
Tuesday, September 13, 2016
 Harris "Handy" Gelbard, M.D., Ph.D., director of the Center for Neural Development & Disease, is slated to receive the Hilary Koprowski Prize in Neurovirology at this year's International Symposium on Molecular Medicine and Infectious Disease at Drexel University. Gelbard will be recognized for developing an unconventional drug that shows promise in treating brain disorders associated with HIV.
Harris "Handy" Gelbard, M.D., Ph.D., director of the Center for Neural Development & Disease, is slated to receive the Hilary Koprowski Prize in Neurovirology at this year's International Symposium on Molecular Medicine and Infectious Disease at Drexel University. Gelbard will be recognized for developing an unconventional drug that shows promise in treating brain disorders associated with HIV.
Gelbard's drug, URMC-099, calms the immune system when it goes awry, as happens in HIV Associated Neurocognitive Disorder (HAND). In HAND, immune reactions to HIV particles in the brain damage nerve cells and cause dementia. Because patients affected by HAND also have HIV, it was imperative that URMC-099 not interfere with the antiretroviral drugs that keep HIV-positive patients alive.
KL2 award helps researcher pave his career path
Friday, September 9, 2016

David Auerbach, senior instructor in medicine,
says his KL2 award has "opened many doors for me."
David Auerbach's interest in pursuing a scientific career began during a hockey game his first year of college, when a teammate — who turned out to be a chief medical examiner — asked Auerbach if he would like to observe a case.
Now Auerbach's career is taking a major step forward with a two-yearKL2 Mentored Career Development Program award from the University's Clinical and Translational Science Institute.
"It has opened up many doors for me," says Auerbach, including lead authorship of a recent paper in Neurology.
Auerbach, a senior instructor in the Department of Medicine/Aab Cardiovascular Research Institute, is taking a multisystem approach to understanding the mechanisms that cause electrical disturbances in both the hearts and brains of patients with genetic ion channel diseases. Ion channels, located in the plasma membrane of cells, are narrow tunnels that open and close at precise times to allow the flow of ions into or out of the cell, thus shaping the electrical activity in the heart and brain.
As a postdoc working with Lori Isom, a professor at the University of Michigan, Auerbach demonstrated that people with severe genetic forms of epilepsy were at a higher risk not only of electrical disturbances in the brain, resulting in seizures, but also of electrical disturbances in the heart, causing arrhythmias.
In order to establish an independent line of research, Auerbach decided to approach the problem in reverse: are people with long QT syndrome — a classically studied genetic cardiac disease that causes arrhythmias — also at an increased risk of seizures?
He came to Rochester in 2014 specifically because of its research strengths in this area, including the opportunity to work with such experts as Arthur Moss, the Bradford Berk Distinguished Professor of Medicine; Robert Dirksen, the Lewis Pratt Ross Professor and chair of pharmacology and physiology; and Charles Lowenstein, chief of cardiology and director of the Aab Cardiovascular Research Institute.
2016 Convocation Award Winners from Neuroscience
Thursday, September 8, 2016
Congratulations to the following people for winning teaching and student achievement awards at this year's SMD Opening Convocation.
Faculty Teaching, Mentoring & Diversity Awards
- Deborah Cory-Slechta, PhD
- John Olschowka, PhD
Medical & Graduate Student Achievement Awards
- Alexandra McHale - Irving L. Spar Fellowship Award
- Gavin Jenkins - Merritt and Marjorie Cleveland Fellowship
- Neal Shah - J. Newell Stannard Graduate Student Scholarship Award
- Grayson Sipe - Outstanding Student Mentor Award
Make sure to congratulate each of them when you see them.
Ann M. Dozier, PhD named Albert David Kaiser Chair of Public Health Sciences
Thursday, September 8, 2016
Ann M. Dozier, PhD, Professor Public Health Sciences, in the Center for Community Health, and of Clinical Nursing, named Albert David Kaiser Chair of Public Health Sciences at the Opening Convocation for the School of Medicine & Dentistry on September 8, 2016.
DOD Grant Explores New Drugs to Thwart Impact of Trauma, Stroke, and Cardiac Arrest
Thursday, September 8, 2016
A $2.3 million Department of Defense grant will help neuroscientists develop new treatments for the emergency room and the battlefield. The research will focus on the development of new therapies that could help protect brain and other at risk organs following a trauma, heart attack, or stroke.
“While we have made significant progress in our ability to restore blood flow after stroke or cardiac arrest, the medical community does not have drugs at its disposal to prevent the secondary damage that occurs after these events,” said University of Rochester Medical Center neurologist Marc Halterman, M.D., Ph.D., the principal investigator of the study. “This grant will further our research on a promising class of drugs that possess both anti-inflammatory and cytoprotective properties that we believe will be suitable for use in both military and emergency conditions.”
Kelly Thevenet-Morrison, M.S. awarded Outstanding Graduate Student Teacher award
Thursday, September 8, 2016
Kelly Thevenet-Morrison, M.S., Lead Programmer Analyst in the Department of Public Health Sciences, awarded Outstanding Graduate Student Teacher award at the Opening Convocation for the School of Medicine & Dentistry on September 8, 2016.
Anna Bird Receives Two Awards
Sunday, September 4, 2016
Anna Bird has received the Elkes Foundation Scholarship ($1200 in travel funds to the Keystone Symposium in Stockholm, Sweden) and the American Association of Immunologists (AAI) Trainee Abstract Award ($750 in travel funds for AAI Conference, Seattle 2016).
Automatic cortical representation of auditory pitch changes in Rett syndrome - John Foxe et al.
Thursday, September 1, 2016
Over the typical course of Rett syndrome, initial language and communication abilities deteriorate dramatically between the ages of 1 and 4 years, and a majority of these children go on to lose all oral communication abilities. It becomes extremely difficult for clinicians and caretakers to accurately assess the level of preserved auditory functioning in these children, an issue of obvious clinical import. Non-invasive electrophysiological techniques allow for the interrogation of auditory cortical processing without the need for overt behavioral responses. In particular, the mismatch negativity (MMN) component of the auditory evoked potential (AEP) provides an excellent and robust dependent measure of change detection and auditory sensory memory. Here, we asked whether females with Rett syndrome would produce the MMN to occasional changes in pitch in a regularly occurring stream of auditory tones.
Hayley Martin, MD-PhD student in Epidemiology receives student scholarship
Thursday, September 1, 2016
Hayley Martin, MD-PhD student in Epidemiology received a student scholarship to attend the 2016 Family Medicine Education Consortium Annual Meeting in Pittsburgh, PA. This meeting is aimed at family medicine physicians, residents and medical students in the north east interested in "improving the health of the community by strengthening Family Medicine / Primary Care services and medical education.
Biochemistry & Biophysics Faculty Member and Photojournalist Barry Goldstein Covers Republican National Convention for The American Scholar
Wednesday, August 31, 2016
Barry Goldstein is a photographer specializing in portraiture and documentary themes. Originally trained as a physician and biophysicist, he is Associate Professor of Medical Humanities at the University of Rochester Medical Center, Visiting Professor of Humanities at Williams College, and Adjunct Professor of Humanism in Medicine at the NYU Medical School. He was the first Artist-in-Residence at the New York University Medical School on September 11, 2001, an experience that led to his collection Being There: Medical Student Morgue Volunteers Following 9/11. His most recent book, Gray Land: Soldiers on War, is a collection of portraits of, and interviews with soldiers in Iraq and at home. He lectures and exhibits widely, and is a recipient of a Massachusetts Cultural Council Artists Grant in Photography.
Most recently, Barry provided coverage of the Republication National Convention for the American Scholar. His RNC work can be seen at The American Scholar and on his website.
Heilbronner and Yule Receive Awards at 2016 Convocation
Monday, August 29, 2016
Sarah R. Heilbronner, PhD, will receive the Postdoctoral Achievement Award at the 2016 Convocation, on September 8. She is currently a postdoc in Dr. Suzanne Haber's lab, where she is studying the neural circuitry associated with reward processing, decision-making, and executive function.
Along with the other members of Dr. Haber's team, Sarah is working to determine the anatomical connections that are affected by neurosurgical interventions for psychiatric disorders (such as deep brain stimulation).
Professor David I. Yule, Ph.D., will also receive the Faculty Teaching Award, specifically the Trainee Academic Mentoring Award in Basic Science, as well as the Louis C. Lasagna Endowed Professorship at convocation. The Yule Lab studies intracellular calcium signaling in cells which are typically, electrically non-excitable. In cells such as the liver, exocrine, pancreas, salivary glands and various cells in the blood, increases in intracellular calcium are fundamentally important for diverse processes including secretion of digestive enzymes and fluid, glucose metabolism together with cellular growth and differentiation.
Congrats Sarah and David!
Dr. Diana Fernandez assumes co-chair position of the Latino Health Coalition
Monday, August 15, 2016
Dr. Diana Fernandez, Associate Professor of Public Health Sciences, is assuming the co-chair position of the Latino Health Coalition convened by the Finger Lakes Health Systems Agency
Blanton Tolbert Wins Morton L. Mandel Award For Outstanding Chemistry Faculty
Tuesday, August 9, 2016

Blanton Tolbert
Former Biophysics student, Blanton Tolbert (PhD 2006), mentored by Doug Turner & Ravi Basavappa, has been awarded the Morton L. Mandel endows award for outstanding chemistry faculty members at Case Western Reserve University.
Associate Professor Blanton S. Tolbert, whose work focuses closely on elucidating molecular details of the human immunodeficiency virus, more commonly known as HIV. A member of the Case Western Reserve faculty since 2012, Tolbert paired the honor with extraordinary achievements during the past academic year:
- A featured cover story in the Journal of Molecular Biology that described new three-dimensional structures of molecules in the life cycle of HIV
- A second article about HIV’s molecular structures, published in the Journal of Biological Chemistry that became the journal’s most-viewed RNA (Ribonucleic acid) paper in December 2015
- Multiple online mentions of the work, including the Nov. 16 Science Highlights of the Advanced Photon Source at the U.S. Department of Energy’s Argonne National Library
- Service as director of the chemistry department’s Research Experience for Undergraduates (REU) grant from the National Science Foundation. In that role, Tolbert led efforts that identified and recruited a diverse group of students to work on federally sponsored projects. As part of the 10-week experience learning cutting-edge science, the students also participated in teambuilding and professional development activities. The work proved so successful that one undergraduate was cited as co-author on a publication from his summer project.
Congratulations Blanton!
How a Stone Spearhead Found in a Whale Could Help Solve the Mystery of Cancer
Monday, August 8, 2016
Bowhead whales can live over 200 years, but there is no evidence of a bowhead ever having cancer. "The biggest questions are what are the extra protections that whales have against cancer," says Vera Gorbunova, the Doris Johns Cherry Professor in the Department of Biology. "We would really like to understand the mechanism."
Lin Honored as 'Brilliant New Investigator'
Thursday, August 4, 2016
 University of Rochester Assistant Professor of Nursing Feng (Vankee) Lin, Ph.D., R.N. will be presented with the Brilliant New Investigator Award from the Council for the Advancement of Nursing Science (CANS) at the organization’s 2016 State of the Science Congress on Nursing Research, Sept. 15-17 in Washington, D.C.
University of Rochester Assistant Professor of Nursing Feng (Vankee) Lin, Ph.D., R.N. will be presented with the Brilliant New Investigator Award from the Council for the Advancement of Nursing Science (CANS) at the organization’s 2016 State of the Science Congress on Nursing Research, Sept. 15-17 in Washington, D.C.
The award recognizes the contributions of scientists early in their research careers who show extraordinary potential to develop sustained programs of research certain to have significant impact on the science and practice of nursing and health care. Nominees must show a record of building research productivity in an area of major significance to nursing and health care, research dissemination and translation to practice and/or policy, and emerging leadership related to the advancement of nursing science.
Pasternak Research Paper to be Published in J. Neuroscience
Thursday, August 4, 2016
 The paper "Prefrontal Neurons Represent Motion Signals from Across
the Visual Field but for Memory-Guided Comparisons
Depend on Neurons Providing these Signals" will be published in J. Neuroscience shortly.
The paper "Prefrontal Neurons Represent Motion Signals from Across
the Visual Field but for Memory-Guided Comparisons
Depend on Neurons Providing these Signals" will be published in J. Neuroscience shortly.
Visual decisions often involve comparisons of sequential visual motion that can appear at any location in the visual field. We
show that during such comparisons, the lateral prefrontal cortex (LPFC) contains accurate representation of visual motion
from across the visual field, supplied by motion processing neurons. However, at the time of comparison, LPFC neurons can
only use this information to compute the differences between the stimuli, if stimuli appear at the same retinal location,
implicating neurons with localized receptive fields in the comparison process. These findings show that sensory comparisons
rely on the interactions between LPFC and sensory neurons that not only supply sensory signals but also actively
participate in the comparison of these signals at the time of the decision.
Make sure to read the article when it comes out.
University of Rochester rising junior completes research project on infant feeding
Monday, August 1, 2016
Yareni Sime, a University of Rochester rising junior and Scholar in the Ronald E. McNair Post-Baccalaureate Achievement Program, was hosted this summer by Dr. Ann Dozier and her team that is studying infant feeding, health and safety. Ms. Sime's summer research project was entitled Suboptimal Infant Feeding Practices Among Hispanic/Latino Women in Monroe County.
Maquat Featured at Cornell-Ithaca Creativity Workshop
Saturday, July 30, 2016
J. Lowell Orbison Chair of Biochemistry and Biophysics, and of Oncology, Lynne Maquat, PhD, was a featured speaker at The Creativity Spark: a creativity workshop for scientists, a workshop put on by Cornell University, July 25.
The creativity workshop featured award winning scientists and scholars, including two Nobel Laureates, as they discussed the Creativity Spark and its role in science exploration.
Luebke and Bennetto Explore Hearing Test That May Identify Autism Risk
Monday, July 25, 2016
Researchers have identified an inner ear deficiency in children with Autism that may impact their ability to recognize speech. The findings, which were published in the journal Autism Research, could ultimately be used as a way to identify children at risk for the disorder at an early age.
“This study identifies a simple, safe, and non-invasive method to screen young children for hearing deficits that are associated with Autism,” said Anne Luebke, Ph.D., an associate professor in the University of Rochester Medical Center Departments of Biomedical Engineering and Neuroscience and a co-author of the study. “This technique may provide clinicians a new window into the disorder and enable us to intervene earlier and help achieve optimal outcomes.”
“Auditory impairment has long been associated with developmental delay and other problems, such as language deficits,” said Loisa Bennetto, Ph.D., an associate professor in the University of Rochester Department of Clinical and Social Sciences in Psychology and a co-author of the study. “While there is no association between hearing problems and autism, difficulty in processing speech may contribute to some of the core symptoms of the disease. Early detection could help identify risk for ASD and enable clinicians to intervene earlier. Additionally, these findings can inform the development of approaches to correct auditory impairment with hearing aids or other devices that can improve the range of sounds the ear can process.”
McNair Summer Scholar Ashley Bui Talks Amygdala Circuits
Friday, July 22, 2016
 Congratulations to Ashley Bui, a rising senior in Brain and Cognitive Sciences, on her presentation July 22, 2016. Her talk Projections from the Temporal Cortex to the Basal Nucleus of the Amygdala in the Macaque highlighted data from her summer project in our lab. The amygdala is required for computing which of the complex sensory stimuli that an individual encounters are emotionally meaningful, so that appropriate action can be taken. Ashley’s preliminary data shows that specific portions of the temporal cortex, which are critical for processing complex visual and auditory information, communicate with different regions of the amygdala. The results suggest that cortical areas that process complex visual information on 'what' and 'where' an object is (or is moving) are communicating with specific amygdala subregions. Thus, while determining the emotional importance of ‘what or who’ is important, biologic movements also likely influence amygdala activity and coding. We are happy that she will continue this work through the Fall semester.
Congratulations to Ashley Bui, a rising senior in Brain and Cognitive Sciences, on her presentation July 22, 2016. Her talk Projections from the Temporal Cortex to the Basal Nucleus of the Amygdala in the Macaque highlighted data from her summer project in our lab. The amygdala is required for computing which of the complex sensory stimuli that an individual encounters are emotionally meaningful, so that appropriate action can be taken. Ashley’s preliminary data shows that specific portions of the temporal cortex, which are critical for processing complex visual and auditory information, communicate with different regions of the amygdala. The results suggest that cortical areas that process complex visual information on 'what' and 'where' an object is (or is moving) are communicating with specific amygdala subregions. Thus, while determining the emotional importance of ‘what or who’ is important, biologic movements also likely influence amygdala activity and coding. We are happy that she will continue this work through the Fall semester.
NGP student plays with RPO
Thursday, July 21, 2016
Second year NGP student, Monique Mendes, had a unique opportunity to play alongside the Rochester Philharmonic Orchestra in their Side-by-Side Reading Session – a program that pairs amateur and professional musicians in a joint rehearsal and performance at Kodak Hall on July 21st.
Congratulations Monique!
Ryan Dawes defends thesis
Monday, July 18, 2016
Ryan Dawes successfully defended his thesis, "β-Adrenergic Receptor Signaling Constrains Breast Cancer Progression and
Modulates Tumor-Associated Exosome Content And Function" on July 18, 2016.
Congratulations Dr. Dawes!
Minsoo Kim Wins Dolph O. Adams Award
Friday, July 15, 2016
Congratulations to professor of Microbiology and Immunology and The Center for Vaccine Biology and Immunology, Minsoo Kim, who has won the 2016 Dolph O. Adams Award. Dr. Kim will be accepting the award at the SLB Annual Meeting in September in Verona Italy!
This annual award is named in honor of the outstanding macrophage researcher Dolph O. Adams, M.D., Ph.D. The award is to recognize excellence of an investigator working in the area of cellular and molecular mechanisms of host defense and inflammation.
Lisa A. DeLouise Receives Patent for Microfluidic Device
Friday, July 8, 2016
Lisa A. DeLouise, Ph.D., M.P.D., associate professor of Dermatology, Biomedical Engineering, Material Chemistry and Electrical and Computer Engineering and a member of the Environmental Health and Science Center, has received a patent for her microfluidic device and a method of manufacturing the device.
Research in the DeLouise Lab – funded by NYSTAR, NSF, DCFAR, CTSI and URVentures – has led to the development of a single cell screening technology platform based on microbubble well array. Single cell screening technologies can facilitate the discovery of rare cells.
DeLouise’s current work, in collaboration with James J. Kobie, Ph.D., assistant professor of Infectious Diseases, seeks to sort antigen-specific antibody-secreting B cells for the development of therapeutic monoclonal antibodies and the detection of cancer stem cells that harbor genetic mutations that confer their tumor-initiating and drug-resistant properties.
Meet Karl Smith, the Typewriting Tale Teller
Friday, July 8, 2016

Karl Smith
The "Friends of Joe's Big Idea" is a vibrant community of talented people we think you should meet. With our feature, FOJBI Friday, we're introducing some of these cool communicators of science in their own words. This week: Karl Smith.
Background
I'm a fifth-year biophysics doctoral candidate at the University of Rochester, where I study porous ultrathin silicon membranes. At the moment I'm taking a brief break from my research to be an American Academy for the Advancement of Science Mass Media Fellow at the Manhattan office of Scientific American.
Importance of science communication
I love science communication because it's both hard and important. People need to be told what scientists have discovered and what it means for their lives, but to do that well requires balancing the storytelling needs of journalism against objectivity and sober contextualization. Also, I personally find scientists to be generally fascinating people to write about and hear from.
Current projects
Along with my co-producer Madeline Sofia, I created The Bench Warmer's Podcast, which tells stories of misadventures and victories in science using interviews with current and former graduate students. I think the stories that don't often get told about science — the scoops, the failures, the dead ends, rewrites and rejections — are just as important to tell as the wild success stories. Not only that, but I think we short-sell our successes by not highlighting how rare they are. So, in the podcast, Maddie and I ask questions like "What's the most expensive thing you ever broke in lab?" and "Have you ever embarrassed yourself by dislocating your knee while singing karaoke onstage in front of hundreds of your scientific peers and possible future employers?"
I also write "10-cent stories" for children at the Rochester Museum and Science Center and at a few other places around Rochester. The children give me a prompt and in five minutes I use my typewriter to type them a tale. I've been doing this for about three years now, and I've written well over 800 stories. Sometimes the stories have a STEM bend to them, but sometimes they're just stories. I love this project for a lot of reasons, but most of all because it lets me make the world a stranger, more whimsical place.
Future plans
I've only been at my fellowship for a few weeks, so I'm still deciding if I want to be a science journalist or if my plans lie elsewhere. This is a time of great flux for me, so I don't know yet where I'm heading. But I'm enjoying figuring it out.
Rebecca Lowery Defends Thesis
Thursday, July 7, 2016

Rebecca Lowery has successfully defended her thesis, "The Role of Microglia and Fractalkine Signaling in Experience-dependent Synaptic Plasticity". Congratulate her when you see her.
Congratulations Dr. Lowery!
David Yule appointed Louis C. Lasagna Professor
Tuesday, July 5, 2016

David Yule
David Yule, professor of pharmacology and physiology, has been appointed the Louis C. Lasagna Professor in Experimental Therapeutics for five years, effective July 1. He retains his joint appointments as professor of medicine and as professor in the Center for Oral Biology.
For the past 15 years, Yule has studied calcium’s role in disorders in which calcium signaling and secretions are disrupted, such as Sjögren’s syndrome—in which patients experience dry mouth due to a lack of saliva—and acute pancreatitis.
Using state-of-the art imaging and electrophysiological techniques, Yule’s lab monitors calcium signals to achieve a better understanding of the mechanisms that underlie these signals with the goal that the studies will give insight into the control of important physiological processes in both normal physiology and disease states.
Yule received his PhD in physiology from the University of Liverpool in the United Kingdom. His research has been published in numerous journals, including the Journal of Biological Chemistry, Proceedings of the National Academy of Sciences, Science Signaling, and the Journal of Physiology. Yule serves on the editorial board for Gastroenterology, the preeminent journal in the field of gastrointestinal disease.
The Lasagna professorship honors Louis Lasagna, who served as chair of the Department for Pharmacology from 1970 to 1983, and brought the department to national recognition as a center of training and research. Lasagna, who was known for pioneering the study of placebos and writing an alternative Hippocratic oath, died in 2003.
URMC Team Revises Understanding of Genetic Code
Friday, July 1, 2016

Beth Grayhack, Ph.D., with lab
members and grad students
Christina Brule and Jiyu Wang
Scientists for years have known that the genetic code found in all living things contains many layers of complexity. But new research from the University of Rochester cracks the code more deeply, clarifying for example why some genes are inefficiently translated into proteins.
In a study published in the journal Cell, the researchers, co-led by Beth Grayhack, Ph.D., of the UR School of Medicine and Dentistry, discovered the existence and identity of 17 pairs of inefficient codons (DNA nucleotides or bases) within the genetic code.
Scientists have generally considered each piece of the genetic code (or codon) as a single “word” in a language. But the new data suggests some codon combinations act as compound words or phrases whose order and pairing has a significant impact on the translation of genes into proteins.
“Consider the words ‘pancake’ versus ‘cake pan,’ “ said Grayhack, an associate professor of Biochemistry and Biophysics, Pediatrics, and Cancer, in the Center for RNA Biology, at the UR Medical Center.
The Sleep Hack Neuroscience Says Gives Your Brain Optimal Rest
Thursday, June 30, 2016
Sleep is critical for rest and rejuvenation. A human being will actually die of sleep deprivation before starvation--it takes about two weeks to starve, but only 10 days to die if you go without sleep.
The CDC has also classified insufficient sleep as a public health concern. Those who don't get enough sleep are more likely to suffer from chronic diseases that include hypertension, diabetes, depression, obesity, and cancer.
It's thus vital to get enough shuteye, but it turns out your sleep position also has a significant impact on the quality of rest you get.
Now, a neuroscience study suggests that of all sleep positions, one is most helpful when it comes to efficiently cleaning out waste from the brain: sleeping on your side.
The study, published in the Journal of Neuroscience, used dynamic contrast-enhanced MRI to image the brain's "glymphatic pathway." This is the system by which cerebrospinal fluid filters through the brain and swaps with interstitial fluid (the fluid around all other cells in the body).
"It is interesting that the lateral [side] sleep position is already the most popular in humans and most animals--even in the wild," said University of Rochester's Maiken Nedergaard. "It appears that we have adapted the lateral sleep position to most efficiently clear our brain of the metabolic waste products that build up while we are awake."
6th Annual Stem Cell and Regenerative Medicine Symposium
Monday, June 27, 2016

Dr. Jack Kessler
In celebration of the NYSTEM-funded training program in stem cell biology at the University of Rochester, researchers convened for a day of presentations and discussions on advances in stem cell biology. To emphasize the excellence of our junior scientists, five NYSTEM trainees (both pre- and post-doctoral, took turns with leaders in the field of stem cell medicine to present their work. The meeting kicked off with a presentation by Dr. Jack Kessler, Northwestern University Feinberg School of Medicine) describing the factors controlling adult neural stem cell maintenance -- a key determinant of cognitive health.

Dr. Kunle Odusi

Dr. Angela Christiano (center)
Dr. Kunle Odunsi (Roswell Park Cancer Institute) spoke in his role as director of the immune-therapy program on the importance of gene-engineered, tumor recognizing CD4 T-cells in anti-tumor therapy.
Dr. Angela Christiano (Columbia University) provided an impressive example of the power of iPSC technology with the development of 3D-skin tissue for treatment of such devastating skin diseases as epidermolysis bulbosa.

NYSTEM Trainees
Presentations by NYSTEM trainees Fanju Meng (Biteau lab), Wenxuan Liu (Chakkalakal Lab), Michael Rudy (Mayer-Proschel Lab), Dr. Andrew Campbell (Proschel Labs), and Dr. Nicole Scott (Noble Lab) rounded out a day full of exciting new work that highlights the broad impact of stem cell biology on medicine today -- and the success of the SCRMI training program. The meeting was buoyed by good vibes and food provided by the backdrop of the Rochester International Jazz Festival.
Congratulations To This Year's Poster Prize Winners
Graduate Student Category
- Zhonghe Ke, High Levels of Niche Ha of the NMR Mediates the Maintenance of LT-HSC by reducing ROS Levels, Gorbunova Lab
- Jayme Olsen, Generation of Human Erythroblasts with Increased EX Vivo Self-Renewal, Palis Lab
- Michael Trembley, Novel Mechanisms of the Epicardial-Derived Cell Mobilization, Small Lab
Postdoctoral Category
- Pearl Quijada, Novel Mechanisms of Epicardium Dependent Cardiac Repair, Small Lab
Thank you to all participants for a great event. See you again in 2017!
Review: Giving Gene Editing Technology CRISPR-Cas9 a Boost
Thursday, June 23, 2016
A new gene editing technology called CRISPR-Cas9 has taken the scientific world by storm. It allows researchers to quickly and easily make changes to the DNA of humans, animals and plants. The hope is that CRISPR-Cas9 may be used in the future to eliminate or correct faulty genes that cause disease.
In a recent issue of the journal Cell, Lynne E. Maquat, Ph.D. and Maximilian W. Popp, Ph.D. of the University of Rochester Center for RNA Biology describe how scientists can make this technology more efficient. Understanding the principles of nonsense-mediated mRNA decay (NMD), a cellular mechanism that Maquat discovered early in her career, will help anyone employing the technology achieve a better result -- namely, a more complete knock out or deletion of a desired gene.
Catherine Ovitt receives 2016 IADR Innovation in Oral Care Award
Wednesday, June 22, 2016
Catherine Ovitt is one of this year’s three recipients of the 2016 IADR Innovation in Oral Care Awards. She accepted the award from IADR President Dr. Marc Heft at the IADR/APR General Session & Exhibition in Seoul, Republic of Korea. The three prestigious awards recognize research in innovative oral care technologies that may maintain and improve oral health, and are supported by GlaxoSmithKline.
Elissa Wong receives Neuman Scholarship Award
Tuesday, June 21, 2016

Elissa Wong, a fifth year toxicology graduate student in Ania Majewska’s lab, received the Margaret and William F. Neuman Scholarship Award in Environmental Medicine for exemplary scholarship and citizenship. Dr. William Neuman was the chair of the Department of Radiation Biology and Biophysics for many years and helped to create the Toxicology Training Program and the Environmental Health Science Center. Dr. Margaret Neuman received her PhD in Biochemistry from the University of Rochester. Later, working here, she researched the effects of uranium on bone biochemistry, and was an expert on the regulation of bone minerals.
The criteria for receiving this are as follows: 1) scholarship, 2) scientific excellence, 3) productivity, and 4) exceptional citizenship to the field of toxicology.
Congratulations Elissa!
Claire McCarthy Wins Travel Award
Monday, June 13, 2016
Congratulations to Claire McCarthy, the newest recipient of the Medical Faculty Council UR-SMD Trainee/Student Travel Award for Spring 2016.
Clara Kielkopf Receives EvansMDS Discovery Research Grant
Saturday, June 11, 2016
Biochemistry & Biophysics Associate Professor, Clara Kielkopf's project, entitled, Structural mechanisms and targeting of MOS-relevant pre-mRNA splicing factors has been selected by EvansMDS for funding for 2016. This year EvansMDS requested 12 full DRG proposals and were able to fund 6 of them. Their hope is that these findings will translate into improvements in therapy that can be delivered to MDS patients.
The Kielkopf lab investigates splicing defects in hematologic malignancies; roles of human pre-mRNA splicing factors in HIV-1 infectivity; development of engineered splicing factors for correction of splicing defects and splice sites and their associated proteins as therapeutic targets.
Mallory Scott Selected for Summer Internship at Bayer Pharmaceuticals
Friday, June 10, 2016
Mallory Scott, Biophysics, Structural and Computational Biology PhD student, in the laboratory of Dr. Paul Kammermeier, has been selected for a summer internship at Bayer Pharmaceuticals in Whippany, NJ in Global Regulatory Affairs. Mallory will gain valuable work experience in the healthcare industry while working with regulatory professionals on various projects to learn about the role of regulatory affairs in drug development and product registration as well as the regulatory landscape. Mallory will be working in the Chemistry, Manufacturing and Controls (CMC) division. Her project is focused on quality by design in continuous manufacturing.
Swapping Sick for Healthy Brain Cells Slows Huntington’s Disease
Tuesday, June 7, 2016

Researchers have successfully reduced the symptoms and slowed the progression of Huntington’s disease in mice using healthy human brain cells. The findings, which were published today in the journal Nature Communications, could ultimately point to a new method to treat the disease.
The research entailed implanting the animals with human glia cells derived from stem cells. One of the roles of glia, an important support cell found in the brain, is to tend to the health of neurons and the study’s findings show that replacing sick mouse glia with healthy human cells blunted the progress of the disease and rescued nerve cells at risk of death.
“The role that glia cells play in the progression of Huntington’s disease has never really been explored,” said Steve Goldman, M.D., Ph.D., co-director of the University of Rochester Center for Translational Neuromedicine. “This study shows that these cells are not only important actors in the disease, but may also hold the key to new treatment strategies.”
Harold Smith Publishes Commentary on RNA and DNA Editing
Sunday, June 5, 2016
Epigenetics is a popular, yet still mysterious concept in health and medicine. It’s the study of a variety of biological processes that alter the expression of our genes. Sometimes this involves modifying the structure of our chromosomes to mask or unmask genes, and other times the actual genetic code is changed in certain cells. Harold C. Smith, Ph.D., a longtime professor of Biochemistry and Biophysics at the University of Rochester School of Medicine and Dentistry has studied epigenetics in a research focus known as RNA and DNA editing since it was introduced two decades ago. He was invited to write a commentary on the progress and future of this research, published today in Trends in Biochemical Sciences, and answers a few questions about the subject.
Post-doctoral Fellow wins the 2016 Weiss Toxicology Scholar Award
Wednesday, June 1, 2016
Dr. Luisa Caetano-Davies (Biomedical Genetics) was the postdoctoral winner of the third annual Weiss Toxicology Award. The award was created to strengthen training and research in the Toxicology Training Program by enhancing support of talented future leaders in the field of toxicology, particularly those with an interest in neurotoxicology. The award is presented annually to a meritorious trainee with an interest in Neurotoxicology. Dr. Caetano-Davies is member of the Proschel lab and is studying the effects of environmental toxicants on early stages of Parkinson Disease pathology, in particular with a focus on astrocyte dysfunction. Carolyn Klocke (Cory-Schlechta Lab) was the winner of the graduate student category. Congratulations!
2016 Graduating Class
Wednesday, June 1, 2016
We had 22 total graduates this year: 1 Epidemiology PHD, 17 Masters’ in Public Health, 3 Masters’ in Clinical Investigation and 1 Masters in Health Services Research and Policy. Congratulations to all of our graduates!
Dr. Michael Nussenzweig Gives Melville A. Hare Lecture
Monday, May 30, 2016
 Michael Nussenzweig, M.D., Ph.D. gave the Melville Hare Memorial Lecture on May 12. The lecture, "The HIV Vaccine Problem" was organized by the students of the Department of Microbiology and Immunology and co-sponsored by the University of Rochester Center for AIDS Research.
Michael Nussenzweig, M.D., Ph.D. gave the Melville Hare Memorial Lecture on May 12. The lecture, "The HIV Vaccine Problem" was organized by the students of the Department of Microbiology and Immunology and co-sponsored by the University of Rochester Center for AIDS Research.
GDSC Graduate Nirmalya Chatterjee reports a novel role of Bet proteins in the control of the oxidative stress response pathway.
Friday, May 27, 2016
Bet proteins are a subclass of bromodomain containing epigenetic "readers". These proteins have complex and incompletely understood functions in the control of gene expression and chromatin organization. The human Bet proteins Brd3 and Brd4 have been implicated in cancer and thanks to the availability of specific inhibitors, have emerged as promising drug targets. The paper by Nirmalya Chatterjee, Min Tian and others describes experiments in Drosophila that discovered a novel function for Bet proteins: the regulation of the transcription factor Nrf2. The reported data show that a Drosophila Bet protein is part of a previously unknown pathway that can control Nrf2 activity. This is of interest as Nrf2 plays a prominent role in the defense against oxidative stress, protection against various diseases, and aging. Nirmalya Chatterjee, a recent member of the Bohmann Lab, received the PhD last September and is currently working as a postdoc in the group of Norbert Perrimon at Harvard Medical School.
Nirmalya Chatterjee2, Min Tian3, M., Kerstin Spirohn, Michael Boutros & Dirk Bohmann (2016) Keap1-Independent Regulation of Nrf2 Activity by Protein Acetylation and a BET Bromodomain Protein, PLoS Genetics, will go to press 5/27/2016. PMID: 27233051
Congratulations to all of our Retreat awards winners
Thursday, May 26, 2016
The Toxicology program would like to congratulate the following on their 2016 Retreat awards:
- Elissa Wong won the William F. and Margaret W. Neuman Award for exemplary scholarship and citizenship in the Toxicology Training Program
- Amanda Croasdell won the (student) Robert F. Infurna Award for publishing the best research paper in toxicology (this award was started in 1998)
- Lisbeth Boule won the (postdoc) Robert F. Infurna Award for publishing the best research paper in toxicology (this award was started in 1998)
- Carolyn Klocke won the (student) Weiss Toxicology Scholar Award
- Luisa Caetano-Davies won the (Postdoc) Weiss Toxicology Scholar Award
Luisa Caetano-Davies wins “Best Oral Presentation” Award.
Thursday, May 26, 2016

Luisa Caetano-Davies
Luisa, a post-doctoral fellow in the
Proschel Lab, received the award for her presentation on “Astrocyte dysfunction in Parkinson Disease” at the 2016 Environmental Medicine and Toxicology Training Program retreat. Her presentation described the use of both iPSC-based disease-in-a-dish and in vivo animal models to identify early astrocyte defects in PD disease etiology. Congratulations, Luisa!
Congratulations Karl Smith, AAAS Mass Media Fellowship Recipient
Monday, May 23, 2016
Karl Smith, 5th year graduate student in the Biophysics Structural and Computational Biology PhD program, laboratory of Dr. Jim McGrath, has received an AAAS (American Association for the Advancement of Science) Mass Media Fellowship. Karl, sponsored by the American Physical Society, will be spending 10 weeks this summer working at Scientific American in their Manhattan offices as part of his fellowship.
Since its inception, the AAAS Fellowship Program has supported more than 625 student scientists, engineers and medical professionals who, in some cases, produced the only original science-news reporting at their assigned media outlets over the summer. The current 15 fellows, selected from a pool of 130 outstanding applicants, are likely to generate between 200 and 300 original science stories for print articles, blogs, podcasts, radio segments, and multimedia features.
Past participants in the Mass Media Fellows program include Mark Dumont, Professor, Department of Biochemistry and Biophysics, who received the AAAS Mass Media Fellowship in 1975, the second year that it was in existence. Over that summer, he wrote 26 news articles for the San Diego Union.
Other recipients include Erica Goode and Kenneth Chang of the New York Times; Richard Harris, David Kestenbaum, and Joe Palca of NPR; renowned biologist Eric Lander, co-chair of U.S. President Barack Obama's Council of Advisors on Science and Technology; physician and "Law & Order: Special Victims Unit" Executive Producer Neal Baer; Victoria Bruce, author of No Apparent Danger: The True Story of Volcanic Disaster at Galeras and Nevado Del Ruiz; and many others. - from AAAS's website.
GDSC Student Xuan Li publishes on the role of Cdk12 in response to stress.
Monday, May 23, 2016

Xuan Li
The phosphorylation of RNA polymerase II in the C-terminal domain, or CTD, is an essential step for the transcription of all eukaryotic protein coding genes. The paper be Xuan Li and colleagues describes the unexpected discovery that a certain CTD kinase, called CDK12, is not universally required, but is only needed for the transcription of genes that are inducible by stress, such as heat, DNA damage or reactive oxygen species. This finding suggests that CTD phosphorylation plays a role in the regulation of specific gene expression programs, rather than being a generic step of transcription. This work involved a large-scale robotic RNAi screen in collaboration with the Boutros lab in Heidelberg, as well as genetic and biochemical experiments in the Drosophila model system. Xuan Li, a graduate student in the Bohmann Lab is currently doing an internship at Takeda Pharmaceuticals in Boston and will defend her PhD in November.
Xuan Li1, Nirmalya Chatterjee2,, Kerstin Spirohn, Michael Boutros & Dirk Bohmann (2016) Cdk12 Is A Gene-Selective RNA Polymerase II Kinase That Regulates a Subset of the Transcriptome, Including Nrf2 Target Genes. Scientific Reports, 6:21455. PMID: 26911346
Proposal by Amy Kiernan Receives University Research Award
Monday, May 23, 2016
A collaborative project involving Associate Professor Amy Kiernan of the Flaum Eye Institute has been chosen as one of the 2016-17 University Research Awards. One of just eight applications chosen by senior research leadership, the proposal entitled, "Understanding cell turnover and injury recovery in the corneal endothelium" will be funded $75.000 annually.
Conventional Radiation Therapy May Not Protect Healthy Brain Cells
Wednesday, May 18, 2016
A new study shows that repeated radiation therapy used to target tumors in the brain may not be as safe to healthy brain cells as previously assumed. The findings, which appear in the International Journal of Radiation Oncology, Biology, Physics, show that the treatment also kills important support cells in the brain and may cause as much, if not more damage, than single dose radiation therapy.
“This study suggests that conventional repeated radiation treatments offer no significant benefit to brain tumor patients,” said Kerry O’Banion, M.D., Ph.D., a professor in the University of Rochester Medical Center (URMC) Department of Neuroscience and lead author of the study. “It also shows that certain cell populations in the brain are vulnerable to radiation and this may help explain why so many brain cancer patients experience cognitive problems after treatment.”
Karl Smith places third in University’s Falling Walls Competition
Wednesday, May 18, 2016
Karl Smith, a PhD student in Biophysics and a member of the lab of James McGrath, Professor of Biomedical Engineering, won third place in the University of Rochester's Falling Walls Competition for describing his use of physics to make water behind a filter form a mixer vortex, reducing the difficulty of normal stirring when fluids stick to surfaces. A total of 19 presenters competed.
The competition is associated with the Falling Walls foundation, a non-profit organization that fosters discussions on research and innovation and promotes the latest scientific findings to society. The Rochester winner's idea will compete with others from around the world at the Falling Walls Lab Finale in November in Berlin. This event selects the participants for the annual Falling Walls Conference the following day: an international forum for science and innovation to commemorate the fall of the Berlin Wall. Speakers at the conference have included Angela Merkel, Chancellor of Germany; Nobel Prize winner Sir Paul Nurse; and young inventors from around the world. BBC London said it was where the "brightest minds on the planet" meet.
Last year's Falling Walls Lab Rochester winner, Ryan Trombetta, a BME PhD student in Dr. Awad's lab, finished 12th (out off a 100 finalists worldwide) in the Berlin competition for his description of using 3D printed bone grafts to treat osteomyelitis. See his presentation here.

From left to right, Solomon Abiola, Sara Nowacki and Karl Smith, the top three finishers at the Falling Walls Competition.
Cindy (Xiaowen) Wang in the Noble Lab wins 2016 GSS Poster Prize
Tuesday, May 17, 2016
 Cindy (Xiaowen) Wang in the Noble Lab wins 2016 GSS Poster Prize with her work on:
Identifying c-Cbl as a critical point of intervention in acquired tamoxifen resistant breast cancer.
(Co-authors Jennifer L Stripay, Hsing-Yu Chen and Mark D Noble).
Cindy (Xiaowen) Wang in the Noble Lab wins 2016 GSS Poster Prize with her work on:
Identifying c-Cbl as a critical point of intervention in acquired tamoxifen resistant breast cancer.
(Co-authors Jennifer L Stripay, Hsing-Yu Chen and Mark D Noble).
Garry Coles wins 2016 Vincent du Vigneaud Award For Excellence in Graduate Research
Tuesday, May 17, 2016
 Garry Coles, graduate of the Genetics, Development and Stem Cell program received this years du Vigneaud commencement award. The University of Rochester School of Medicine and Dentistry recognizes outstanding post-baccalaureate research efforts and promising PhD candidates through the Vincent du Vigneaud Award, in honor of Vincent du Vigneaud, himself a PhD graduate of the University of Rochester and recipient of the 1955 Nobel Prize in Chemistry.
Garry Coles, graduate of the Genetics, Development and Stem Cell program received this years du Vigneaud commencement award. The University of Rochester School of Medicine and Dentistry recognizes outstanding post-baccalaureate research efforts and promising PhD candidates through the Vincent du Vigneaud Award, in honor of Vincent du Vigneaud, himself a PhD graduate of the University of Rochester and recipient of the 1955 Nobel Prize in Chemistry.
Gary's PhD thesis, entitled "KIF7 and microtubule dynamics function to regulate cellular proliferation and cell cycle progression" focuses on deciphering the role of Kinesin family member 7 (Kif7) on cell cycle control during mammalian development. The work was conducted in Dr. Kate Ackerman's laboratory and has been published in the Proceedings of National Academy of Sciences (PNAS), PLoS Genetics and Developmental Biology.
Dr. Wellington Cardoso, Director for the Center for Human Development at Columbia University Medical Center, comments: "I have been closely following the work of Dr. Coles and his mentor Dr. Kate Ackerman, since we share a similar research interest. Dr. Coles has made important contributions to our understanding of the mechanisms regulating diaphragm and lung morphogenesis… I am confident that he will continue to make great contributions to the field in his future career."
This outlook is also shared by Dr. Hartmut Land, Chair of the Department for Biomedical Genetics and Director of Research at the Wilmot Cancer Center: "Garry is an incredibly driven and inquisitive scientist, and he has a fabulous enthusiasm for his work…(He) has grown tremendously during his time in graduate school. His maturity and independence are ahead of the curve for most post-doctoral fellows." Dr. Land concludes, "Given (Garry's) exceptional talent to make things work, his curiosity and great persistence, I am certain that he will contribute significantly to any scientific environment... (and)… become a leader in his field".
Class of 2014 Prelim season begins
Thursday, May 12, 2016
On Friday, May 6th, Andrew Albee opened the 2016 season of Prelim Exams. According to his committee, Andrew passed his qualifying exam with flying colors, and the committee looks forward to the outcome of his work. His studies on the function of Lmx Homeobox transcription factors in early somatic progenitors of the Drosophila ovary are also the basis of an F31 application submitted in February of this year. Congratulations, Andrew!
28th Annual Genetics Day Meeting
Wednesday, May 11, 2016

Dr. Michael Levine
This year's Genetics Day provided another opportunity to celebrate the impact of Genetics on science and medicine. An excellent selection of speakers from the University of Rochester Medical Center highlighted the importance of diverse genetic mechanisms ranging from chromatin remodeling in erythropoesis (Laurie Steiner) and DNA damage repair (Xi Bin) to translational control by riboswitches (Joe Wedekind) and di-codon usage (Elizbeth Grayhack). Genetics Day concluded with the Fred Sherman lecture by Dr. Michael Levine (Princeton University). His presentation on visualizing the mechanisms of transcriptional enhancers was equally entertaining and insightful. Originally from the Hollywood area, and by his own admission a closet movie producer, Dr. Levine wowed audiences with in vivo movies of enhancer reporters, shedding new light on what we all thought was an established principle of molecular genetics.
Grad student
-
Manisha Taya – Hammes Lab
The Role of Estrogen Signaling in a Mouse Model for Lymphangioleiomyomatosis (Lam)
-
Sam Carrell – Thornton Lab
Silencing of Myotonic Dystrophy Protein Kinase (Dmpk) Does Not Affect Cardiac or Muscle Function In Mice
Post Docs
-
Walter Knight – Yan Lab
The Role and Mechanism of Cyclic Nucleotide Phosphodiesterase 1c in Regulating Pathological Cardiac Remodeling and Dysfunction
-
Vincent Martinson - Jaenike lab
Gut Microbiota of Distantly Related Drosophila Species Share Similar Bacterial Diversity
Genetics Day has been a long standing tradition at the University of Rochester And more recently includes the Fred Sherman lecture in memory of Fred Sherman a renowned biochemist and geneticist, who led international efforts to establish the yeast Saccharomyces cerevisiae as the premier genetic eukaryotic model system. The lecture is made possible by a generous fund endowed by Fred Sherman's wife, Elena Rustchenko-Bulgac, herself a research professor at the URMC.
When the Physical World is Unreliable: Study Finds Visual and Tactile Processing Deficits in Schizophrenia
Tuesday, May 10, 2016

A new study out today in the journal Translational Psychiatry sheds further light on the idea that schizophrenia is a sensory disorder and that individuals with the condition are impaired in their ability to process stimuli from the outside world. The findings may also point to a new way to identify the disease at an early stage and before symptoms become acute.
Because one of the hallmarks of the disease is auditory hallucinations, such as hearing voices, researchers have long suspected a link between auditory processing and schizophrenia. The new study provides evidence that the filtering of incoming visual information, and also of simple touch inputs, is also severely compromised in individuals with the condition.
“When we think about schizophrenia, the first things that come to mind are the paranoia, the delusions, the disorganized thinking,” said John Foxe, Ph.D., the chair of the University of Rochester Medical Center Department of Neuroscience and senior author of the study. “But there is increasing evidence that there is something fundamentally wrong with the way these patients hear, the way they feel things through their sense of touch, and in the way in which they see the environment.”
Denise Skrombolas Receives Award
Thursday, May 5, 2016
Dr. Denise Skrombolas was awarded the Rochester Vaccine Fellowship created by a donation from Dr. Michael Pichichero in honor of Dr. Porter Anderson one of the pioneers in the Hib vaccine.
Jennifer Judge Wins the University of Rochester’s Three Minute Thesis (3MT) Competition
Wednesday, May 4, 2016

Jennifer Judge presenting at the 3MT competition
Jennifer Judge, a Toxicology graduate student in the Sime Lab, has won the Judge's Vote and People's Choice Award at the University of Rochester's Three Minute Thesis (3MT) competition. The event was held today in the Class of '62 auditorium, as 8 finalists delivered their research in only three minutes.
The judges will picked a winner ($1,000 in travel funds), then the students voted for whom they thought should receive the People's Choice
award ($500 in travel funds!). Congrats to Jennifer for winning both!
SA Government names Professors of the Year
Monday, May 2, 2016

Join us in congratulating Laurel for being selected as one of 4 Professors of the year from a extraordinary field of 63 candidates.
Laurel Carney, professor of biomedical engineering, won in the Engineering field. Her research focuses on the complex network of auditory nerve fibers that transmit the inner ear’s electrical signals to the brain with the goal of better hearing aids.
Carney earned her M.S. and Ph.D degrees in electrical engineering at the University of Wisconsin-Madison. She was an associate professor of biomedical engineering at Boston University and professor of biomedical engineering at Syracuse University before joining the Rochester faulty in 2007. She serves as professor in three departments – biomedical engineering, neurobiology and anatomy, and electrical and computer engineering.
Elissa Wong Awarded Individual Pre-doctoral Fellowship from NIAAA
Sunday, May 1, 2016

Elissa Wong, a 4th year Toxicology Graduate Program student in Dr. Ania Majewska's lab received a perfect 10 review score and was awarded an NIH (NRSA) Individual Pre-doctoral Fellowship from the NIAAA. The title of her project is: Synaptic plasticity and microglial-synapse interactions after developmental alcohol exposure (2016-2018).
Congrats Elissa!
NGP Graduate Alum, Grayson Sipe, Wins Doty Thesis Award
Friday, April 29, 2016

Grayson Sipe, recent doctoral graduate from the Majewska lab, received the Robert Doty prize for the 2016 outstanding dissertation in neuroscience. The Doty prize is named in the honor of longtime faculty member Robert Doty, who made great contributions to neuroscience research at the University of Rochester and nationally. It is awarded on the basis of the impact and importance of research, novelty of experimental design, independence and creativity of the student and research implications and relevance for neuroscience. Grayson’s thesis entitled “The Role of P2Y12 in non-pathological microglial functions during synaptic plasticity”, which he successfully defended on February 19th, 2016, embodied all these characteristics. Grayson has now moved to his postdoctoral position with Dr. Mriganka Sur at MIT. Dr. Peter Shrager presented Grayson the prize at the annual neuroscience retreat on Friday, April 29th.
Congratulations Grayson!!!
Subtle Chemical Changes in Brain Can Alter Sleep-Wake Cycle
Friday, April 29, 2016

A study out today in the journal Science sheds new light on the biological mechanisms that control the sleep-wake cycle. Specifically, it shows that a simple shift in the balance of chemicals found in the fluid that bathes and surrounds brain cells can alter the state of consciousness of animals.
The study, which focuses on a collection of ions that reside in the cerebral spinal fluid (CSF), found that not only do these changes play a key role in stimulating or dampening the activity of nerve cells, but they also appear to alter cell volume causing brain cells to shrink while we sleep, a process that facilitates the removal of waste.
"Understanding what drives arousal is essential to deciphering consciousness and the lack thereof during sleep," said Maiken Nedergaard, M.D., D.M.Sc., co-director of the University of Rochester Center for Translational Neuromedicine and lead author of the study. "We found that the transition from wakefulness to sleep is accompanied by a marked and sustained change in the concentration of key extracellular ions and the volume of the extracellular space."
The current scientific consensus is that the brain is "woken up" by a set of neurotransmitters -- which include compounds such as acetylcholine, hypocretin, histamine, serotonin, noradrenaline, and dopamine -- that originate from structures deep within the brain and the brain stem. This cocktail of chemical messengers serve to activate -- or arouse -- a set of neurons in the cerebral cortex and other parts of the brain responsible for memory, thinking, and learning, placing the brain in a state of wakefulness.
Heather Natola Wins 2016 Edward Peck Curtis Award for Excellence in Undergraduate Teaching
Thursday, April 28, 2016
 We are proud to announce that Heather Natola has been selected to receive the 2016 Edward Peck Curtis Awards for Excellence in Undergraduate Teaching. Ms. Natola received high praise from her students, faculty in the Department of Biomedical Genetics and Rochester Museum and Science Center.
We are proud to announce that Heather Natola has been selected to receive the 2016 Edward Peck Curtis Awards for Excellence in Undergraduate Teaching. Ms. Natola received high praise from her students, faculty in the Department of Biomedical Genetics and Rochester Museum and Science Center.
Ms. Natola is a graduate student researcher in the Pröschel Lab, where she investigates new therapeutic approaches to spinal cord injury as part of the UR Stem Cell and Regenerative Medicine Institute.
 "Ms. Natola was particularly instrumental in providing students with in-depth and detailed training, which had a significant positive impact on the student's engagement and learning"
"Ms. Natola was particularly instrumental in providing students with in-depth and detailed training, which had a significant positive impact on the student's engagement and learning"
-Hartmut Land, Ph.D., Chair, Department of Biomedical Genetics
"Despite her ambitious and demanding research work, Heather has volunteered for all of these teaching activities. Clearly she has not only become an ambassador for science as a whole, but has helped fulfill the mission of our school. What more can we ask of a graduate student?"
Christoph Proschel, Ph.D., Program Director - Genetics Development & Stem Cells Ph.D. Program
Heather is enthusiastic and committed to promoting interest in science and an attitude of life-long learning
-Kara Verno, Program Supervisor - Rochester Museum and Science Center
Community Talk on Zika Virus Features Infectious Disease Expert from Brazil
Tuesday, April 19, 2016
As the warm summer season approaches, the possibility of a Zika outbreak in the United States looms large. The greatest concern is for women of childbearing age, as studies continue to link exposure to the virus in pregnancy to serious birth defects like microcephaly, hearing loss and blindness.
Esper Kallas, M.D., Ph.D., an infectious diseases specialist and professor of Medicine at the University of Sao Paulo, Brazil will speak about Zika virus on Monday, May 9 at 7:30 pm in the Eisenhart Auditorium at the Rochester Museum and Science Center. The event is free and open to the public. Kallas will be joined by a panel of experts from the University of Rochester Medical Center:
Congratulations to NGP student Aleta Steevens
Friday, April 8, 2016

Aleta Stevens, an NGP student in Dr. Amy Kiernan's lab, secured a 3-year NIH Individual Pre-doctoral Fellowship, F31 entitled, "Elucidating the role of SOX2 in inner ear development."
Excellent work Aleta!
Harold Smith Inducted into Royal Society of Biology
Friday, April 8, 2016
Dr. Harold Smith, Professor of Biochemistry & Biophysics has been inducted into the Royal Society of Biology.
A long time member of the department, Dr. Smith's primary interest is understanding the composition, regulation and structure of macromolecular complexes involved in regulating gene expression at the level of messenger RNA expression and processing. The lab's focus is on a platform of enzymes that change the genetic code at the DNA or RNA level by deaminating cytidine to form uridine. Current data suggest that this family of cytidine deaminase function with other proteins (auxiliary proteins) as holoenzymes complexes which we refer to as editosomes (for RNA) or mutasomes (for DNA). RNA editing or DNA mutational activity by these enzymes affect the protein coding capacity of mRNAs and thereby can diversify the proteins that are expressed by cells (the proteome). Please visit the Smith Lab for more information. Dr. Smith has a 30 year track record of teaching and mentoring graduate students, medical students and undergraduates at the University of Rochester and has lead curriculum design and reform for these programs.
The Royal Society of Biology (RSB), previously called the Society of Biology, is a learned society in the United Kingdom created to advance the interests of biology in academia, industry, education, and research. Formed in 2009 by the merger of the Biosciences Federation and the Institute of Biology, the society has around 16,000 individual members, and over 100 member organizations. In addition to engaging the public on matters related to the life sciences, the society seeks to develop the profession and to guide the development of related policies.
Neuroscience Graduate Students Win Award for Teaching
Friday, April 8, 2016

Neuroscience Graduate Program students, Aleta Steevens (Dr. Amy Kiernan lab) and Heather Natola (Dr. Chris Pröschel lab) were awarded the 2016 Edward Peck Curtis Award for Excellence for Graduate Student Teaching.
Only a handful of these are awarded each year, and all this year's nominees were extremely well-qualified.
Congratulations to both!!!
Christina Cloninger Defends Thesis
Thursday, April 7, 2016
 Congratulations Dr. Cloninger on successfully defending your thesis!!
Congratulations Dr. Cloninger on successfully defending your thesis!!
“Honeycomb” of Nanotubes Could Boost Genetic Engineering
Wednesday, April 6, 2016

Electron microscope image of animal cells (colored blue) cultured on an array of carbon nanotubes
Researchers have developed a new and highly efficient method for gene transfer. The technique, which involves culturing and transfecting cells with genetic material on an array of carbon nanotubes, appears to overcome the limitations of other gene editing technologies.
The device, which is described in a study published today in the journal Small, is the product of a collaboration between researchers at the University of Rochester Medical Center (URMC) and the Rochester Institute of Technology (RIT).
“This platform holds the potential to make the gene transfer process more robust and decrease toxic effects, while increasing amount and diversity of genetic cargo we can deliver into cells,” said Ian Dickerson, Ph.D., an associate professor in the Department of Neuroscience at the URMC and co-author of the paper.
Study: The Science behind Bodily Secretions
Tuesday, April 5, 2016
The salivary gland secretes saliva that helps us chew and swallow the food we eat. The pancreas secretes digestive juices that enable our bodies to break down the fat, protein, and carbohydrates in the food. Secretions like these are important in countless activities that keep our bodies running day and night. A study published today in the journal Science Signaling uncovers a previously mysterious process that makes these secretions possible.
At the heart of the new study is calcium, which is present in all of our cells and is a gatekeeper of sorts: an increase in calcium in our cells opens up “gates” or “channels” that are required for the production and secretion of fluids like saliva. If calcium doesn’t increase inside cells the gates won’t open, a problem that occurs in diseases like Sjögren’s syndrome. Sjögren’s patients experience dry mouth due to a lack of saliva and have difficulty chewing, swallowing, and speaking, which severely hampers quality of life.
For the past 15 years David I. Yule, Ph.D., professor in the department of Pharmacology and Physiology at the University of Rochester School of Medicine and Dentistry has studied calcium’s role in Sjögren’s and other disorders in which calcium and secretions are disrupted, like acute pancreatitis. In the new study he answers an important question that has stumped scientists for years: what does it take for a particularly important calcium channel to open and start these processes?
Professor Harold Smith to Organize Meeting on Drug Discovery
Thursday, March 31, 2016
The Clinical Science and Drug Discovery Conference had its inaugural meeting in 2015 in Baltimore, MD where Dr. Smith was asked to serve as a Keynote Speaker (and judge for poster sessions). The organizers of that meeting nominated him to organize this years meeting in Dundee, Scotland along with Drs. Ian Catchpole from GlaxoSmithKline in the UK and Nikolai Zhelev, professor at Abertay University, the hosting institution. The meeting will be held July 27-29. Dr. Smith will also deliver a keynote lecture at this meeting and chair a special topics session that he is bringing together on 'Host Cell Factors as Therapeutic Targets'. For more information, please visit the Drug Discovery Summit site, see also the CSDD Brochure.
Q&A: Biologist earns raves for work with yeast
Wednesday, March 23, 2016
David Goldfarb, professor of biology and biochemistry, researches yeast as a model organism for understanding the aging process in humans. Goldfarb joined the Rochester faculty in 1988, five years after earning his PhD in biochemistry at the University of California, Davis, and completing postdoctoral work at Stanford University.
Goldfarb holds four patents and has been recognized with more than a dozen honors, including the Johnson & Johnson “Focused Giving Program” Award, the National Society of Collegiate Scholars Distinguished Member Award, and the March of Dimes Health Leadership Award in Education.
Early Wiring of Brain's “Fear” Centers Could Produce Long-term Consequences
Monday, March 21, 2016

New research shows that our brains may be hardwired to become sensitive to stressful environments at an early age and, if overstimulated, this may contribute to anxiety disorders and even psychotic syndromes later in life.
The study, which appears in the journal Brain Structure and Function, focuses on two structures deep in the brain. The central nucleus of the amygdala (Ce) is thought to be involved in responses to immediate threats and stimulus, such as becoming startled or freezing in reaction to a loud noise. The bed nucleus of the stria terminalis (BST) is thought to be involved in regulating a person’s state of vigilance, such as determining whether or not an environment or a situation poses a potential threat. Animal and human studies show that when the BST is activated by a threatening situation, we tend to slow down, become quieter, and stress hormones spike.
While Ce and BST reside in different parts of the brain, the two areas are hardwired to each other by axonal tracts – basically, bundles of long distance axon fibers that enable the separate regions to communicate with each other. However, until now it has not been clear when these connections form or the way in which they interact with each other.
In the study published today, a team of researchers led by Julie Fudge, M.D., with the Department of Neuroscience observed that these connections are made at a very early stage of development in non-human primates. They also found that the direction of the connection is essentially a one way street. The Ce – or immediate fear signaling center – conveys information to the BST, the structure that mediates general threat sensing or anxiety states. This arrangement suggests that repeated activation of the Ce by immediately fearful or traumatic events may shape long-term anxiety states in the BST.
Omega 3 Fatty Acids May Reduce Bacterial Lung Infections Associated with COPD
Tuesday, March 15, 2016
Compounds derived from omega-3 fatty acids -- like those found in salmon -- might be the key to helping the body combat lung infections, according to researchers at the University of Rochester School of Medicine and Dentistry.
The omega-3 derivatives were effective at clearing a type of bacteria called Nontypeable Haemophilus influenzae (NTHi), which often plagues people with inflammatory diseases like chronic obstructive pulmonary disease (COPD).
COPD, which is most often caused by years of smoking, is characterized by inflammation and excessive mucus in the lungs that blocks airflow. Quitting can slow the progress of COPD, but it doesn't halt the disease. Anti-inflammatory drugs are the most common treatment, however they suppress the immune system, which can put people with COPD at risk for secondary infections, most commonly NTHi bacterial infections.
"Our biggest concern with patients who have COPD is bacterial infections, which often put their lives at risk," says Richard Phipps, Ph.D. professor of Environmental Medicine and director of the URSMD Lung Biology and Disease Program. "If we can figure out how to predict who is likely to get an infection, physicians could put them on a preventative medication."
In his recent study, which was featured in the top ten percent of the March 15 issue of The Journal of Immunology, Phipps and lead author, Amanda Croasdell, a graduate student in the Toxicology program, tested the effectiveness of an inhalable omega-3 derivative to prevent NTHi lung infections in mice.
The Brain’s Gardeners: Immune Cells ‘Prune’ Connections Between Neurons
Monday, March 7, 2016
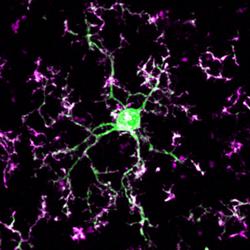
Microglia (green) with purple representing the P2Y12 receptor which the study shows is a critical regulator in the process of pruning connections between nerve cells.
A new study out today in the journal Nature Communications shows that cells normally associated with protecting the brain from infection and injury also play an important role in rewiring the connections between nerve cells. While this discovery sheds new light on the mechanics of neuroplasticity, it could also help explain diseases like autism spectrum disorders, schizophrenia, and dementia, which may arise when this process breaks down and connections between brain cells are not formed or removed correctly.
“We have long considered the reorganization of the brain’s network of connections as solely the domain of neurons,” said Ania Majewska, Ph.D., an associate professor in the Department of Neuroscience at the University of Rochester Medical Center (URMC) and senior author of the study. “These findings show that a precisely choreographed interaction between multiple cells types is necessary to carry out the formation and destruction of connections that allow proper signaling in the brain.”
The study is another example of a dramatic shift in scientists’ understanding of the role that the immune system, specifically cells called microglia, plays in maintaining brain function. Microglia have been long understood to be the sentinels of the central nervous system, patrolling the brain and spinal cord and springing into action to stamp out infections or gobble up dead cell tissue. However, scientists are now beginning to appreciate that, in addition to serving as the brain’s first line of defense, these cells also have a nurturing side, particularly as it relates to the connections between neurons.
The Future of Photonics
Tuesday, February 23, 2016
As hundreds of millions of dollar pour into Rochester to establish the nation's first Photonics Hub, Mark Gruba has a closer look at the technology in a News 8 special report, "The Future of Photonics."
Photonics is the science and technology of generating, controlling and detecting photons, which are particles of light. A display at the Rochester Museum & Science Center houses examples of its many applications. In one, a transmitter converts an audio signal from electrical pulses into light pulses. The laser beam sends that information to the receiver, which converts the light pulses back to electrical pulses and sends them to the speaker for your listening enjoyment.
"We work on optical bio sensors," said Dr. Ben Miller, a researcher at the University of Rochester Medical Center. He's creating a sensor that can detect the presence of hundreds of viruses from a single blood sample, in real time. "We're working to make devices so that you can immediately get that information in the doctor's office," said Dr. Miller.
Hope, Hype, and Wishful Thinking
Monday, February 22, 2016
In a perspective piece appearing in the journal Cell Stem Cell, URMC neurologist Steve Goldman, M.D., Ph.D., lays out the current state of affairs with respect to stem cell medicine and how close we are to new therapies for neurological disorders.
The dawn of stem cell medicine some 25 years ago was greeted with great enthusiasm, particularly by scientists who study diseases in the central nervous system (CNS). Many of the diseases found in the brain and spinal cord are degenerative in nature; meaning that over time populations of cells are lost due to genetic factors, infection, or injury. Because stem cell medicine holds the potential to repair or replace damaged or destroyed cells, scientists have considered these diseases as promising candidates for new therapies.
However, as with other emerging fields of medicine, the race to cures has turned out to be more of marathon than a sprint. While scientists have become very adept at manipulating stem and progenitor cells and understanding the complex choreography of genetic and chemical signals that instruct these cells to divide, differentiate, and proliferate, researchers are still grappling with the challenges of how to integrate new cells into the complex network of connections that comprise the human brain.
Goldman, co-director of the URMC Center for Translational Neuromedicine, takes a sweeping view of where we stand and which CNS diseases may or may not ultimately benefit from future stem cell-based therapies.
Congratulations Dr. Sipe!
Wednesday, February 17, 2016
Make sure you congratulate Grayson Sipe on defending his thesis.
Way to go Grayson!
Richard Aslin's Rochester Baby Lab Shows
Wednesday, February 17, 2016
Amelia Smith sits on the floor of a newly remodeled wing of the University of Rochester's department of Brain and Cognitive Sciences. The 8-month old wears a headband of cottony roses, and tiny bubbles form in the corner of her mouth. She's completely entranced by the commotion around her.
Though few adults in the room can resist oohing and aww-ing, little Amelia is not there to be fawned over. She's there to work. Researchers at the UR's Baby Lab want to know what she's thinking, what she's learned so far in her young life, and how she learned it.
But there's a problem: Amelia can't talk yet.
The work being investigated in Richard Aslin's Baby Lab was written up in the City Newspaper article "Signs of Intelligent Life".
Tracking Melanoma Metastasis Leads to Key Gene Discovery
Wednesday, February 17, 2016
A Wilmot Cancer Institute investigator discovered a gene that's required for the initiation of melanoma and the growth of disseminated melanoma cancer cells in the lungs.
The findings suggest that the gene's signaling pathway may be proof that melanoma stem cells exist, a question that's being debated by scientists.
Lei Xu, Ph.D., associate professor of Biomedical Genetics at the University of Rochester Medical Center, is lead author of the study, which was recently published in PLOS ONE and funded by a Wilmot Cancer Institute pilot grant. The Xu lab investigates the multiple, complex steps that occur as cancer cells spread from the original tumor to other parts of the body.
Report Recommends More Treatment, Research, for Gulf War Vets
Monday, February 15, 2016
The cause of Gulf War illness is still a mystery but focusing on treatments and interventions might help the veterans of Operation Desert Storm as well as the troops of the future, according to an Institute of Medicine committee report led by University of Rochester Medical Center Professor Deborah Cory-Slechta.
In 1990 and ’91 nearly 700,000 U.S. troops deployed to the Persian Gulf region for a short, intense war. Few injuries or deaths occurred, but troops were exposed to chemical and biological weapons, vaccines, oil-fires, air pollution, bomb blasts, pesticides, extreme desert temperatures, and constant false alarms and fear of nerve-gas attacks.
After the war ended a high number of the veterans reported debilitating fatigue, muscle and joint pain, headaches, and cognitive problems. This became known as “Gulf War illness.” During the past 25 years, 10 different committees of the nation’s top medical experts have searched for evidence that would better define Gulf War illness and possible treatments. The latest committee, headed by Cory-Slechta, concluded that no single mechanism can explain the multitude of symptoms seen in Gulf War illness—and that it’s unlikely a cause will ever be identified.
Doing something larger than you could ever do on your own
Friday, February 12, 2016
"There is a tendency for many investigators, especially early in their careers, to hold onto their work and not share it," says David Williams, the William G. Allyn Professor of Medical Optics; Dean for Research in Arts, Sciences and Engineering; Director of the Center for Visual Science - and a leading eye expert who pioneered the use of adaptive optics for vision correction.
"They don't realize - and it's one of the things that took me longer to learn than I wish it had - that one of the best ways to build your reputation is to share your ideas or your technology with the hope that they will be adopted.
"I was lucky enough to realize that if I let my students take my adaptive optics technology and use it to build their own labs, for example, it not only helped them get their independent research programs off the mark but also enhanced my reputation because so many more people were able to access and deploy the technology."
Is it any wonder then, that of the five NEI Audacious Goals grants recently awarded to Williams and four other investigators:
- four of the projects use adaptive optics as their core technology?
- three of the other PI's are either current collaborators with Williams or former postdocs in his lab?
- which means that four of the PI's will be cooperating with each other, even as they individually collaborate with other experts in the field on their individual projects - in effect widening the opportunities for synergy?
"That's the excitement of this," Williams says. "Why should we compete when one group can do one piece of it, and a second group can do another, and as along as you can manage authorships and credit appropriately and fairly, we can be much more efficient and effective in getting things done?"
"One of the things I'm proudest about in this community of people around the world doing adaptive optics and retinal imaging is that almost all of us get along really well, and we're moving science forward as rapidly as we can by helping each other. That doesn't always happen in science."
As Dean of Research for Arts, Science and Engineering, Williams is always looking for young faculty throughout AS&E who have the right personality and vision to take on larger, multi-investigator, multi-institutional projects.
"You have to be gregarious and interested in working with other people and tolerating the quirks that they have, just as they have to tolerate the quirks you have," Williams said.
"The largest source of optimism for me about the AS&E research portfolio is the quality of our junior faculty members - their enthusiasm and energy. Many of them have cut their teeth on individual investigator awards and will reach a certain point in mid career when they realize they need to reach out for complementary expertise in order to do more."
Williams' advice: The best collaborator may not be the first one that comes to mind.
"One of the biggest mistakes faculty members make is to choose a collaborator who is just like them, who has the same interests in a problem and the same background and who they can easily begin a conversation with because they are so closely aligned. But that doesn't really help your research. You want to have somebody who . . . has a completely different skills set. As obvious as that is, it doesn't always get factored into planning how to accumulate the necessary wisdom to do something larger than you could ever do on your own."
Scientists Seek to Improve Flu Vaccine for the Very Young
Thursday, February 11, 2016
Scientists at the University of Rochester School of Medicine and Dentistry have discovered a way to make a nasal spray flu vaccine safer for those who are at greatest risk of catching the flu, particularly infants under the age of 2. The work is early and a long way from being applied in people, but offers promise for a vaccine that could better protect the most vulnerable.
Study Sheds Light on Source of Drug Addicts' Risk-Taking Behavior
Wednesday, February 10, 2016
A study out today provides new insight into how the brains of drug addicts may be wired differently. The findings, which appear in the journal Psychopharmacology, show that while drug users have very strong motivation to seek out "rewards," they exhibit an impaired ability to adjust their behavior and are less fulfilled once they have achieved what they desire. Addressing this disconnect between the craving for a drug and the ability to regulate behavior may be one of the keys to breaking the cycle of addiction.
"The vast majority of people, when faced with something they want, will assess how achievable the goal is and adjust their actions and expectations in order to maximize their potential to achieve it," said John Foxe, PhD, the chair of the Department of Neuroscience at the University of Rochester Medical Center and senior author of the study. "However, it appears that the integrity of this system of assessment and self-regulation is impaired in substance abusers and this may contribute to the risk-taking behaviors and poor decision-making commonly associated with this population."
Lead poisoning still an issue in Rochester
Tuesday, February 9, 2016
The Michigan city of Flint became ground zero of the nation's latest public-health outrage when it was learned in recent months that its tap water contained unsafe levels of toxic lead.
Though the aqueous cause of its lead problem is unusual, Flint is otherwise far from unique. Many American cities, including Rochester, continue to struggle with lead poisoning, particularly of children.
In fact, despite years of successful anti-lead work locally, the proportion of children in Rochester found to have elevated levels of lead in their blood still was roughly double that of their counterparts in Flint in 2014, the most recent year for which comparable data are available.
We have had a huge amount of progress here. We’ve had a nearly 90 percent reduction in the number of kids with elevated blood lead levels in the past 15 years,
said Katrina Smith Korfmacher, an associate professor of environmental medicine at the University of Rochester Medical Center who has long been involved in local anti-lead efforts. But I would say it’s still a serious, or an ongoing, problem. The point is, it won’t ever go away entirely, because there is lead in the environment.
Scientists Discover Stem Cells Capable of Repairing Skull, Face Bones
Monday, February 1, 2016
The photo shows a blue-stained stem cell and a red-stained stem cell that each generated new bones cells after transplantation.
A team of Rochester scientists has, for the first time, identified and isolated a stem cell population capable of skull formation and craniofacial bone repair in mice—achieving an important step toward using stem cells for bone reconstruction of the face and head in the future, according to a new paper in Nature Communications.
Senior author Wei Hsu, Ph.D., dean's professor of Biomedical Genetics and a scientist at the Eastman Institute for Oral Health at the University of Rochester Medical Center, said the goal is to better understand and find stem-cell therapy for a condition known as craniosynostosis, a skull deformity in infants. Craniosynostosis often leads to developmental delays and life-threatening elevated pressure in the brain.
Hsu believes his findings contribute to an emerging field involving tissue engineering that uses stem cells and other materials to invent superior ways to replace damaged craniofacial bones in humans due to congenital disease, trauma, or cancer surgery.
For years Hsu's lab, including the study's lead author, Takamitsu Maruyama, Ph.D., focused on the function of the Axin2 gene and a mutation that causes craniosynostosis in mice. Because of a unique expression pattern of the Axin2 gene in the skull, the lab then began investigating the activity of Axin2-expressing cells and their role in bone formation, repair and regeneration. Their latest evidence shows that stem cells central to skull formation are contained within Axin2 cell populations, comprising about 1 percent—and that the lab tests used to uncover the skeletal stem cells might also be useful to find bone diseases caused by stem cell abnormalities.
The team also confirmed that this population of stem cells is unique to bones of the head, and that separate and distinct stem cells are responsible for formation of long bones in the legs and other parts of the body, for example.
The National Institutes of Health and NYSTEM funded the research.
Strong Star Certificate of Appreciation Awarded to Dr. Jermaine “JJ" Jenkins
Tuesday, January 26, 2016
On January 22, 2016, Dr. Jermaine Jenkins, who runs the Structural Biology and Biophysics Facility, was nominated for going 'above and beyond' in his work for a client of the facility. The client commented in their nomination:
The clinical trials group had a very tight timeline to meet for one of our clients. Testing had to be completed by the 25th of January so the client could present the data to the FDA. JJ was aware of the required quick turnaround time and he met the challenge. He worked the weekend so that our client's needs would be met. It is so impressive to work with such a dedicated scientist who takes his job so seriously. With JJ's help, URMC Labs Clinical Trials group made a very good impression on a client.
As Facility Manager of the Structural Biology and Biophysics Facility, Dr. Jenkins offers support services to determine macromolecular x-ray crystal structures, and to investigate protein-protein, protein-nucleic acid or protein-small molecule interactions. Professor Clara Kielkopf – a long time user and co-founder of the Facility commented, JJ quickly, calmly and reliably responds to user needs
. Co-director of the Facility Professor Joseph Wedekind added, Dr. Jenkins is an outstanding and dedicated scientist. We are fortunate to have such a great colleague
. Please join us in expressing your gratitude to JJ for his service and dedication.
CTSI Trainee Pilot supports better understanding of lupus
Friday, January 15, 2016
Lupus is a devastating disease that affects around 1 in 2,000 people in the U.S., and involves chronic inflammation and tissue damage in various organs including the skin, kidneys, and joints. Although the mortality rate for lupus has improved in recent decades, a diagnosis of lupus often means elevated risk of early mortality and lifetime of immunosuppressive therapy, which can carry significant side effects.
The Scientist as Storyteller
Friday, January 15, 2016
Graduate students Clarence Ling (left), Jon Baker,
and Karl Smith rehearse a script for The Bootleggers
at the WRUR studios in Todd Union. (Photo: Adam Fenster)
For Karl Smith, the storytelling bug began with a Montgomery Ward No. 22 typewriter purchased for $5 at a moving sale.
Typewriter perched on his lap, the doctoral student in biophysics has become a fixture at the Rochester Public Market, Corn Hill Arts Festival, and other Rochester-area arts-oriented venues. For 10 cents, he crafts a half-sheet-long tale about grandchildren, lost loves, pets, or the absurd. The clacking of keys on paper draws a curious crowd.
I derive a lot of meaning and joy from making things that other people draw joy from,
says Smith.
As a graduate student at Rochester, Smith has been finding lots of ways to share his love of storytelling. In addition to his peripatetic typewriting, he’s the leader of Rocket Radio Theater, a troupe of radio performers whose core membership includes fellow like-minded medical science graduate students Clarence Ling, Jon Baker, Carolyn Klocke, Bronwyn Lucas, and Matt Payea.
The project began in 2013 with a recording at Smith’s kitchen table. The group, which now records in the studios of campus radio station WRUR, hosts several serial drama podcasts and stand-alone stories created by Smith. Its feature series, The Bootleggers, takes place during prohibition-era Rochester, playing up aspects of local history and landscapes.
In his research as a biophysicist, Smith explores nanoporous silicon membranes in the lab of James McGrath, professor of biomedical engineering. Smith describes the membranes as coffee filters made of glass that are 10,000 times thinner than a human hair.
But he hopes to continue to combine storytelling and science after graduation, perhaps as a science journalist or a podcaster.
I want to live in a world,
he says, where people are standing on street corners writing stories.
What Frogs Can Teach Us About Tumors
Wednesday, January 13, 2016
Researchers at the University of Rochester School of Medicine and Dentistry are using frogs as a model to study human diseases. These frogs, called South African clawed frogs or Xenopus laevis, may not resemble humans on the outside, but they are very similar on a genetic level.